Search
- Page Path
- HOME > Search
Review Article
- Diabetes, obesity and metabolism
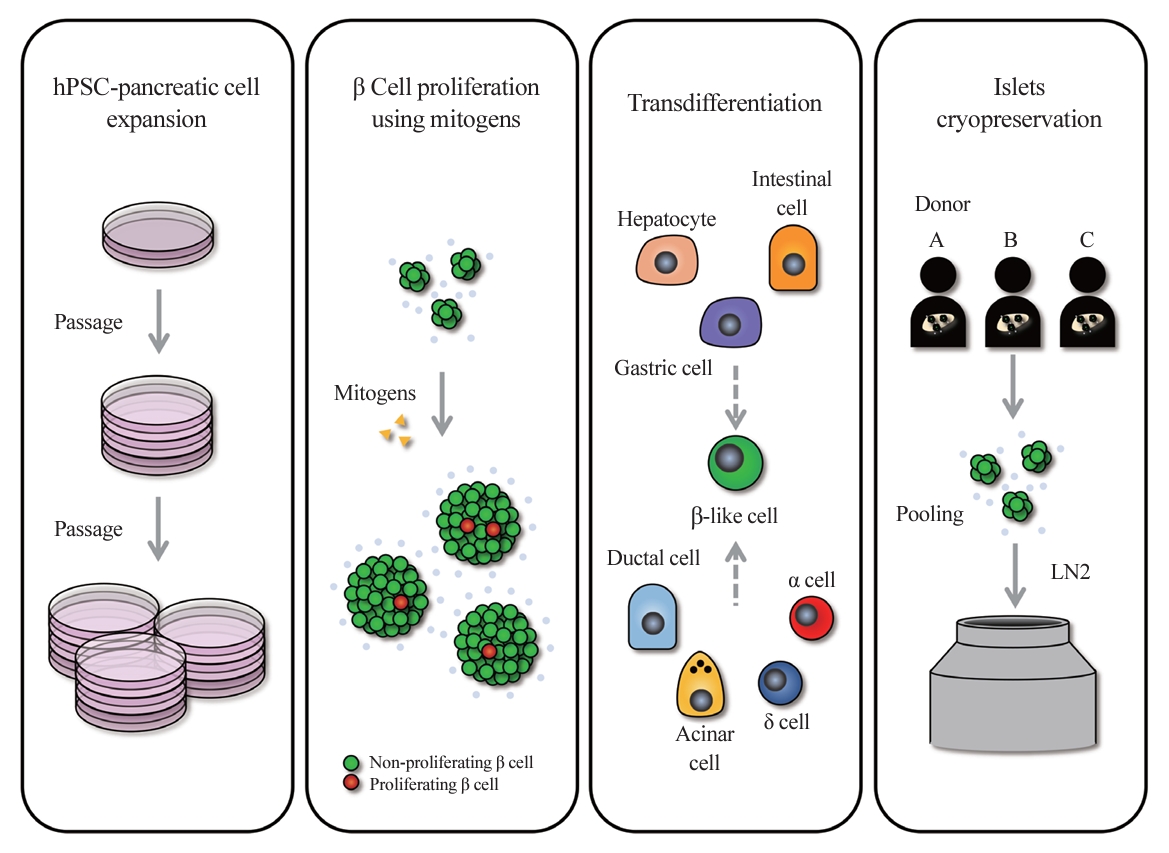
- Scaling Insulin-Producing Cells by Multiple Strategies
- Jinhyuk Choi, Fritz Cayabyab, Harvey Perez, Eiji Yoshihara
- Endocrinol Metab. 2024;39(2):191-205. Published online April 4, 2024
- DOI: https://doi.org/10.3803/EnM.2023.1910
- 1,616 View
- 82 Download
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - In the quest to combat insulin-dependent diabetes mellitus (IDDM), allogenic pancreatic islet cell therapy sourced from deceased donors represents a significant therapeutic advance. However, the applicability of this approach is hampered by donor scarcity and the demand for sustained immunosuppression. Human induced pluripotent stem cells are a game-changing resource for generating synthetic functional insulin-producing β cells. In addition, novel methodologies allow the direct expansion of pancreatic progenitors and mature β cells, thereby circumventing prolonged differentiation. Nevertheless, achieving practical reproducibility and scalability presents a substantial challenge for this technology. As these innovative approaches become more prominent, it is crucial to thoroughly evaluate existing expansion techniques with an emphasis on their optimization and scalability. This manuscript delineates these cutting-edge advancements, offers a critical analysis of the prevailing strategies, and underscores pivotal challenges, including cost-efficiency and logistical issues. Our insights provide a roadmap, elucidating both the promises and the imperatives in harnessing the potential of these cellular therapies for IDDM.

Original Article
- Standardization of Isolation Procedure and Analysis of Variables on Successful Isolation of Islet from the Human Pancreas.
- Song Cheol Kim, Duck Jong Han, Ik Hee Kim, Yoo Me We, Yang Hee Kim, Jin Hee Kim, Ji He Back, Dong Gyun Lim
- J Korean Endocr Soc. 2006;21(1):22-31. Published online February 1, 2006
- DOI: https://doi.org/10.3803/jkes.2006.21.1.22
- 1,628 View
- 19 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Identifying the donor and isolation-related factors during the islet isolation would be greatly helpful to improve the result of human islet isolation for successful clinical islet transplantation. METHODS: Sixty-nine pancreata from cadaveric donors were isolated with standard protocol and analyzed to identify the donor factors and isolation variables for successful isolation. Islet isolations recovered > or = 100,000 Islet Equivalent (IEQ, n=53) were compared to islet mass less than 100,000 IEQ (n=16). RESULTS: The mean islet recovery was 216.0 x 10(3) +/- 173.7 x 10(3) (IEQ) before purification and 130.6 x 10(3) +/- 140.2 x 10(3) (IEQ) after purification. Mean purity was 54 +/- 31%. Mean age of donor was 31.2 +/- 13.2 year and mean cold ischemic time was 6.9 +/- 6.2 hour. Quality of isolated islets was acceptable in terms of bacterial culture, viability and secretory function in vitro and in vivo. In univariate analysis on successful isolation, status of pancreas was the only significant factor and sex, duration of collagenase expansion and digestion time were marginal factors. Stepwise multivariate logistic regression analysis showed donor sex, status of pancreas and digestion time were significant factors for the successful islet isolation. CONCLUSION: This study confirms some donor factors and variables in isolation process can influence the ability to obtain the successful isolation of human islet. Enough experiences and pertinent review of donor and isolation factors can make islet isolation successful, supporting the clinical islet transplantation without spending of cost.


 KES
KES
 First
First Prev
Prev



