Search
- Page Path
- HOME > Search
Original Articles
- Hypothalamus and pituitary gland
- Clinical Characteristics, Diagnosis, and Treatment of Thyroid Stimulating Hormone-Secreting Pituitary Neuroendocrine Tumor (TSH PitNET): A Single-Center Experience
- Jung Heo, Yeon-Lim Suh, Se Hoon Kim, Doo-Sik Kong, Do-Hyun Nam, Won-Jae Lee, Sung Tae Kim, Sang Duk Hong, Sujin Ryu, You-Bin Lee, Gyuri Kim, Sang-Man Jin, Jae Hyeon Kim, Kyu Yeon Hur
- Endocrinol Metab. 2024;39(2):387-396. Published online February 5, 2024
- DOI: https://doi.org/10.3803/EnM.2023.1877
- 1,486 View
- 43 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
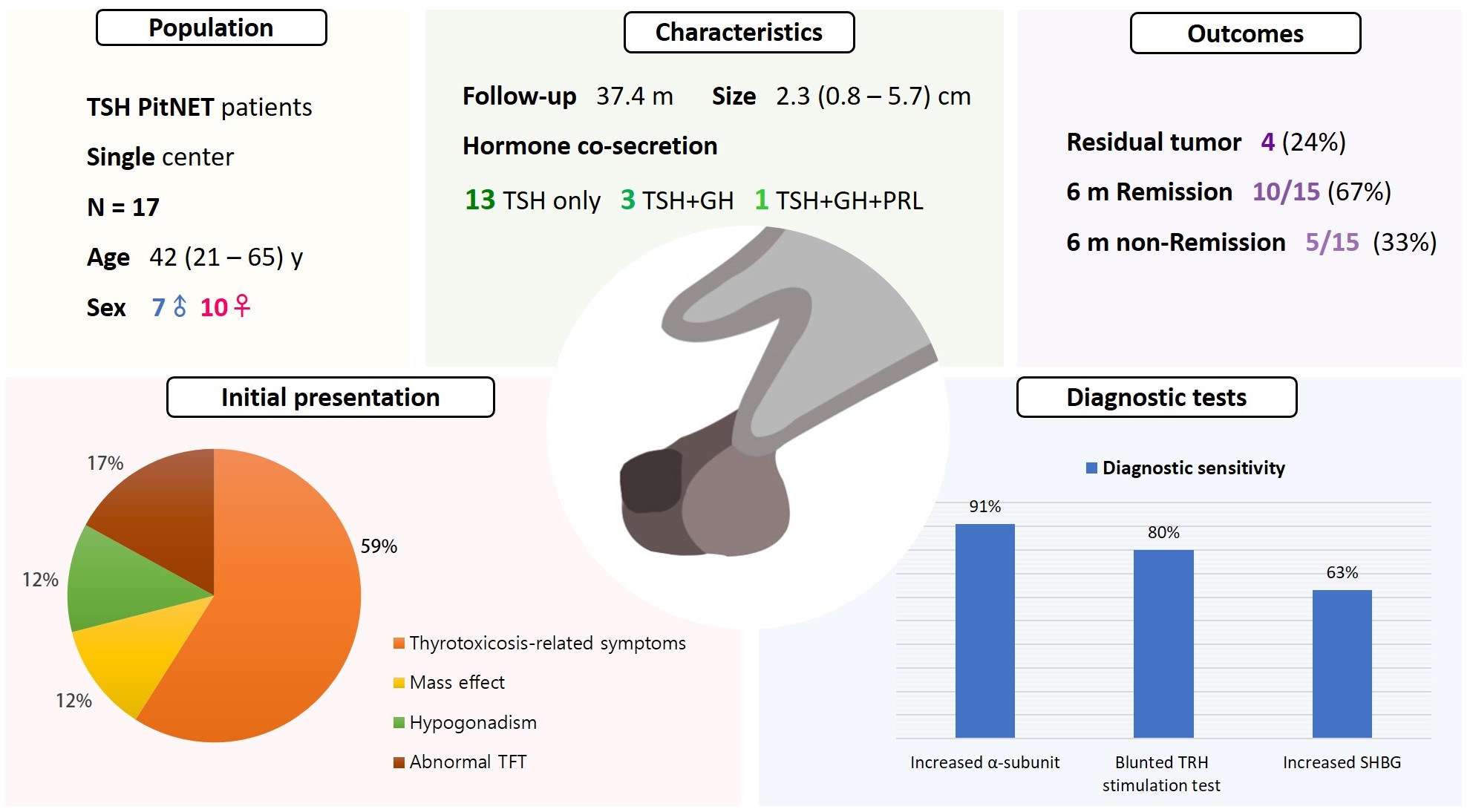
Thyroid-stimulating hormone (TSH)-secreting pituitary neuroendocrine tumor (TSH PitNET) is a rare subtype of PitNET. We investigated the comprehensive characteristics and outcomes of TSH PitNET cases from a single medical center. Also, we compared diagnostic methods to determine which showed superior sensitivity.
Methods
A total of 17 patients diagnosed with TSH PitNET after surgery between 2002 and 2022 in Samsung Medical Center was retrospectively reviewed. Data on comprehensive characteristics and treatment outcomes were collected. The sensitivities of diagnostic methods were compared.
Results
Seven were male (41%), and the median age at diagnosis was 42 years (range, 21 to 65); the median follow-up duration was 37.4 months. The most common (59%) initial presentation was hyperthyroidism-related symptoms. Hormonal co-secretion was present in four (23%) patients. Elevated serum alpha-subunit (α-SU) showed the greatest diagnostic sensitivity (91%), followed by blunted response at thyrotropin-releasing hormone (TRH) stimulation (80%) and elevated sex hormone binding globulin (63%). Fourteen (82%) patients had macroadenoma, and a specimen of one patient with heavy calcification was negative for TSH. Among 15 patients who were followed up for more than 6 months, 10 (67%) achieved hormonal and structural remission within 6 months postoperatively. A case of growth hormone (GH)/TSH/prolactin (PRL) co-secreting mixed gangliocytoma-pituitary adenoma (MGPA) was discovered.
Conclusion
The majority of the TSH PitNET cases was macroadenoma, and 23% showed hormone co-secretion. A rare case of GH/TSH/PRL co-secreting MGPA was discovered. Serum α-SU and TRH stimulation tests showed great diagnostic sensitivity. Careful consideration is needed in diagnosing TSH PitNET. Achieving remission requires complete tumor resection. In case of nonremission, radiotherapy or medical therapy can improve the long-term remission rate.

- Hypothalamus and pituitary gland
- Preoperative Serum Copeptin Can Predict Delayed Hyponatremia after Pituitary Surgery in the Absence of Arginine Vasopressin Deficiency
- Ho Kang, Seung Shin Park, Yoo Hyung Kim, Hwan Sub Lim, Mi-Kyeong Lee, Kyoung-Ryul Lee, Jung Hee Kim, Yong Hwy Kim
- Endocrinol Metab. 2024;39(1):164-175. Published online January 3, 2024
- DOI: https://doi.org/10.3803/EnM.2023.1792
- 1,079 View
- 49 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
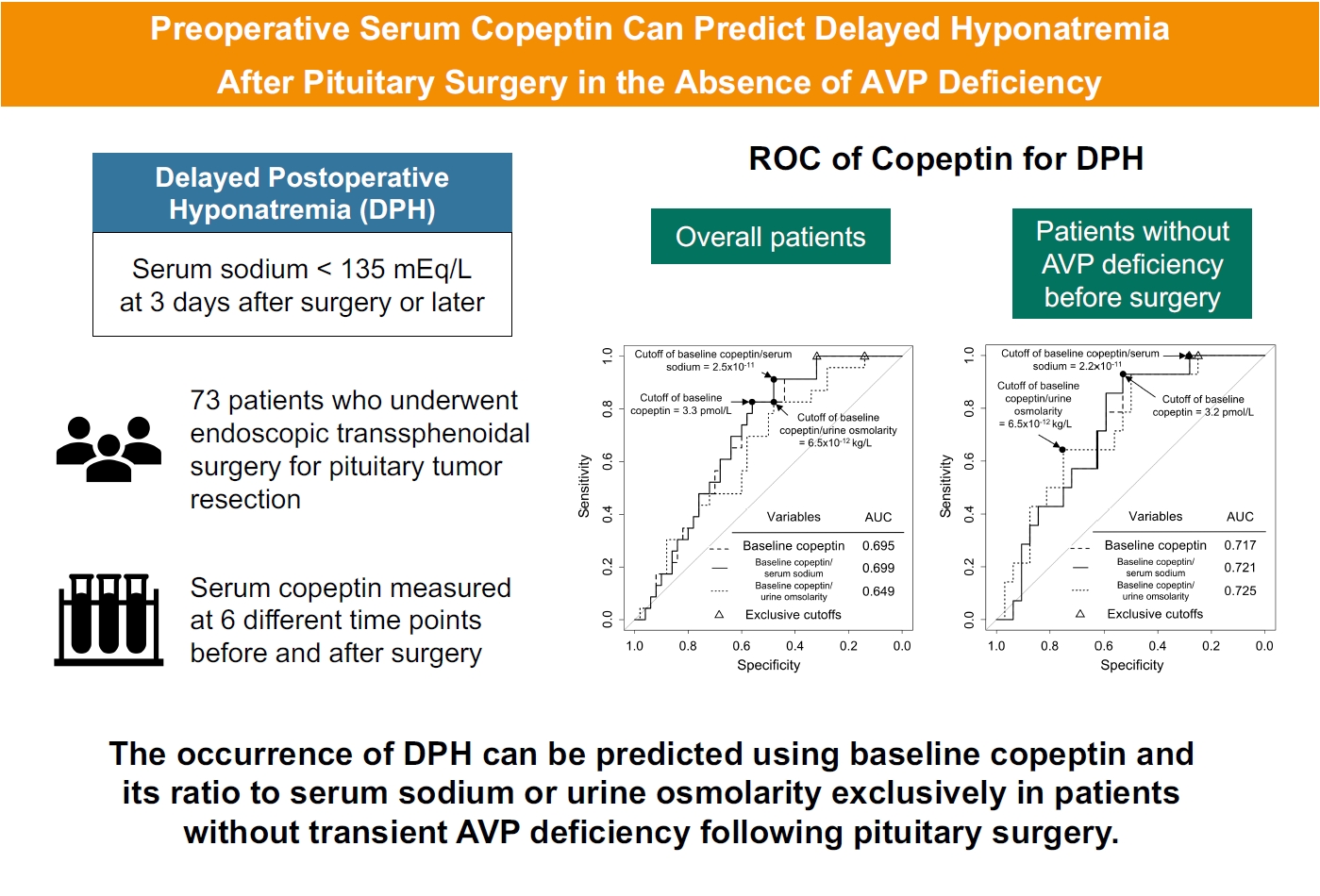
Delayed postoperative hyponatremia (DPH) is the most common cause of readmission after pituitary surgery. In this study, we aimed to evaluate the cutoff values of serum copeptin and determine the optimal timing for copeptin measurement for the prediction of the occurrence of DPH in patients who undergo endoscopic transsphenoidal approach (eTSA) surgery and tumor resection.
Methods
This was a prospective observational study of 73 patients who underwent eTSA surgery for pituitary or stalk lesions. Copeptin levels were measured before surgery, 1 hour after extubation, and on postoperative days 1, 2, 7, and 90.
Results
Among 73 patients, 23 patients (31.5%) developed DPH. The baseline ratio of copeptin to serum sodium level showed the highest predictive performance (area under the curve [AUROC], 0.699), and its optimal cutoff to maximize Youden’s index was 2.5×10–11, with a sensitivity of 91.3% and negative predictive value of 92.0%. No significant predictors were identified for patients with transient arginine vasopressin (AVP) deficiency. However, for patients without transient AVP deficiency, the copeptin-to-urine osmolarity ratio at baseline demonstrated the highest predictive performance (AUROC, 0.725). An optimal cutoff of 6.5×10–12 maximized Youden’s index, with a sensitivity of 92.9% and a negative predictive value of 94.1%.
Conclusion
The occurrence of DPH can be predicted using baseline copeptin and its ratio with serum sodium or urine osmolarity only in patients without transient AVP deficiency after pituitary surgery.

Review Article
- Hypothalamus and pituitary gland
- Update on Current Evidence for the Diagnosis and Management of Nonfunctioning Pituitary Neuroendocrine Tumors
- Elizabeth Whyte, Masahiro Nezu, Constance Chik, Toru Tateno
- Endocrinol Metab. 2023;38(6):631-654. Published online November 15, 2023
- DOI: https://doi.org/10.3803/EnM.2023.1838
- 2,438 View
- 152 Download
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Pituitary neuroendocrine tumors (PitNETs) are the third most frequently diagnosed intracranial tumors, with nonfunctioning PitNETs (nfPitNETs) accounting for 30% of all pituitary tumors and representing the most common type of macroPitNETs. NfPitNETs are usually benign tumors with no evidence of hormone oversecretion except for hyperprolactinemia secondary to pituitary stalk compression. Due to this, they do not typically present with clinical syndromes like acromegaly, Cushing’s disease or hyperthyroidism and instead are identified incidentally on imaging or from symptoms of mass effects (headache, vision changes, apoplexy). With the lack of effective medical interventions, first-line treatment is transsphenoidal surgical resection, however, nfPitNETs often have supra- or parasellar extension, and total resection of the tumor is often not possible, resulting in residual tumor regrowth or reoccurrence. While functional PitNETs can be easily followed for recurrence using hormonal biomarkers, there is no similar parameter to predict recurrence in nfPitNETs, hence delaying early recognition and timely management. Therefore, there is a need to identify prognostic biomarkers that can be used for patient surveillance and as therapeutic targets. This review focuses on summarizing the current evidence on nfPitNETs, with a special focus on potential new biomarkers and therapeutics.
-
Citations
Citations to this article as recorded by- Phenotype Transformation of PitNETs
Zhenwei Li, Yinzi Wu, Guannan He, Renzhi Wang, Xinjie Bao
Cancers.2024; 16(9): 1731. CrossRef
- Phenotype Transformation of PitNETs

Songwon Lecture 2022
- Hypothalamus and pituitary gland
- Multiomics Approach to Acromegaly: Unveiling Translational Insights for Precision Medicine
- Kyungwon Kim, Cheol Ryong Ku, Eun Jig Lee
- Endocrinol Metab. 2023;38(5):463-471. Published online October 13, 2023
- DOI: https://doi.org/10.3803/EnM.2023.1820
- 1,758 View
- 123 Download
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
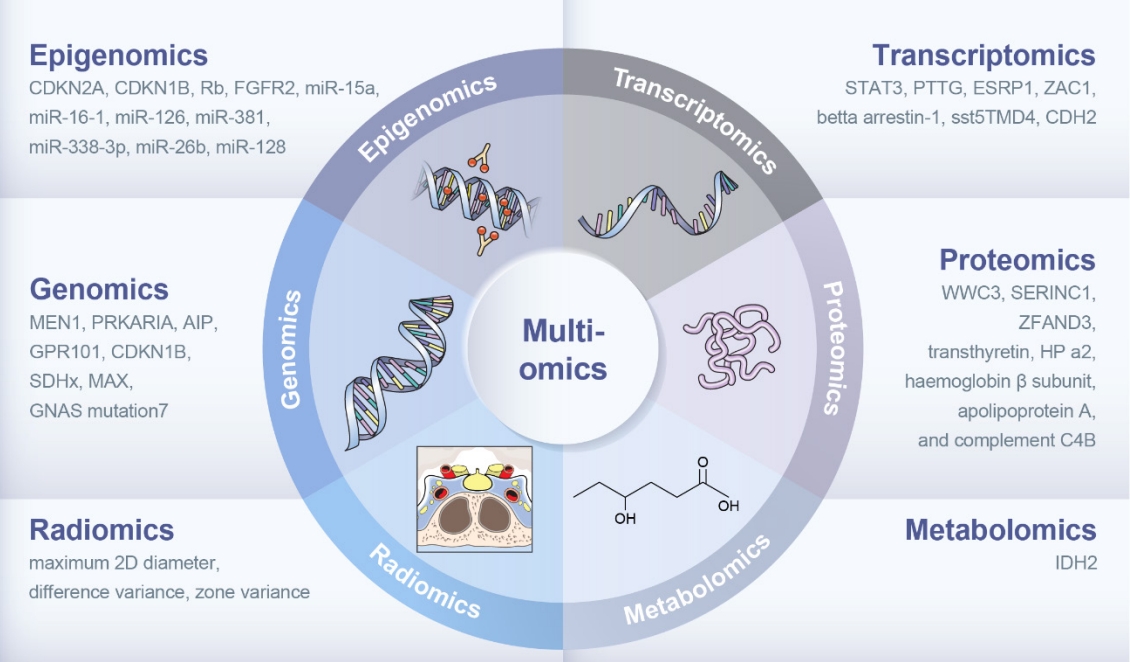
ePub - The clinical characteristics and prognoses of acromegaly vary among patients. Assessment of current and novel predictors can lead to multilevel categorization of patients, allowing integration into new clinical guidelines and a reduction in the increased morbidity and mortality associated with acromegaly. Despite advances in the diagnosis and treatment of acromegaly, its pathophysiology remains unclear. Recent advancements in multiomics technologies, including genomics, transcriptomics, proteomics, metabolomics, and radiomics, have offered new opportunities to unravel the complex pathophysiology of acromegaly. This review comprehensively explores the emerging role of multiomics approaches in elucidating the molecular landscape of acromegaly. We discuss the potential implications of multiomics data integration in the development of novel diagnostic tools, identification of therapeutic targets, and the prospects of precision medicine in acromegaly management. By integrating diverse omics datasets, these approaches can provide valuable insights into disease mechanisms, facilitate the identification of diagnostic biomarkers, and identify potential therapeutic targets for precision medicine in the management of acromegaly.
-
Citations
Citations to this article as recorded by- “Micromegaly”: Acromegaly with apparently normal GH, an entity on its own?
Lucio Vilar, Luciana Ansaneli Naves, Manoel Ricardo Alves Martins, Antônio Ribeiro-Oliveira Jr
Best Practice & Research Clinical Endocrinology & Metabolism.2024; : 101878. CrossRef
- “Micromegaly”: Acromegaly with apparently normal GH, an entity on its own?

Special Article
- Miscellaneous
- Immune Checkpoint Inhibitors and Endocrine Disorders: A Position Statement from the Korean Endocrine Society
- Hyemi Kwon, Eun Roh, Chang Ho Ahn, Hee Kyung Kim, Cheol Ryong Ku, Kyong Yeun Jung, Ju Hee Lee, Eun Heui Kim, Sunghwan Suh, Sangmo Hong, Jeonghoon Ha, Jun Sung Moon, Jin Hwa Kim, Mi-kyung Kim, The Committee of Clinical Practice Guideline of the Korean Endocrine Society
- Endocrinol Metab. 2022;37(6):839-850. Published online December 26, 2022
- DOI: https://doi.org/10.3803/EnM.2022.1627

- 3,579 View
- 322 Download
- 2 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Immune checkpoint inhibitors (ICIs) including an anti-cytotoxic T-lymphocyte-associated antigen 4 inhibitor, anti-programmed cell death protein 1 (PD-1) inhibitors, and anti-PD-ligand 1 inhibitors are representative therapeutics for various malignancies. In oncology, the application of ICIs is currently expanding to a wider range of malignancies due to their remarkable clinical outcomes. ICIs target immune checkpoints which suppress the activity of T-cells that are specific for tumor antigens, thereby allowing tumor cells to escape the immune response. However, immune checkpoints also play a crucial role in preventing autoimmune reactions. Therefore, ICIs targeting immune checkpoints can trigger various immune-related adverse events (irAEs), especially in endocrine organs. Considering the endocrine organs that are frequently involved, irAEs associated endocrinopathies are frequently life-threatening and have unfavorable clinical implications for patients. However, there are very limited data from large clinical trials that would inform the development of clinical guidelines for patients with irAEs associated endocrinopathies. Considering the current clinical situation, in which the scope and scale of the application of ICIs are increasing, position statements from clinical specialists play an essential role in providing the appropriate recommendations based on both medical evidence and clinical experience. As endocrinologists, we would like to present precautions and recommendations for the management of immune-related endocrine disorders, especially those involving the adrenal, thyroid, and pituitary glands caused by ICIs.
-
Citations
Citations to this article as recorded by- Pembrolizumab plus lenvatinib for radically unresectable or metastatic renal cell carcinoma in the Japanese population
Ryo Fujiwara, Takeshi yuasa, kenichi kobayashi, tetsuya yoshida, susumu kageyama
Expert Review of Anticancer Therapy.2023; 23(5): 461. CrossRef - Incidence of Endocrine-Related Dysfunction in Patients Treated with New Immune Checkpoint Inhibitors: A Meta-Analysis and Comprehensive Review
Won Sang Yoo, Eu Jeong Ku, Eun Kyung Lee, Hwa Young Ahn
Endocrinology and Metabolism.2023; 38(6): 750. CrossRef
- Pembrolizumab plus lenvatinib for radically unresectable or metastatic renal cell carcinoma in the Japanese population

Review Articles
- Hypothalamus and Pituitary Gland
- Independent Skeletal Actions of Pituitary Hormones
- Se-Min Kim, Farhath Sultana, Funda Korkmaz, Daria Lizneva, Tony Yuen, Mone Zaidi
- Endocrinol Metab. 2022;37(5):719-731. Published online September 28, 2022
- DOI: https://doi.org/10.3803/EnM.2022.1573
- 3,772 View
- 236 Download
- 4 Web of Science
- 4 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
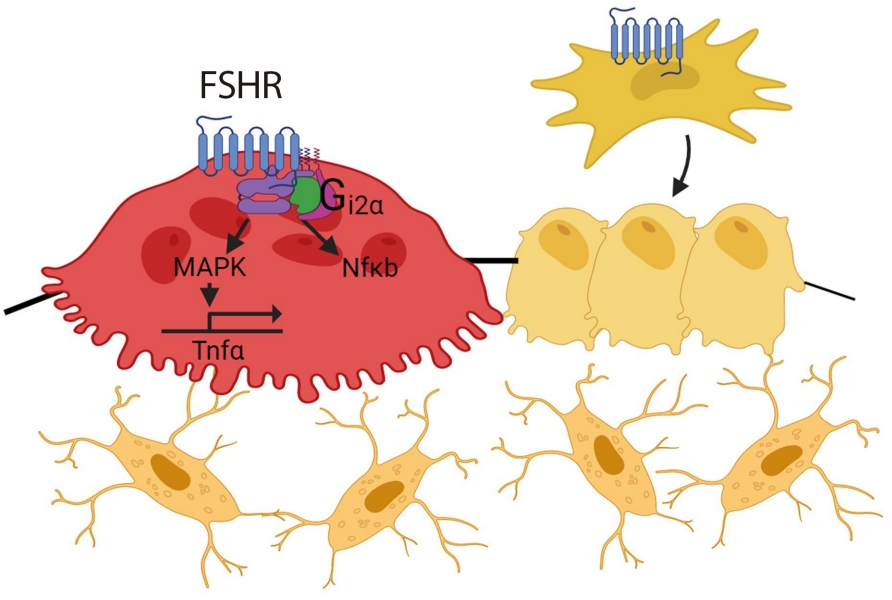
ePub - Over the past years, pituitary hormones and their receptors have been shown to have non-traditional actions that allow them to bypass the hypothalamus-pituitary-effector glands axis. Bone cells—osteoblasts and osteoclasts—express receptors for growth hormone, follicle stimulating hormone (FSH), thyroid stimulating hormone (TSH), adrenocorticotrophic hormone (ACTH), prolactin, oxytocin, and vasopressin. Independent skeletal actions of pituitary hormones on bone have been studied using genetically modified mice with haploinsufficiency and by activating or inactivating the receptors pharmacologically, without altering systemic effector hormone levels. On another front, the discovery of a TSH variant (TSH-βv) in immune cells in the bone marrow and skeletal action of FSHβ through tumor necrosis factor α provides new insights underscoring the integrated physiology of bone-immune-endocrine axis. Here we discuss the interaction of each pituitary hormone with bone and the potential it holds in understanding bone physiology and as a therapeutic target.
-
Citations
Citations to this article as recorded by- New tools for bone health assessment in secreting pituitary adenomas
Meliha Melin Uygur, Stefano Frara, Luigi di Filippo, Andrea Giustina
Trends in Endocrinology & Metabolism.2023; 34(4): 231. CrossRef - A Causality between Thyroid Function and Bone Mineral Density in Childhood: Abnormal Thyrotropin May Be Another Pediatric Predictor of Bone Fragility
Dongjin Lee, Moon Ahn
Metabolites.2023; 13(3): 372. CrossRef - The mechanism of oxytocin and its receptors in regulating cells in bone metabolism
Liu Feixiang, Feng Yanchen, Li Xiang, Zhang Yunke, Miao Jinxin, Wang Jianru, Lin Zixuan
Frontiers in Pharmacology.2023;[Epub] CrossRef - To investigate the mechanism of Yiwei Decoction in the treatment of premature ovarian insufficiency-related osteoporosis using transcriptomics, network pharmacology and molecular docking techniques
Weisen Fan, Yan Meng, Jing Zhang, Muzhen Li, Yingjie Zhang, Xintian Qu, Xin Xiu
Scientific Reports.2023;[Epub] CrossRef
- New tools for bone health assessment in secreting pituitary adenomas

- Hypothalamus and Pituitary Gland
- Advances in Pituitary Surgery
- Yoon Hwan Byun, Ho Kang, Yong Hwy Kim
- Endocrinol Metab. 2022;37(4):608-616. Published online August 19, 2022
- DOI: https://doi.org/10.3803/EnM.2022.1546
- 3,280 View
- 156 Download
- 2 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
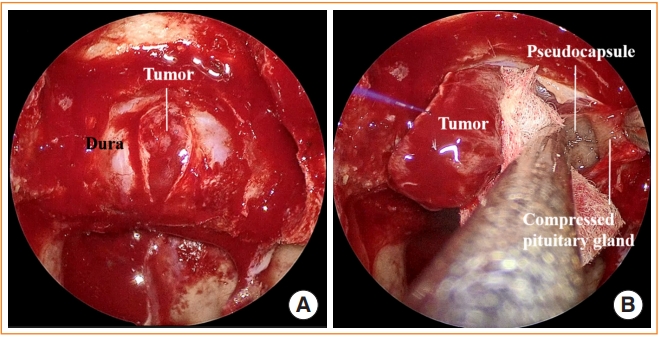
ePub - Pituitary surgery has advanced considerably in recent years with the exploration and development of various endoscopic approaches and techniques. Different endoscopic skull base approaches are being applied to access sellar tumors in different locations. Moreover, extracapsular dissection and cavernous sinus exploration have enabled gross total resection of sellar tumors where it could not have been achieved in the past. Techniques for skull base reconstruction have also progressed, allowing surgeons to remove larger and more complicated tumors than before. This review article discusses different endoscopic skull base approaches, surgical techniques for removing pituitary adenomas, and reconstruction methods for repairing postoperative low-flow and high-flow cerebrospinal fluid leakage.
-
Citations
Citations to this article as recorded by- Serum and hair steroid profiles in patients with nonfunctioning pituitary adenoma undergoing surgery: A prospective observational study
Seung Shin Park, Yong Hwy Kim, Ho Kang, Chang Ho Ahn, Dong Jun Byun, Man Ho Choi, Jung Hee Kim
The Journal of Steroid Biochemistry and Molecular Biology.2023; 230: 106276. CrossRef - Pituitary disease and anaesthesia
Kim Rhodes, Robert John, Astri Luoma
Anaesthesia & Intensive Care Medicine.2023; 24(6): 348. CrossRef - Орфанні ендокринні захворювання: сучасні тенденції хірургічного лікування в Україні
M.D. Tronko, B.B. Guda
Endokrynologia.2022; 27(4): 287. CrossRef
- Serum and hair steroid profiles in patients with nonfunctioning pituitary adenoma undergoing surgery: A prospective observational study

Original Article
- Hypothalamus and Pituitary Gland
- Metabolic Impacts of Discontinuation and Resumption of Recombinant Human Growth Hormone Treatment during the Transition Period in Patients with Childhood-Onset Growth Hormone Deficiency
- Yun Jeong Lee, Yunha Choi, Han-Wook Yoo, Young Ah Lee, Choong Ho Shin, Han Saem Choi, Ho-Seong Kim, Jae Hyun Kim, Jung Eun Moon, Cheol Woo Ko, Moon Bae Ahn, Byung-Kyu Suh, Jin-Ho Choi
- Endocrinol Metab. 2022;37(2):359-368. Published online April 25, 2022
- DOI: https://doi.org/10.3803/EnM.2021.1384
- 4,489 View
- 185 Download
- 3 Web of Science
- 4 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Discontinuing growth hormone (GH) treatment during the transition to adulthood has been associated with adverse health outcomes in patients with childhood-onset growth hormone deficiency (CO-GHD). This study investigated the metabolic changes associated with interrupting GH treatment in adolescents with CO-GHD during the transition period.
Methods
This study included 187 patients with CO-GHD who were confirmed to have adult GHD and were treated at six academic centers in Korea. Data on clinical parameters, including anthropometric measurements, metabolic profiles, and bone mineral density (BMD) at the end of childhood GH treatment, were collected at the time of re-evaluation for GHD and 1 year after treatment resumption.
Results
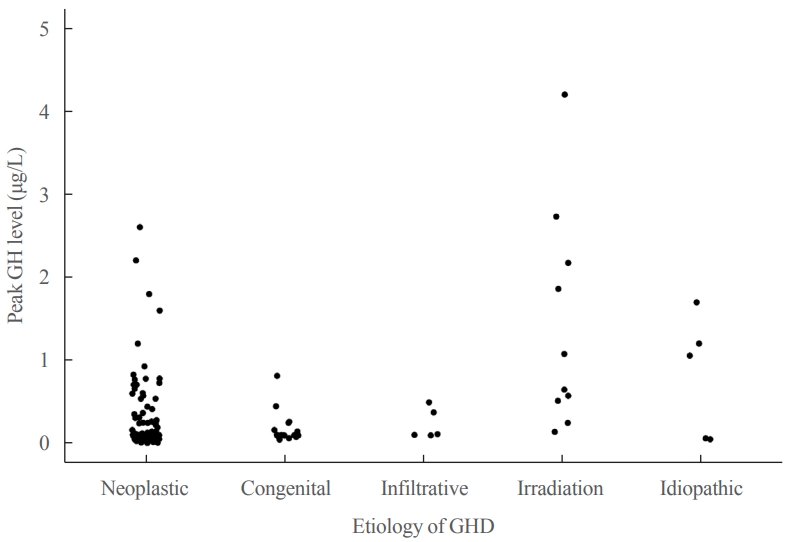
Most patients (n=182, 97.3%) had organic GHD. The median age at treatment discontinuation and re-evaluation was 15.6 and 18.7 years, respectively. The median duration of treatment interruption was 2.8 years. During treatment discontinuation, body mass index Z-scores and total cholesterol, low-density lipoprotein, and non-high-density lipoprotein (HDL) cholesterol levels increased, whereas fasting glucose levels decreased. One year after GH treatment resumption, fasting glucose levels, HDL cholesterol levels, and femoral neck BMD increased significantly. Longer GH interruption (>2 years, 60.4%) resulted in worse lipid profiles at re-evaluation. The duration of interruption was positively correlated with fasting glucose and non-HDL cholesterol levels after adjusting for covariates.
Conclusion
GH treatment interruption during the transition period resulted in worse metabolic parameters, and a longer interruption period was correlated with poorer outcomes. GH treatment should be resumed early in patients with CO-GHD during the transition period. -
Citations
Citations to this article as recorded by- Ghrelin regulating liver activity and its potential effects on liver fibrosis and Echinococcosis
Jiang Zhu, Tanfang Zhou, Meng Menggen, Kalibixiati Aimulajiang, Hao Wen
Frontiers in Cellular and Infection Microbiology.2024;[Epub] CrossRef - Composición de la microbiota en pacientes con déficit de hormona de crecimiento antes y después de recibir tratamiento
Patricia García Navas, María Yolanda Ruíz del Prado, Pablo Villoslada Blanco, Emma Recio Fernández, María Ruíz del Campo, Patricia Pérez Matute
Anales de Pediatría.2024;[Epub] CrossRef - Relationship between the Stimulated Peak Growth Hormone Level and Metabolic Parameters in Children with Growth Hormone Deficiency
Seong Yong Lee
The Ewha Medical Journal.2023;[Epub] CrossRef - Dyslipidaemia and growth hormone deficiency – A comprehensive review
Matthias Hepprich, Fahim Ebrahimi, Emanuel Christ
Best Practice & Research Clinical Endocrinology & Metabolism.2023; 37(6): 101821. CrossRef
- Ghrelin regulating liver activity and its potential effects on liver fibrosis and Echinococcosis

Special Article
- Miscellaneous
- COVID-19 Vaccination for Endocrine Patients: A Position Statement from the Korean Endocrine Society
- Cheol Ryong Ku, Kyong Yeun Jung, Chang Ho Ahn, Jun Sung Moon, Ju Hee Lee, Eun Heui Kim, Hyemi Kwon, Hee Kyung Kim, Sunghwan Suh, Sangmo Hong, Jeonghoon Ha, Eun Roh, Jin Hwa Kim, Mi-kyung Kim, the Committee of Clinical Practice Guideline of the Korean Endocrine Society
- Endocrinol Metab. 2021;36(4):757-765. Published online August 17, 2021
- DOI: https://doi.org/10.3803/EnM.2021.404
- 10,444 View
- 419 Download
- 19 Web of Science
- 21 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Since the first outbreak of coronavirus disease 2019 (COVID-19), ongoing efforts have been made to discover an efficacious vaccine against COVID-19 to combat the pandemic. In most countries, both mRNA and DNA vaccines have been administered, and their side effects have also been reported. The clinical course of COVID-19 and the effects of vaccination against COVID-19 are both influenced by patients’ health status and involve a systemic physiological response. In view of the systemic function of endocrine hormones, endocrine disorders themselves and the therapeutics used to treat them can influence the outcomes of vaccination for COVID-19. However, there are very limited data to support the development of clinical guidelines for patients with specific medical backgrounds based on large clinical trials. In the current severe circumstances of the COVID-19 pandemic, position statements made by clinical specialists are essential to provide appropriate recommendations based on both medical evidence and clinical experiences. As endocrinologists, we would like to present the medical background of COVID-19 vaccination, as well as precautions to prevent the side effects of COVID-19 vaccination in patients with specific endocrine disorders, including adrenal insufficiency, diabetes mellitus, osteoporosis, autoimmune thyroid disease, hypogonadism, and pituitary disorders.
-
Citations
Citations to this article as recorded by- COVID-19 mRNA vaccine may trigger subacute thyroiditis
Mehmet Sözen, Ömercan Topaloğlu, Berrin Çetinarslan, Alev Selek, Zeynep Cantürk, Emre Gezer, Damla Köksalan, Taner Bayraktaroğlu
Human Vaccines & Immunotherapeutics.2024; 17(12): 5120. CrossRef - The role of co-morbidities in the development of an AEFI after COVID-19 vaccination in a large prospective cohort with patient-reported outcomes in the Netherlands
C. Ouaddouh, J.W. Duijster, T. Lieber, F.P.A.M. van Hunsel
Expert Opinion on Drug Safety.2024; 23(3): 323. CrossRef - Thyroid dysfunction in COVID-19
David Tak Wai Lui, Chi Ho Lee, Yu Cho Woo, Ivan Fan Ngai Hung, Karen Siu Ling Lam
Nature Reviews Endocrinology.2024;[Epub] CrossRef - Adult-Onset Type 1 Diabetes Development Following COVID-19 mRNA Vaccination
Hyeyeon Moon, Sunghwan Suh, Mi Kyoung Park
Journal of Korean Medical Science.2023;[Epub] CrossRef - Prior immunization status of COVID-19 patients and disease severity: A multicenter retrospective cohort study assessing the different types of immunity
Javaria Aslam, Faisal Shahzad Khan, Muhammad Talha Haris, Hewad Hewadmal, Maryam Khalid, Mohammad Y. Alshahrani, Qurrat-ul-ain Aslam, Irrum Aneela, Urooj Zafar
Vaccine.2023; 41(2): 598. CrossRef - Mortality and Severity of Coronavirus Disease 2019 in Patients with Long-Term Glucocorticoid Therapy: A Korean Nationwide Cohort Study
Eu Jeong Ku, Keeho Song, Kyoung Min Kim, Gi Hyeon Seo, Soon Jib Yoo
Endocrinology and Metabolism.2023; 38(2): 253. CrossRef - Pituitary Diseases and COVID-19 Outcomes in South Korea: A Nationwide Cohort Study
Jeonghoon Ha, Kyoung Min Kim, Dong-Jun Lim, Keeho Song, Gi Hyeon Seo
Journal of Clinical Medicine.2023; 12(14): 4799. CrossRef - Inactivated SARS-CoV-2 vaccination does not disturb the clinical course of Graves’ disease: An observational cohort study
Shichen Xu, Huixin Yu, Xian Cheng, Jing Wu, Jiandong Bao, Li Zhang
Vaccine.2023; 41(38): 5648. CrossRef - Adrenal Crisis Associated With COVID-19 Vaccination in Patients With Adrenal Insufficiency
Yukako Kurematsu, Takako Mohri, Sadanori Okada, Yutaka Takahashi
JCEM Case Reports.2023;[Epub] CrossRef - Adverse Events Associated with COVID-19 Vaccination in Adolescents with Endocrinological Disorders: A Cross-Sectional Study
İbrahim Mert Erbaş, İrem Ceren Erbaş, Gözde Akın Kağızmanlı, Kübra Yüksek Acinikli, Özge Besci, Korcan Demir, Ece Böber, Nurşen Belet, Ayhan Abacı
Journal of Clinical Research in Pediatric Endocrinology.2023; 15(3): 248. CrossRef - Neue Aspekte der Glukokortikoidsubstitution bei Nebennierenrindeninsuffizienz
Tina Kienitz, Gesine Meyer
Der Internist.2022; 63(1): 12. CrossRef - Endocrine Follow-up During Post-Acute COVID-19: Practical Recommendations Based on Available Clinical Evidence
Rimesh Pal, Ameya Joshi, Sanjay K. Bhadada, Mainak Banerjee, Suresh Vaikkakara, Satinath Mukhopadhyay
Endocrine Practice.2022; 28(4): 425. CrossRef - Safety of Inactivated and mRNA COVID-19 Vaccination Among Patients Treated for Hypothyroidism: A Population-Based Cohort Study
Xi Xiong, Carlos King Ho Wong, Ivan Chi Ho Au, Francisco Tsz Tsun Lai, Xue Li, Eric Yuk Fai Wan, Celine Sze Ling Chui, Esther Wai Yin Chan, Franco Wing Tak Cheng, Kristy Tsz Kwan Lau, Chi Ho Lee, Yu Cho Woo, David Tak Wai Lui, Ian Chi Kei Wong
Thyroid.2022; 32(5): 505. CrossRef - The New Entity of Subacute Thyroiditis amid the COVID-19 Pandemic: From Infection to Vaccine
Mihaela Popescu, Adina Ghemigian, Corina Maria Vasile, Andrei Costache, Mara Carsote, Alice Elena Ghenea
Diagnostics.2022; 12(4): 960. CrossRef - Adrenal Crisis Secondary to COVID-19 Vaccination in a Patient With Hypopituitarism
Nikolina Markovic, Anila Faizan, Chirag Boradia, Sridhar Nambi
AACE Clinical Case Reports.2022; 8(4): 171. CrossRef - The Effect of Inactivated SARS-CoV-2 Vaccines on TRAB in Graves’ Disease
LingHong Huang, ZhengRong Jiang, JingXiong Zhou, YuPing Chen, HuiBin Huang
Frontiers in Endocrinology.2022;[Epub] CrossRef - Osteoporosis in Patients With Respiratory Diseases
Yue Ma, Shui Qiu, Renyi Zhou
Frontiers in Physiology.2022;[Epub] CrossRef - Pilot Findings on SARS-CoV-2 Vaccine-Induced Pituitary Diseases: A Mini Review from Diagnosis to Pathophysiology
Ach Taieb, El Euch Mounira
Vaccines.2022; 10(12): 2004. CrossRef - Forty Years Together, New Leap Forward! The 40th Anniversary of the Korean Endocrine Society
Jong Chul Won, Ki-Hyun Baek
Endocrinology and Metabolism.2022; 37(6): 851. CrossRef - No need of glucocorticoid dose adjustment in patients with adrenal insufficiency before COVID-19 vaccine
Tania Pilli, Cristina Dalmiglio, Gilda Dalmazio, Alfonso Sagnella, Raffaella Forleo, Lucia Brilli, Fabio Maino, Cristina Ciuoli, Maria Grazia Castagna
European Journal of Endocrinology.2022; 187(1): K7. CrossRef - Diabetes and COVID-19 Vaccination
Hae Dong Choi, Jun Sung Moon
The Journal of Korean Diabetes.2021; 22(4): 221. CrossRef
- COVID-19 mRNA vaccine may trigger subacute thyroiditis

Original Articles
- Hypothalamus and Pituitary Gland
Big Data Articles (National Health Insurance Service Database) - Descriptive Epidemiology and Survival Analysis of Prolactinomas and Cushing’s Disease in Korea
- Jin Sun Park, Soo Jin Yun, Jung Kuk Lee, So Young Park, Sang Ouk Chin
- Endocrinol Metab. 2021;36(3):688-696. Published online June 28, 2021
- DOI: https://doi.org/10.3803/EnM.2021.1000
- 4,766 View
- 136 Download
- 3 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
Only a few studies have established the epidemiology of prolactinoma and Cushing’s disease in Korea. Furthermore, the incidence of these disease are increasing than before associated with the development of technologies. This study was designed to evaluate the epidemiology of prolactinoma and Cushing’s disease and their survival analysis according to treatment.
Methods
The nationwide, population-based study evaluated incidence and prevalence of prolactinoma and Cushing’s disease using de-identified claims data in The Korean Health Insurance Review and Assessment Service database between 2013 and 2017. The survival analysis investigated regarding treatment over a period of 6 years. A log-rank test and Cox proportional hazard regression analysis were used.
Results
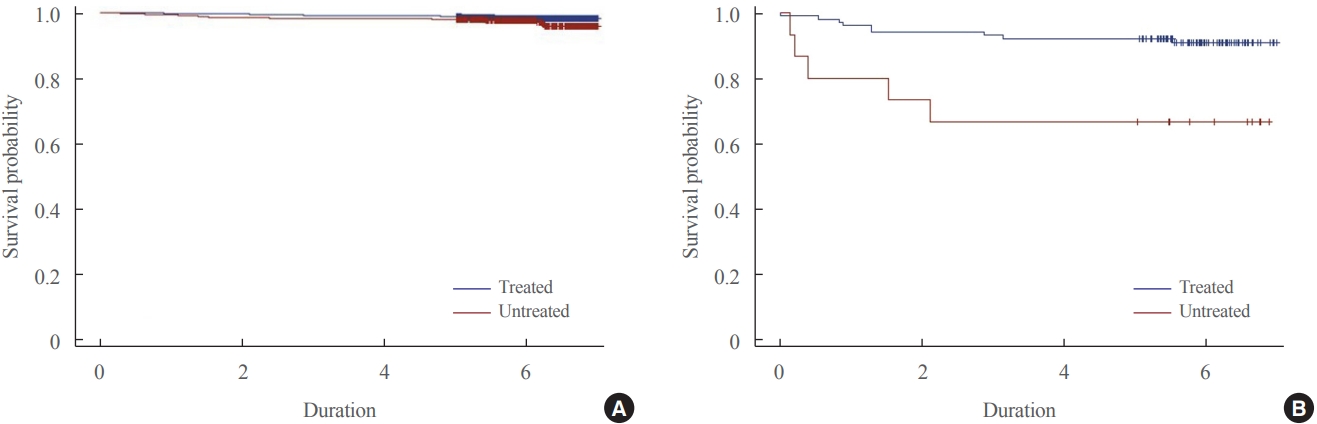
The 6,056 patients with newly diagnosed prolactinoma and 584 patients with Cushing’s disease were recorded between 2013 and 2017. The annual incidence of prolactinoma was 23.5 cases per million, and its prevalence was 82.5 cases per million, and 2.3 cases per million/year and 9.8 cases per million for Cushing’s disease. The survival benefit was insignificant in prolactinoma according to treatment, but treatment of Cushing’s disease ameliorated the survival rate significantly.
Conclusion
Overall, the incidence of prolactinoma and Cushing’s disease was similar with those found previously, but the prevalence of two diseases were inconsistent when compared with the early studies. The present study also proposed necessity of treatment in Cushing’s disease for improving the survival rate. -
Citations
Citations to this article as recorded by- Big Data Research in the Field of Endocrine Diseases Using the Korean National Health Information Database
Sun Wook Cho, Jung Hee Kim, Han Seok Choi, Hwa Young Ahn, Mee Kyoung Kim, Eun Jung Rhee
Endocrinology and Metabolism.2023; 38(1): 10. CrossRef - Cushing Syndrome
Martin Reincke, Maria Fleseriu
JAMA.2023; 330(2): 170. CrossRef - Clinical Biology of the Pituitary Adenoma
Shlomo Melmed, Ursula B Kaiser, M Beatriz Lopes, Jerome Bertherat, Luis V Syro, Gerald Raverot, Martin Reincke, Gudmundur Johannsson, Albert Beckers, Maria Fleseriu, Andrea Giustina, John A H Wass, Ken K Y Ho
Endocrine Reviews.2022; 43(6): 1003. CrossRef
- Big Data Research in the Field of Endocrine Diseases Using the Korean National Health Information Database

- Hypothalamus and Pituitary Gland
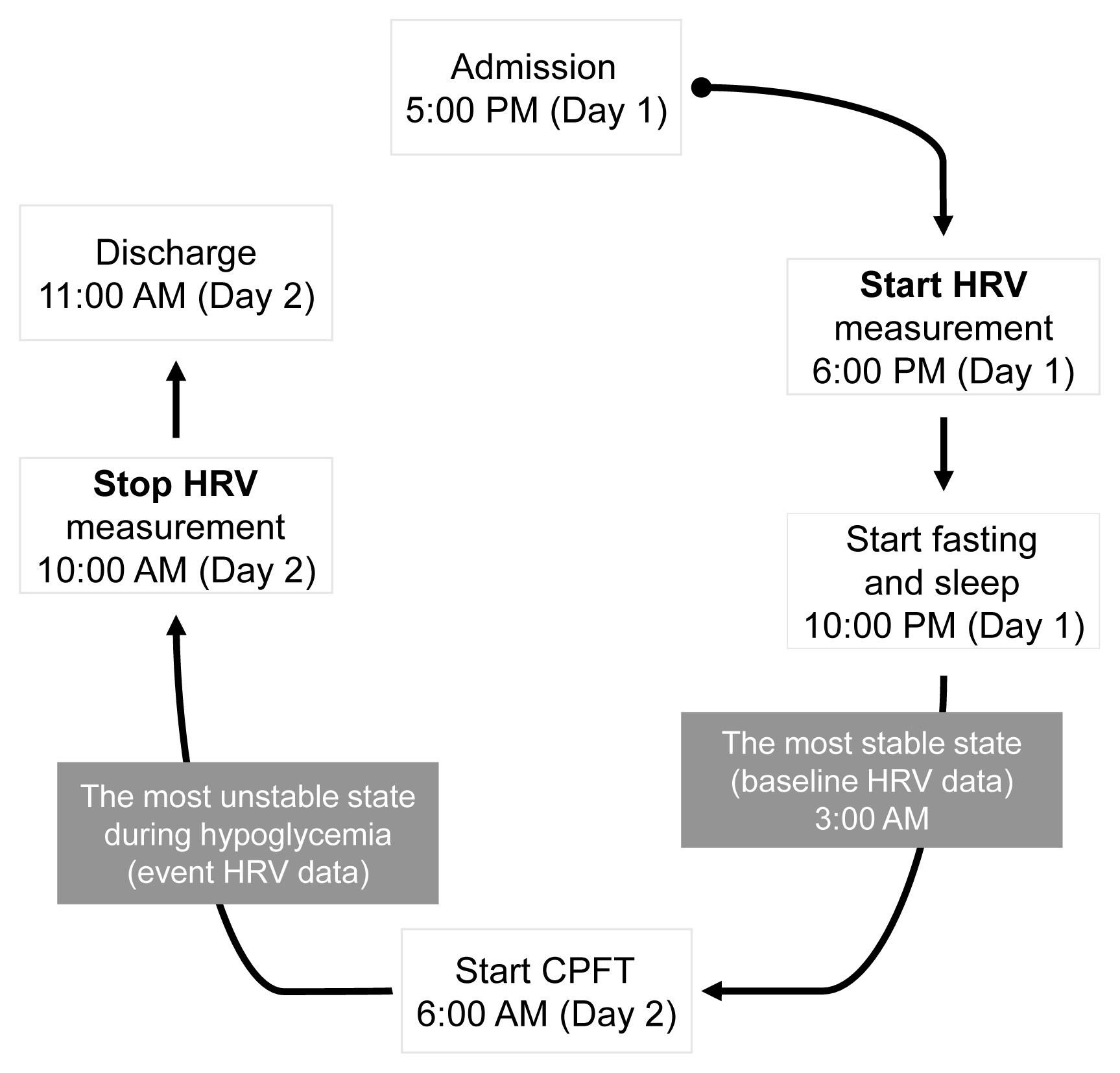
- Heart Rate Variability in Postoperative Patients with Nonfunctioning Pituitary Adenoma
- Jeonghoon Ha, Hansang Baek, Chaiho Jeong, Minsoo Yeo, Seung-Hwan Lee, Jae Hyoung Cho, Ki-Hyun Baek, Moo Il Kang, Dong-Jun Lim
- Endocrinol Metab. 2021;36(3):678-687. Published online June 10, 2021
- DOI: https://doi.org/10.3803/EnM.2021.978
- 4,599 View
- 109 Download
- 3 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Decreased heart rate variability (HRV) has been reported to be associated with cardiac autonomic dysfunction. Hypopituitarism in nonfunctioning pituitary adenoma (NFPA) is often linked to increased cardiovascular mortality. We therefore hypothesized that postoperative NFPA patients with hormone deficiency have an elevated risk of HRV alterations indicating cardiac autonomic dysfunction.
Methods
A total of 22 patients with NFPA were enrolled in the study. Between 3 and 6 months after surgery, a combined pituitary function test (CPFT) was performed, and HRV was measured. The period of sleep before the CPFT was deemed the most stable period, and the hypoglycemic period that occurred during the CPFT was defined as the most unstable period. Changes in HRV parameters in stable and unstable periods were observed and compared depending on the status of hormone deficiencies.
Results
In patients with adrenocorticotropic hormone (ACTH) deficiency with other pituitary hormone deficiencies, the low frequency to high frequency ratio, which represents overall autonomic function and is increased in the disease state, was higher (P=0.005). Additionally, the standard deviation of the normal-to-normal interval, which decreases in the autonomic dysfunction state, was lower (P=0.030) during the hypoglycemic period. In panhypopituitarism, the low frequency to high frequency ratio during the hypoglycemic period was increased (P=0.007).
Conclusion
HRV analysis during CPFT enables estimation of cardiac autonomic dysfunction in patients with NFPA who develop ACTH deficiency with other pituitary hormone deficiencies or panhypopituitarism after surgery. These patients may require a preemptive assessment of cardiovascular risk. -
Citations
Citations to this article as recorded by- Heart Rate Variability in Subjects with Severe Allergic Background Undergoing COVID-19 Vaccination
Maria Bernadette Cilona, Filippo D’Amico, Chiara Asperti, Giuseppe Alvise Ramirez, Stefano Turi, Giovanni Benanti, Shai Marc Bohane, Serena Nannipieri, Rosa Labanca, Matteo Gervasini, Federica Russetti, Naomi Viapiana, Martina Lezzi, Giovanni Landoni, Lor
Vaccines.2023; 11(3): 567. CrossRef - Pituitary Diseases and COVID-19 Outcomes in South Korea: A Nationwide Cohort Study
Jeonghoon Ha, Kyoung Min Kim, Dong-Jun Lim, Keeho Song, Gi Hyeon Seo
Journal of Clinical Medicine.2023; 12(14): 4799. CrossRef - Effect of a 16-Session Qigong Program in Non-Hodgkin Lymphoma Survivors: A Randomized Clinical Trial
Keyla Vargas-Román, Emilia I. De la Fuente-Solana, Jonathan Cortés-Martín, Juan Carlos Sánchez-García, Christian J. González-Vargas, Lourdes Díaz-Rodríguez
Journal of Clinical Medicine.2022; 11(12): 3421. CrossRef
- Heart Rate Variability in Subjects with Severe Allergic Background Undergoing COVID-19 Vaccination

Review Article
- Hypothalamus and Pituitary gland
- Best Achievements in Pituitary and Adrenal Diseases in 2020
- Chang Ho Ahn, Jung Hee Kim
- Endocrinol Metab. 2021;36(1):51-56. Published online February 24, 2021
- DOI: https://doi.org/10.3803/EnM.2021.108

- 4,546 View
- 162 Download
- 2 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Significant progress in pituitary and adrenal diseases was made in 2020. This review presents major translational and clinical advances in research on pituitary and adrenal diseases, encompassing their epidemiology, pathogenesis, diagnosis, and clinical management. We address the controversy regarding the nomenclature of pituitary neuroendocrine tumors, omics-based molecular classification of pituitary adenomas, and novel drugs for Cushing’s disease in the field of pituitary diseases. In the field of adrenal diseases, we cover big data-driven epidemiology of adrenal tumors, steroid profiling as a new diagnostic tool, and the utility of scoring systems in the decision-making process of managing primary aldosteronism. This brief article will broaden readers’ understanding of pituitary and adrenal diseases.
-
Citations
Citations to this article as recorded by- Surgical management and outcomes of spinal metastasis of malignant adrenal tumor: A retrospective study of six cases and literature review
Xiangzhi Ni, Jing Wang, Jiashi Cao, Kun Zhang, Shuming Hou, Xing Huang, Yuanjin Song, Xin Gao, Jianru Xiao, Tielong Liu
Frontiers in Oncology.2023;[Epub] CrossRef - 2023 Korean Endocrine Society Consensus Guidelines for the Diagnosis and Management of Primary Aldosteronism
Jeonghoon Ha, Jung Hwan Park, Kyoung Jin Kim, Jung Hee Kim, Kyong Yeun Jung, Jeongmin Lee, Jong Han Choi, Seung Hun Lee, Namki Hong, Jung Soo Lim, Byung Kwan Park, Jung-Han Kim, Kyeong Cheon Jung, Jooyoung Cho, Mi-kyung Kim, Choon Hee Chung
Endocrinology and Metabolism.2023; 38(6): 597. CrossRef
- Surgical management and outcomes of spinal metastasis of malignant adrenal tumor: A retrospective study of six cases and literature review

Special Article
- Miscellaneous
- Diagnosis and Treatment of Growth Hormone Deficiency: A Position Statement from Korean Endocrine Society and Korean Society of Pediatric Endocrinology
- Jung Hee Kim, Hyun Wook Chae, Sang Ouk Chin, Cheol Ryong Ku, Kyeong Hye Park, Dong Jun Lim, Kwang Joon Kim, Jung Soo Lim, Gyuri Kim, Yun Mi Choi, Seong Hee Ahn, Min Ji Jeon, Yul Hwangbo, Ju Hee Lee, Bu Kyung Kim, Yong Jun Choi, Kyung Ae Lee, Seong-Su Moon, Hwa Young Ahn, Hoon Sung Choi, Sang Mo Hong, Dong Yeob Shin, Ji A Seo, Se Hwa Kim, Seungjoon Oh, Sung Hoon Yu, Byung Joon Kim, Choong Ho Shin, Sung-Woon Kim, Chong Hwa Kim, Eun Jig Lee
- Endocrinol Metab. 2020;35(2):272-287. Published online June 24, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.2.272
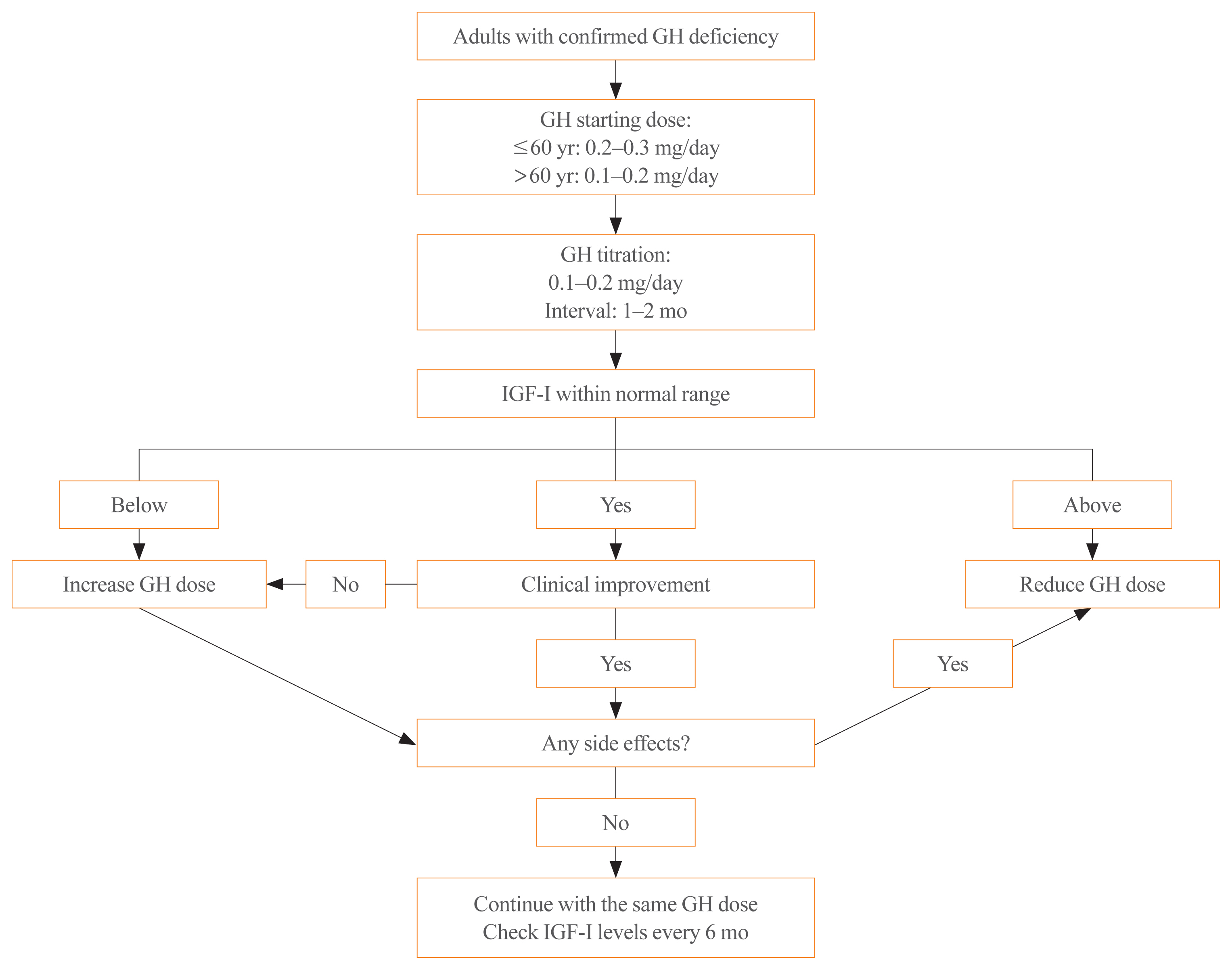
- 9,554 View
- 431 Download
- 14 Web of Science
- 16 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Growth hormone (GH) deficiency is caused by congenital or acquired causes and occurs in childhood or adulthood. GH replacement therapy brings benefits to body composition, exercise capacity, skeletal health, cardiovascular outcomes, and quality of life. Before initiating GH replacement, GH deficiency should be confirmed through proper stimulation tests, and in cases with proven genetic causes or structural lesions, repeated GH stimulation testing is not necessary. The dosing regimen of GH replacement therapy should be individualized, with the goal of minimizing side effects and maximizing clinical improvements. The Korean Endocrine Society and the Korean Society of Pediatric Endocrinology have developed a position statement on the diagnosis and treatment of GH deficiency. This position statement is based on a systematic review of evidence and expert opinions.
-
Citations
Citations to this article as recorded by- Once-Weekly Somapacitan as an Alternative Management of Growth Hormone Deficiency in Prepubertal Children: A Systematic Review and Meta-Analysis of Randomized Controlled Trial
Ghina Tsurayya, Cut Alifiya Nazhifah, Muhammad Rahmat Pirwanja, Putri Oktaviani Zulfa, Muhammad Raihan Ramadhan Tatroman, Fajar Fakri, Muhammad Iqhrammullah
Children.2024; 11(2): 227. CrossRef - Efficacy, safety, and patient satisfaction of norditropin and sogroya in patients with growth hormone deficiency: a systematic review and meta-analysis of randomized controlled trials
Obieda Altobaishat, Mohamed Abouzid, Mostafa Hossam El Din Moawad, Abdulrahman Sharaf, Yazan Al-Ajlouni, Tungki Pratama Umar, Abdallah Bani-salameh, Mohammad Tanashat, Omar Abdullah Bataineh, Abdulqadir J. Nashwan
Endocrine.2024;[Epub] CrossRef - Evaluation of Adult Height in Patients with Non-Permanent Idiopathic GH Deficiency
Agnese Murianni, Anna Lussu, Chiara Guzzetti, Anastasia Ibba, Letizia Casula, Mariacarolina Salerno, Marco Cappa, Sandro Loche
Endocrines.2023; 4(1): 169. CrossRef - The effect of hypothalamic involvement and growth hormone treatment on cardiovascular risk factors during the transition period in patients with childhood-onset craniopharyngioma
Sang Hee Park, Yun Jeong Lee, Jung-Eun Cheon, Choong Ho Shin, Hae Woon Jung, Young Ah Lee
Annals of Pediatric Endocrinology & Metabolism.2023; 28(2): 107. CrossRef - Continuous Glucose Monitoring: A Possible Aid for Detecting Hypoglycemic Events during Insulin Tolerance Tests
Soo Yeun Sim, Moon Bae Ahn
Sensors.2023; 23(15): 6892. CrossRef - The risk patients with AGHD have of developing CVD
Eisha Javed, Maha Zehra, Naz Elahi
International Journal of Cardiology Cardiovascular Risk and Prevention.2023; 19: 200221. CrossRef - Diagnosis of GH Deficiency Without GH Stimulation Tests
Anastasia Ibba, Sandro Loche
Frontiers in Endocrinology.2022;[Epub] CrossRef - Metabolic Impacts of Discontinuation and Resumption of Recombinant Human Growth Hormone Treatment during the Transition Period in Patients with Childhood-Onset Growth Hormone Deficiency
Yun Jeong Lee, Yunha Choi, Han-Wook Yoo, Young Ah Lee, Choong Ho Shin, Han Saem Choi, Ho-Seong Kim, Jae Hyun Kim, Jung Eun Moon, Cheol Woo Ko, Moon Bae Ahn, Byung-Kyu Suh, Jin-Ho Choi
Endocrinology and Metabolism.2022; 37(2): 359. CrossRef - A Radiomics-Based Model with the Potential to Differentiate Growth Hormone Deficiency and Idiopathic Short Stature on Sella MRI
Taeyoun Lee, Kyungchul Song, Beomseok Sohn, Jihwan Eom, Sung Soo Ahn, Ho-Seong Kim, Seung-Koo Lee
Yonsei Medical Journal.2022; 63(9): 856. CrossRef - Phenotypic spectrum of patients with mutations in CHD7: clinical implications of endocrinological findings
Ja Hye Kim, Yunha Choi, Soojin Hwang, Gu-Hwan Kim, Han-Wook Yoo, Jin-Ho Choi
Endocrine Connections.2022;[Epub] CrossRef - Immune Checkpoint Inhibitors and Endocrine Disorders: A Position Statement from the Korean Endocrine Society
Hyemi Kwon, Eun Roh, Chang Ho Ahn, Hee Kyung Kim, Cheol Ryong Ku, Kyong Yeun Jung, Ju Hee Lee, Eun Heui Kim, Sunghwan Suh, Sangmo Hong, Jeonghoon Ha, Jun Sung Moon, Jin Hwa Kim, Mi-kyung Kim
Endocrinology and Metabolism.2022; 37(6): 839. CrossRef - Laron syndrome: clinic, diagnostics (а clinical case)
P.M. Lіashuk, R.P. Lіashuk, N.I. Stankova, M.B. Kudina
INTERNATIONAL JOURNAL OF ENDOCRINOLOGY (Ukraine).2022; 18(3): 193. CrossRef - Diagnosis for Pheochromocytoma and Paraganglioma: A Joint Position Statement of the Korean Pheochromocytoma and Paraganglioma Task Force
Eu Jeong Ku, Kyoung Jin Kim, Jung Hee Kim, Mi Kyung Kim, Chang Ho Ahn, Kyung Ae Lee, Seung Hun Lee, You-Bin Lee, Kyeong Hye Park, Yun Mi Choi, Namki Hong, A Ram Hong, Sang-Wook Kang, Byung Kwan Park, Moon-Woo Seong, Myungshin Kim, Kyeong Cheon Jung, Chan
Endocrinology and Metabolism.2021; 36(2): 322. CrossRef - Asian Conference on Tumor Ablation Guidelines for Adrenal Tumor Ablation
Byung Kwan Park, Masashi Fujimori, Shu-Huei Shen, Uei Pua
Endocrinology and Metabolism.2021; 36(3): 553. CrossRef - Asian Conference on Tumor Ablation guidelines for renal cell carcinoma
Byung Kwan Park, Shu-Huei Shen, Masashi Fujimori, Yi Wang
Investigative and Clinical Urology.2021; 62(4): 378. CrossRef - Diagnosis and Treatment of Adult Growth Hormone Deficiency
Jung Hee Kim
The Korean Journal of Medicine.2021; 96(5): 400. CrossRef
- Once-Weekly Somapacitan as an Alternative Management of Growth Hormone Deficiency in Prepubertal Children: A Systematic Review and Meta-Analysis of Randomized Controlled Trial

Review Articles
- Hypothalamus and Pituitary gland
- Epidemiology of Functioning Pituitary Adenomas
- Sang Ouk Chin
- Endocrinol Metab. 2020;35(2):237-242. Published online June 24, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.2.237
- 7,967 View
- 258 Download
- 11 Web of Science
- 12 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Pituitary adenomas (PAs) are defined as benign monoclonal tumors in the pituitary gland that cause symptoms due to either hormonal hypersecretion or a space-occupying effect, and are classified as functioning or non-functioning. Because of their rarity and slow-growing with symptomless nature in most cases, it has been challenging to investigate the epidemiology of PAs. Considering their public health impact and association with increased morbidity and mortality, however, it is essential to understand the prevalence and incidence of PAs in order to improve patient outcomes and to minimize the resultant burden on the health care system. Fortunately, developments in imaging modalities and easier access to large-scale population data have enabled investigators to analyze the epidemiology of PAs more accurately. This review summarizes previously reported epidemiologic data on functioning PAs in Korea and other countries.
-
Citations
Citations to this article as recorded by- Spherical type of amyloidogenic pituitary prolactinoma in a 50 year old male
Madhala Divya, Balasubramanian Archana, Lawrence D Cruze, D. Balasubramanian
Interdisciplinary Neurosurgery.2024; 36: 101957. CrossRef - Transcriptome of GH-producing pituitary neuroendocrine tumours and models are significantly affected by somatostatin analogues
Rihards Saksis, Olesja Rogoza, Helvijs Niedra, Kaspars Megnis, Ilona Mandrika, Inga Balcere, Liva Steina, Janis Stukens, Austra Breiksa, Jurijs Nazarovs, Jelizaveta Sokolovska, Ilze Konrade, Raitis Peculis, Vita Rovite
Cancer Cell International.2023;[Epub] CrossRef - Prognostic Models in Growth-Hormone- and Prolactin-Secreting Pituitary Neuroendocrine Tumors: A Systematic Review
Roxana-Ioana Dumitriu-Stan, Iulia-Florentina Burcea, Teodor Salmen, Catalina Poiana
Diagnostics.2023; 13(12): 2118. CrossRef - Salivary microbiome profiles for different clinical phenotypes of pituitary adenomas by single-molecular long-read sequencing
Xuefei Ji, Pingping Li, Qinglong Guo, Liao Guan, Peng Gao, Bingshan Wu, Hongwei Cheng, Jin Xiao, Lei Ye, Justin R. Kaspar
Microbiology Spectrum.2023;[Epub] CrossRef - Endoscopic endonasal surgical management of giant pituitary adenomas with extension into ventricle system
Mykola O. Guk, Oleksii V. Ukrainets
Ukrainian Neurosurgical Journal.2023; 29(4): 13. CrossRef - Uso de resonancia magnética nuclear intraoperatoria en la resección transesfenoidal de adenomas hipofisiarios: ¿qué resultados se han obtenido?
María Laura Boschetti Saer, Levino Roberto Boschetti, Jose Pastor Linarez Veloz, Michael Ortega-Sierra
Archivos de Neurociencias.2023;[Epub] CrossRef - Epidemiology of common and uncommon adult pituitary tumors in the U.S. according to the 2017 World Health Organization classification
Luz E. Castellanos, Catherine Gutierrez, Timothy Smith, Edward R. Laws, J. Bryan Iorgulescu
Pituitary.2022; 25(1): 201. CrossRef - The kinome, cyclins and cyclin-dependent kinases of pituitary adenomas, a look into the gene expression profile among tumors from different lineages
Keiko Taniguchi-Ponciano, Lesly A. Portocarrero-Ortiz, Gerardo Guinto, Sergio Moreno-Jimenez, Erick Gomez-Apo, Laura Chavez-Macias, Eduardo Peña-Martínez, Gloria Silva-Román, Sandra Vela-Patiño, Jesús Ordoñez-García, Sergio Andonegui-Elguera, Aldo Ferreir
BMC Medical Genomics.2022;[Epub] CrossRef - IL-10 Gene Rs1800871, Rs1800872, and Rs1800896 Polymorphisms and IL-10 Serum Levels Association with Pituitary Adenoma
Migle Palivonaite, Greta Gedvilaite, Brigita Glebauskiene, Loresa Kriauciuniene, Vita Rovite, Rasa Liutkeviciene
Biomedicines.2022; 10(8): 1921. CrossRef - Genome wide analysis of circulating miRNAs in growth hormone secreting pituitary neuroendocrine tumor patients’ plasma
Helvijs Niedra, Raitis Peculis, Helena Daiga Litvina, Kaspars Megnis, Ilona Mandrika, Inga Balcere, Mihails Romanovs, Liva Steina, Janis Stukens, Austra Breiksa, Jurijs Nazarovs, Jelizaveta Sokolovska, Rasa Liutkeviciene, Alvita Vilkevicute, Ilze Konrade,
Frontiers in Oncology.2022;[Epub] CrossRef - Identification of mutant K-RAS in pituitary macroadenoma
Veronica Aran, Manoela Heringer, Paulo Jose da Mata, Leandro Kasuki, Renan Lyra Miranda, Felipe Andreiuolo, Leila Chimelli, Paulo Niemeyer Filho, Monica Roberto Gadelha, Vivaldo Moura Neto
Pituitary.2021; 24(5): 746. CrossRef - Surgery is a safe, effective first-line treatment modality for noninvasive prolactinomas
Ji Yong Park, Wonsuk Choi, A Ram Hong, Jee Hee Yoon, Hee Kyung Kim, Woo-Youl Jang, Shin Jung, Ho-Cheol Kang
Pituitary.2021; 24(6): 955. CrossRef
- Spherical type of amyloidogenic pituitary prolactinoma in a 50 year old male

- Hypothalamus and Pituitary gland
- Precision Therapy in Acromegaly Caused by Pituitary Tumors: How Close Is It to Reality?
- Cheol Ryong Ku, Vladimir Melnikov, Zhaoyun Zhang, Eun Jig Lee
- Endocrinol Metab. 2020;35(2):206-216. Published online June 24, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.2.206
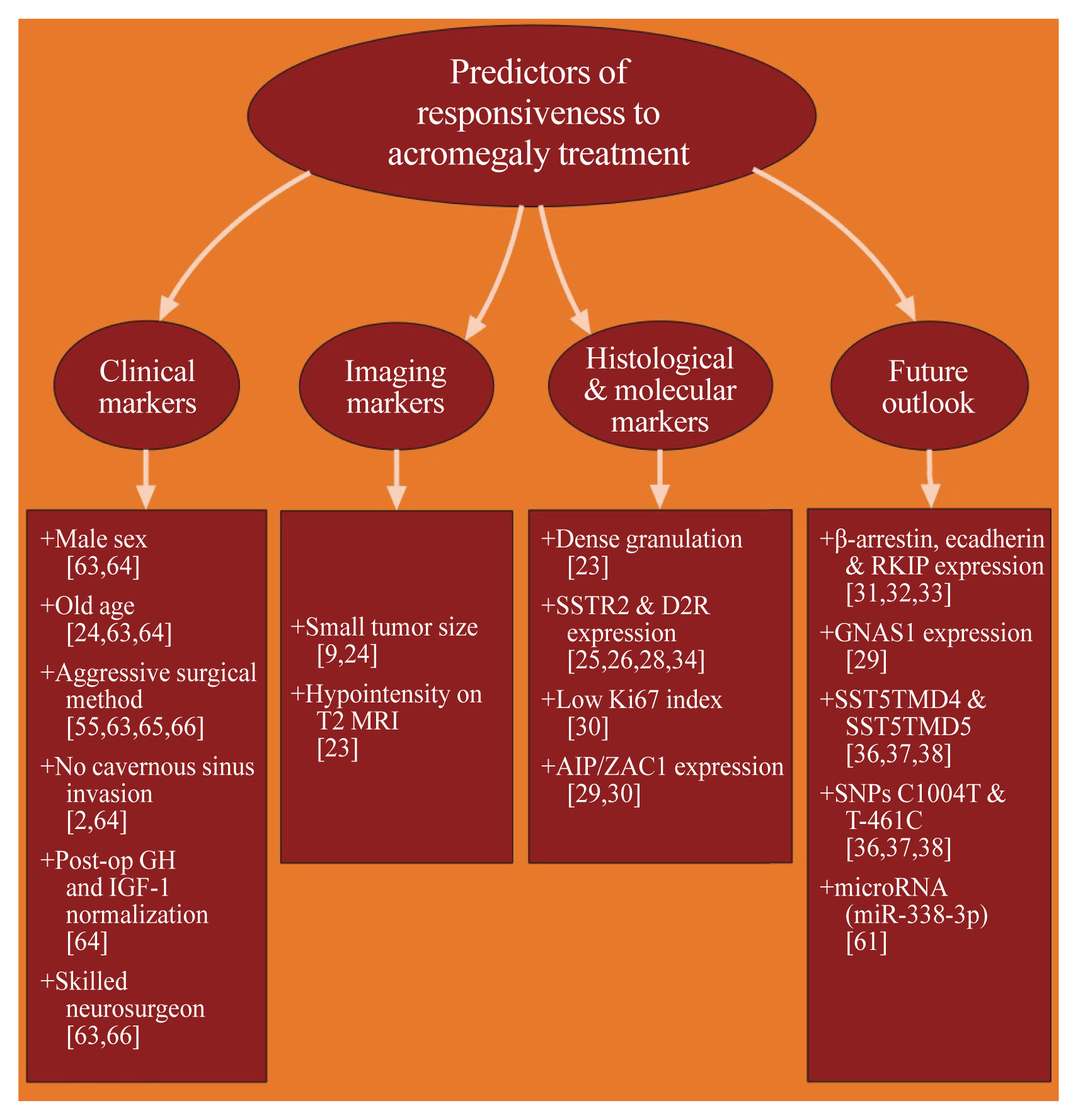
- 6,795 View
- 253 Download
- 8 Web of Science
- 8 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Acromegaly presents with an enigmatic range of symptoms and comorbidities caused by chronic and progressive growth hormone elevations, commonly due to endocrinologic hypersecretion from a pituitary gland tumor. Comprehensive national acromegaly databases have been appearing over the years, allowing for international comparisons of data, although still presenting varying prevalence and incidence rates. Lack of large-scale analysis in geographical and ethnic differences in clinical presentation and management requires further research. Assessment of current and novel predictors of responsiveness to distinct therapy can lead to multilevel categorization of patients, allowing integration into new clinical guidelines and reduction of increased morbidity and mortality associated with acromegaly. This review compares current data from epidemiological studies and assesses the present-day application of prognostic factors in medical practice, the reality of precision therapy, as well as its future prospects in acromegaly, with a special focus on its relevance to the South Korean population.
-
Citations
Citations to this article as recorded by- Biomarkers of response to treatment in acromegaly
Leandro Kasuki, Elisa Lamback, Ximene Antunes, Mônica R. Gadelha
Expert Review of Endocrinology & Metabolism.2024; 19(1): 71. CrossRef - Multiomics Approach to Acromegaly: Unveiling Translational Insights for Precision Medicine
Kyungwon Kim, Cheol Ryong Ku, Eun Jig Lee
Endocrinology and Metabolism.2023; 38(5): 463. CrossRef - Risk of depression in patients with acromegaly in Korea (2006-2016): a nationwide population-based study
Shinje Moon, Sangmo Hong, Kyungdo Han, Cheol-Young Park
European Journal of Endocrinology.2023; 189(3): 363. CrossRef - The Future of Somatostatin Receptor Ligands in Acromegaly
Monica R Gadelha, Luiz Eduardo Wildemberg, Leandro Kasuki
The Journal of Clinical Endocrinology & Metabolism.2022; 107(2): 297. CrossRef - Innovative therapeutics in acromegaly
Leandro Kasuki, Mônica R. Gadelha
Best Practice & Research Clinical Endocrinology & Metabolism.2022; 36(6): 101679. CrossRef - Risk of Neurodegenerative Diseases in Patients With Acromegaly
Sangmo Hong, Kyungdo Han, Kyung-Soo Kim, Cheol-Young Park
Neurology.2022;[Epub] CrossRef - Machine Learning-based Prediction Model for Treatment of Acromegaly With First-generation Somatostatin Receptor Ligands
Luiz Eduardo Wildemberg, Aline Helen da Silva Camacho, Renan Lyra Miranda, Paula C L Elias, Nina R de Castro Musolino, Debora Nazato, Raquel Jallad, Martha K P Huayllas, Jose Italo S Mota, Tobias Almeida, Evandro Portes, Antonio Ribeiro-Oliveira, Lucio Vi
The Journal of Clinical Endocrinology & Metabolism.2021; 106(7): 2047. CrossRef - Skin anomalies in acromegalic patients (Review of the practical aspects)
Florica Sandru, Adelina Popa, Dan Paduraru, Alexandru Filipescu, Mara Carsote, Adina Ghemigian
Experimental and Therapeutic Medicine.2021;[Epub] CrossRef
- Biomarkers of response to treatment in acromegaly


 KES
KES

 First
First Prev
Prev



