Protein Expression of Cyclin B1, Transferrin Receptor, and Fibronectin Is Correlated with the Prognosis of Adrenal Cortical Carcinoma
Article information
Abstract
Background
Adrenal cortical carcinoma (ACC) is a rare cancer with a variable prognosis. Several prognostic factors of ACC have been previously reported, but a proteomic analysis has not yet been performed. This study aimed to investigate prognostic biomarkers for ACC using a proteomic approach.
Methods
We used reverse-phase protein array data from The Cancer Proteome Atlas, and identified differentially expressed proteins in metastatic ACCs. Multivariate Cox regression analysis adjusted by age and staging was used for survival analysis, and the C-index and category-free net reclassification improvement (cfNRI) were utilized to evaluate additive prognostic value.
Results
In 46 patients with ACC, cyclin B1, transferrin receptor (TfR1), and fibronectin were significantly overexpressed in patients with distant metastasis. In multivariate models, high expression of cyclin B1 and TfR1 was significantly associated with mortality (hazard ratio [HR], 6.13; 95% confidence interval [CI], 1.02 to 36.7; and HR, 6.59; 95% CI, 1.14 to 38.2; respectively), whereas high fibronectin expression was not (HR, 3.92; 95% CI, 0.75 to 20.4). Combinations of high cyclin B1/high TfR1, high cyclin B1/high fibronectin, and high TfR1/high fibronectin were strongly associated with mortality ([HR, 13.72; 95% CI, 1.89 to 99.66], [HR, 9.22; 95% CI, 1.34 to 63.55], and [HR, 18.59; 95% CI, 2.54 to 135.88], respectively). In reclassification analyses, cyclin B1, TfR1, fibronectin, and combinations thereof improved the prognostic performance (C-index, 0.78 to 0.82–0.86; cfNRI, all P values <0.05).
Conclusion
In ACC patients, the overexpression of cyclin B1, TfR1, and fibronectin and combinations thereof were associated with poor prognosis.
INTRODUCTION
Adrenal cortical carcinoma (ACC) is a rare cancer, with an annual incidence of about 1–2 per million [1]. ACC has a poor prognosis, with a 5-year overall survival rate of less than 40% for all cancers and 10% for metastatic cancers [2]. However, even in metastatic ACC, the prognosis is quite variable, with reported survival ranging from a few months to more than 10 years, suggesting the heterogeneity of these tumors [3]. Tumor, node, metastasis (TNM) staging has been used as a predictor of survival and has been modified to improve its prognostic power; currently, the eighth edition of TNM staging and European Network for the Study of Adrenal Tumors (ENSAT) staging systems are used [456]. Nevertheless, a significant proportion of patients with localized disease according to TNM staging experience recurrence after surgery (up to 70% within 3 years), which is the only curative treatment to date [2]. Tumor grade, as assessed by mitotic count and proliferation indices such as the Ki-67 index, has shown good predictive value for localized ACC [7]. However, tumor grade still has the limitation of poor reproducibility [8]. Molecular studies have focused on several potential driver genes, but their independent prognostic values are controversial [910]. Recently, distinct molecular subgroups have been identified via pan-genomic approaches using DNA copy number, mRNA expression, miRNA expression, and DNA methylation, and these subgroups have shown associations with different survival outcomes [1112]. Moreover, this classification demonstrated high prognostic value when combining with clinical staging and tumor grade [13]. However, the pan-genomic approach is quite complex and not easy to use in clinical practice. In addition, pan-genomic biomarkers had limited value in prognostic performance for metastatic ACC [13].
Few studies have investigated the use of a proteomic approach to predict the prognosis of ACC. Moreover, protein markers can be useful in clinical practice since they can be straightforwardly analyzed using immunohistochemistry or Western blots. Therefore, we aimed to investigate prognostic biomarkers for ACCs using a proteomic approach.
METHODS
Study subjects and data source
In this study, ACC datasets from The Cancer Proteome Atlas (TCPA) database were extracted from the cBioPortal website (http://www.cbioportal.org/) using The Cancer Genome Atlas (TCGA) provisional dataset [14]. We obtained data from reverse-phase protein arrays (RPPA), an antibody-based quantitative method assessing multiple protein markers in a cost-effective, sensitive, and high-throughput manner [14]. RPPA data that had been normalized using the z-score, as provided in cBio-Portal, were used. Among the 91 ACC patients in the TCGA database, we included 46 patients with RPPA data. Clinical data, including staging (ENSAT), treatment, and survival information, were also obtained from the same source [5].
Differential protein expression analysis
Differential protein expression analysis of the RPPA data was performed using the Perseus software (http://www.perseus-framework.org). Differentially expressed proteins (DEPs) in metastatic and non-metastatic ACCs were identified, and the false-discovery rate (FDR) was controlled using the Benjamini-Hochberg method. The list of DEPs was limited to proteins showing a fold-change of equal to or greater than +2, or equal to or less than −2, and an FDR lower than 0.05. Protein expression above the median value was defined as high expression.
Statistical analysis
Kaplan-Meier analysis and the log-rank test were used to construct the survival curve. Cox proportional-hazards regression analyses were used to calculate the hazard ratios (HRs) of DEPs for all-cause mortality. Age and staging were adjusted in the multivariable Cox regression analyses. To determine the incremental discriminative value of DEPs additive to age and staging for prognosis, we calculated C-statistics and the category-free net reclassification improvement (cfNRI). In addition, we performed a subgroup analysis of non-metastatic patients. All P values were two-sided, and P values of lower than 0.05 were considered to indicate statistical significance in all analyses. The statistical analyses were performed using STATA software version 13 (StataCorp, College Station, TX, USA; https://www.stata.com) and R version 3.6.1 (R Foundation, Vienna, Austria; https://www.r-project.org).
Ethical statement
Written informed consent by subjects was waived due to the public database. All data were anonymized, and this study received Institutional Review Board (IRB) approval at Seoul National University Hospital (IRB No. E-1910-005-1067).
RESULTS
Baseline characteristics and clinical outcomes
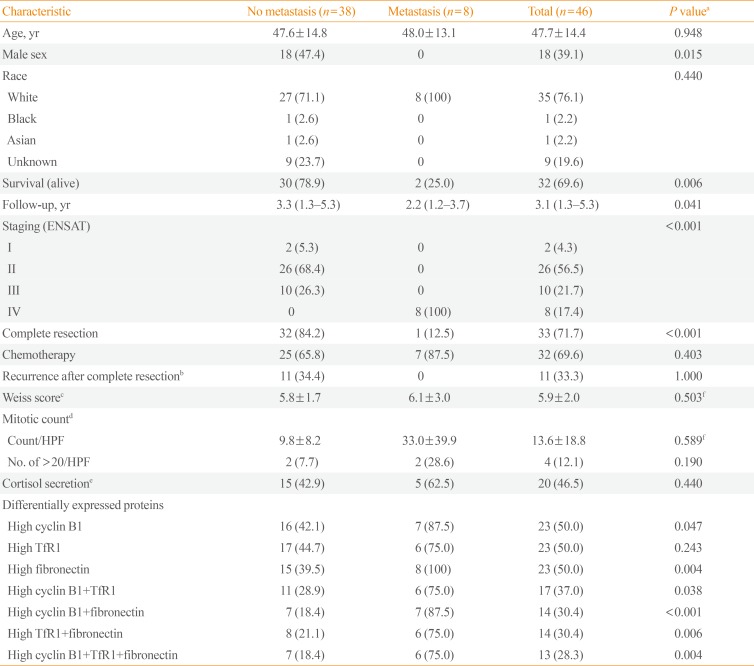
The baseline characteristics of the participants are shown in Table 1. Their mean age was 47.7±14.4 years, and 18 (39.1%) were male. Thirty-five (76.1%) were white, two (4.4%) were non-white, and the others' racial information was unknown. The number of patients with ENSAT stage I, II, III, and IV disease was two (4.3%), 26 (56.5%), 10 (21.7%), and eight (17.4%), respectively. Complete resection was performed in 33 (71.7%) patients, of whom 11 experienced recurrence. Thirty-two patients (69.6%) received adjuvant chemotherapy after surgery. The patients' mean Weiss score was 5.9±2.0.
Differential expression of proteins
The expression levels of 198 proteins in the RPPA data are shown in Supplemental Table S1. In patients with distant metastasis, three proteins were found to be upregulated (fold-change ≥2)—CCNB1 (cyclin B1), TFRC (transferrin receptor [TfR1]), and FN1 (fibronectin)—and none was found to be downregulated (fold-change ≤−2) A volcano plot of DEPs according to metastasis is presented in Fig. 1.
Survival analyses
During the median follow-up period of 3.1 years (interquartile range, 1.3 to 5.3), mortality occurred in 14 patients (31.4%). Kaplan-Meier curves for each of the above-mentioned DEPs and combinations thereof are shown in Fig. 2. The survival rate was significantly lower in patients with high expression levels of each of these three proteins than in those with low expression levels (overall log-rank P value <0.001 for all). In addition, significantly lower survival rates were seen for all combinations of these proteins (high cyclin B1/high TfR1, high cyclin B1/high fibronectin, high TfR1/high fibronectin, high cyclin B1/high TfR1/high fibronectin; overall log-rank P value <0.001 for all).

Kaplan-Meier curves for overall survival based on the expression status of differentially expressed proteins. (A) Cyclin B1. (B) Transferrin receptor (TfR1). (C) Fibronectin. (D) Cyclin B1 and TfR1. (E) Cyclin B1 and fibronectin. (F) TfR1 and fibronectin. (G) Cyclin B1, TfR1, and fibronectin.
In the unadjusted Cox regression analyses, high expression of each protein and all combinations thereof were significantly associated with mortality (Table 2). In multivariate Cox regression analyses adjusted for age and staging, high expression levels of cyclin B1 and TfR1 were significantly associated with high mortality. However, the expression level of fibronectin failed to predict mortality in the multivariate Cox regression model. All combinations of each protein expression signature (high cyclin B1/high TfR1, high cyclin B1/high fibronectin, high TfR1/high fibronectin, and high cyclin B1/high TfR1/high fibronectin) significantly predicted mortality. The HRs for mortality of combinations involving each DEP were higher than those of each protein individually.
Reclassification analyses of individual DEPs and combinations thereof for overall survival
C-statistics and the cfNRI were used to evaluate the prognostic values of the DEPs additive to age and staging (Table 3). The C-index of the reference model (age and staging) was 0.78, and it increased when each protein (high cyclin B1, high TfR1, and high fibronectin) was added, to 0.82, 0.83, and 0.82, respectively. The C-index increased further when combinations of the proteins were considered (high cyclin B1/high TfR1, high cyclin B1/high fibronectin, high TfR1/high fibronectin, and high cyclin B1/high TfR1/high fibronectin), which yielded C-index values of 0.84, 0.86, 0.86, and 0.86, respectively. In addition, the cfNRI values were also significant for each protein, individually and in combination with others (P value <0.05 for all). The event and nonevent cfNRIs were both positive in all models.
Subgroup analysis of non-metastatic ACC patients
We performed a subgroup analysis of non-metastatic patients (Table 4). Some combinations of DEPs (high cyclin B/high TfR1 and high TfR1/high fibronectin) showed significant associations with mortality, while other combinations (high cyclin B1/high fibronectin and high cyclin B1/high TfR1/high fibronectin) showed near-significant associations with mortality.
DISCUSSION
In the present study, we identified three proteins (cyclin B1, TfR1, and fibronectin) that were highly expressed in metastatic ACC in the TCPA database. With a median follow-up of 3.1 years, high expression of each of these three proteins was associated with a poor survival rate. Subjects with high expression levels of a combination of two DEPs were at a higher risk for mortality than those with a high expression levels of only one DEP. Cyclin B1, TfR1, and all combinations of the three DEPs showed meaningful prognostic performance independent of age and staging. Moreover, among non-metastatic patients, combinations of these three DEPs showed significant or near-significant associations with mortality. The reason for the non-significance of fibronectin alone needs to be elucidated, but the small number of patients may have contributed to this finding. In addition, the C-index and cfNRI values of high cyclin B1/high TfR1/high fibronectin were the same as those of high cyclin B1/high fibronectin. This phenomenon may be attributed to the similar number of subjects with high expression of all three DEPs and with high cyclin B1 and high fibronectin expression.
We conducted a proteomic analysis to identify prognostic markers for ACC. Several studies have explored prognostic markers for ACC, but most studies have reported clinical or pathological prognostic markers [57]. Molecular studies revealed that mutations in several potential driver genes such as CTNNB1 and TP53 may serve as prognostic markers [1015]. Nevertheless, the independent prognostic value of these markers is controversial [910]. Recently, pan-genomic approaches—encompassing DNA copy number, mRNA expression, miRNA expression, and DNA methylation—classified ACC patients into distinct molecular subgroups, which showed different survival outcomes [111213]. Despite the good performance of pan-genomic prognostic markers in previous studies, the relevant analyses are quite complex to perform, and their prognostic values were insufficient in advanced ACC (stage IV) [13]. To overcome these limitations, our present study utilized a proteomic approach, which has not been investigated in previous research on ACC. In this study, the expression status of three DEPs had prognostic value in addition to age and staging, as assessed by C-index and cfNRI. In addition, the highest C-index of our prediction model was quite high (0.86), which is similar to the result of a previous study [13], given that we included stage IV patients. Therefore, the DEPs that we identified can be applied as relatively simple and powerful prognostic markers in ACC.
The three identified DEPs—cyclin B1, TfR1, and fibronectin—have been studied as prognostic markers for other cancers. Cyclin B1 is a regulatory protein that plays an important role in mitosis by forming a complex with Cdk1 [16]. It acts as a switch-like manner in the decision to progress from G2 to the M phase in the cell. In many cancers, such as breast, cervical, colorectal, esophageal, lung, and prostate cancers, high expression levels of cyclin B1 have been found [1718192021] and are associated with a poor prognosis [1721]. In addition, the use of small interfering RNA that targeted cyclin B1 downregulated tumor proliferation and enhanced sensitivity to systemic chemotherapy [22]. Moreover, in a previous study using ACC cell lines (H295R and SW-13 cells), the mechanism of mitotane was found to involve the cyclin B1/Cdk complex [23].
TfR1 is a transmembrane glycoprotein that imports iron-bound transferrin into cells by endocytosis [24]. It has also been reported that TfR1 is overexpressed in many cancers, such as breast, prostate, colon, liver, lung, brain, ovarian, and hematologic cancers [25262728293031]. This high expression of TfR1 in cancer is explained by the high iron requirements of cancer cells, as well as by the regulatory role of TfR1 in anti-apoptotic regulation, reactive oxygen species production, and mitochondrial respiration [303233]. In addition, as potential TfR1 inhibitors, curcumin, antibody A24 or JST-TFR09, and miR-320 have been studied in several cancers [34353637].
Fibronectin is a glycoprotein of the extracellular matrix that plays a role in cell growth, differentiation, migration, and wound healing [38]. It has also been reported that abnormal expression of fibronectin promotes invasion and migration of cancers, including breast, ovarian, prostate, lung, and colon cancers [3940414243]. In addition, fibronectin has been reported to play a role in reducing the response of cancer to cytotoxic therapy [4445]. In vitro studies of lung, pancreatic neuroendocrine, and breast cancers have shown that several molecules (e.g., PP2, PF-04554878, pUR4B, and AdF512v1) that inhibit fibronectin and its upstream or downstream pathways reduced its effects on tumor cell proliferation, migration, and angiogenesis [42464748].
As discussed above, previous studies have suggested that all three of the proteins (cyclin B1, TfR1, and fibronectin) identified as DEPs in our study may be prognostic markers in several cancers. However, to the best of our knowledge, no study has yet demonstrated an association of these proteins with mortality in ACC patients. Accordingly, these proteins may also be useful novel markers of poor prognosis in ACC patients.
There are several limitations of our study. The number of subjects was small, and our results were not validated in other cohorts. Thus, the prognostic power of these three DEPs remains to be confirmed. In addition, the RPPA data of ACC patients from the TCPA, which were used as the data source in our study, contained information on a small number of proteins. Therefore, it is possible that other protein markers might predict survival in ACC patients. Advanced proteomics techniques, such as gas or liquid chromatography tandem mass spectrometry, might enable the identification of more prognostic protein markers in ACC patients.
Taken together, the overexpression of cyclin B1, TfR1, and fibronectin proteins may have prognostic value for ACC patients. Further studies need to validate these three proteins as prognostic markers in another ACC cohort.
ACKNOWLEDGMENTS
This study was supported by the Korean Endocrine Hypertension-Adrenal Study Group from the Korean Endocrinology Society. This work was presented in abstract form at the 2019 Endocrine Society in New Orleans, USA.
Notes
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTIONS:
Conception or design: S.J.M., J.H.K., S.H.K., C.S.S.
Acquisition, analysis, or interpretation of data: S.J.M., J.H.K.
Drafting the work or revising: S.J.M., J.H.K.
Final approval of the manuscript: S.J.M., J.H.K., S.H.K., C.S.S.
References
SUPPLEMENTARY MATERIAL
Supplemental Table S1
Protein Expression Data and Differentially Expressed Protein Analysis