Update on Glucocorticoid Induced Osteoporosis
Article information
Abstract
Glucocorticoids are used to treat many autoimmune and inflammatory diseases. However, an adverse systemic effect is a deleterious effect on bone, which may lead to glucocorticoid-induced osteoporosis, characterized by a rapid and transient increase in bone resorption and fracture risk, which may increase rapidly within 3 months of commencing oral glucocorticoids. Therefore, early risk assessment and intervention are crucial for preventing fractures in patients receiving glucocorticoids. Recent practice guidelines recommend an assessment for fracture risk in patients beginning or receiving glucocorticoids for more than 3 months, and they have suggested fracture risk assessment tool values for identifying patients who need preventive treatment. Bisphosphonates are currently the recommended first-line therapy for the prevention and treatment of glucocorticoid-induced osteoporosis. These have been shown to increase the bone mineral density in the spine and hip and to decrease the incidence of vertebral fractures. Recently, a more potent antiresorptive agent, denosumab, has been shown to increase the bone density in patients receiving glucocorticoids. Teriparatide has been shown to have a preventive effect on vertebral fractures, but not on nonvertebral fractures. In this article we aimed to provide an update on glucocorticoid-induced osteoporosis by focusing on the assessment of its risk and treatment options.
INTRODUCTION
Glucocorticoids (GCs) are potent anti-inflammatory drugs first introduced in the 1950s to treat rheumatoid arthritis (RA) [1]. Since then, the efficacy of GCs in a number of autoimmune and inflammatory disorders has been proven [2]. Several studies from the United States and European countries have estimated the prevalence of GC use to range from 0.7% to 17.1% [3–7], and the prescription rate of long-term oral GC has increased by 34% over the past 20 years [4]. In Korea, a recent study demonstrated that the prevalence of chronic oral GC use for ≥30 days increased from 0.16% in 2002 to 0.54% in 2015 [8].
GCs have been reported to result in decreased bone density and increased fracture risk. Approximately 12% of patients on chronic GC treatment are diagnosed with symptomatic fractures [9], and 27% to 51% have radiographic evidence of vertebral fractures [10]. Furthermore, there are multiple risk factors for osteoporosis other than inflammation, including old age, female sex, menopause, and decreased physical activity in patients with inflammatory diseases such as RA [11]. Glucocorticoid-induced osteoporosis (GIOP) is characterized by a rapid and transient increase in bone resorption, accompanied by a long-lasting inhibition of bone formation and osteocyte activity [12]. Consequently, fracture risk increases rapidly within the first 3 months of GC treatment, regardless of decreasing bone mineral density (BMD) [13,14]. Therefore, early intervention for GIOP is critical to prevent fractures. Nevertheless, many patients receiving GC are not evaluated for their skeletal health [15] and are treated inadequately [16]; therefore, awareness of GIOP needs to be emphasized.
In this review article, we intend to provide an update of GIOP, focusing on the assessment of its risk and treatment options. Although the endogenous form of GIOP should also be managed [17], we focused on exogenous GIOP in this article.
PATHOPHYSIOLOGY
The direct effects of GCs on bone have been characterized by two distinct phases [18]. First, osteoclastogenesis during GC treatment increases, which leads to bone resorption mediated by an upregulation of receptor activator of nuclear factor kappa-B ligand (RANKL) and macrophage colony-stimulating factor with decreasing osteoprotegerin [19,20]. A negative uncoupling between bone formation and bone resorption is believed to be crucial for early and rapid bone loss [21]. The next phase is mediated by a reduction in circulating osteoblasts. GCs upregulate the expression of peroxisome proliferator-activated receptor-gamma (PPAR-γ), which subsequently increases the differentiation of precursor cells towards adipocytes rather than osteoblasts, thus reducing osteoblast differentiation and formation [22]. Osteoblastogenesis mediated by the Wnt/β-catenin signaling pathway is also inhibited by GCs. GCs inhibit Wnt binding by increasing the expression of Dickkopfl, which leads to a decrease in osteoblastogenesis [23–25].
EPIDEMIOLOGY
GIOP is the most frequent cause of secondary osteoporosis worldwide [12]. Many epidemiological studies have demonstrated that increased fracture risk is associated with the dose and duration of GC treatment [13,26,27]. Low (2.5 to 7.5 mg) doses of prednisone-equivalents per day can be associated with fracture, especially in vertebral fractures (1.7- to 2.5-fold increase) [13,27]. The risk of this increases rapidly within 3 to 6 months of commencement of GC treatment and is greater in patients exposed to higher doses continuously for a longer period [28,29].
TREATMENT THRESHOLD IN PATIENTS ON GCs
Various guidelines for the management of GIOP have been developed and updated by several academic societies (Table 1) [30–42]. The Korean GIOP guideline was recently developed by adapting established guidelines from other countries [30]. However, the intervention thresholds differ between guidelines. A recent report evaluated and compared the treatment threshold in patients with GC because it is important to set appropriate thresholds to prevent fractures in GIOP [43].
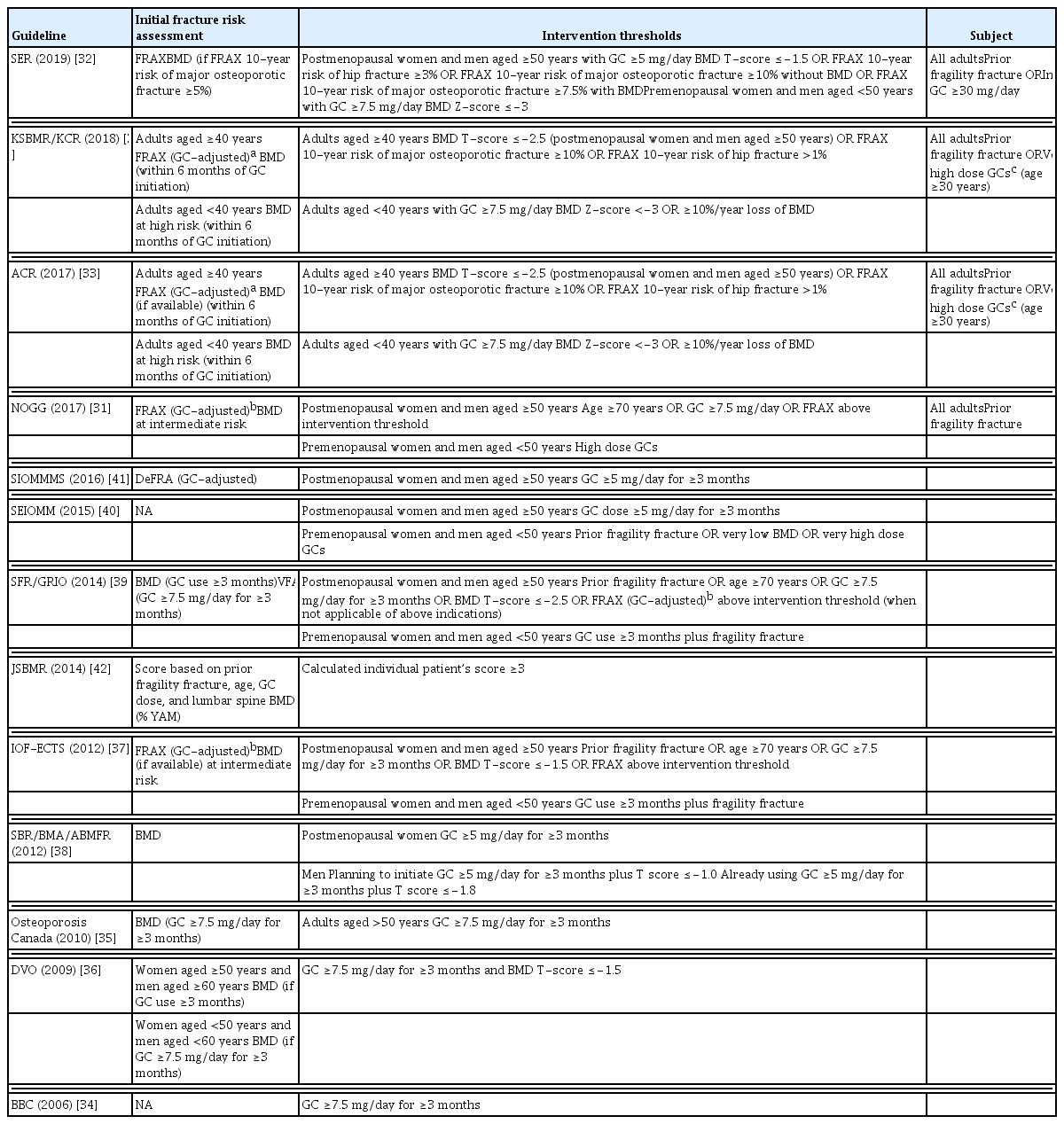
Initial Fracture Risk Assessment and Intervention Thresholds for Glucocorticoid Users in the Guidelines
Most guidelines recommend an assessment of fracture risk in patients beginning or receiving GC treatment for more than 3 months. Although most guidelines that were published before 2012 propose BMD assessment as a single tool for estimating the fracture risk, more recent criteria for identifying patients for preventive treatment have suggested country-specific fracture risk assessment tool (FRAX) values. Some guidelines use both tools to put steps in the assessment; the National Osteoporosis Guideline Group (NOGG) provides an algorithm to perform BMD when an individual is at intermediate risk, as calculated by the FRAX [31], and the Spanish Society of Rheumatology (SER) suggests that BMD is required if the 10-year probability of major osteoporotic fracture using FRAX is greater than 5% [32]. The guidelines from the American College of Rheumatology (ACR) [33] and the Korean Society for Bone and Mineral Research (KSBMR)/the Korean College of Rheumatology (KCR) [30] separately present recommendations for younger patients. They suggested that BMD should be assessed in adults under 40 years of age at a high fracture risk.
Guidelines for GIOP published before 2012 use only a single criterion for the treatment threshold, while recently published guidelines present several criteria for defining thresholds. A dose greater than 5 or 7.5 mg of prednisone equivalents per day with a 3-month minimum duration was proposed as a threshold for preventive treatment in most guidelines [31,34–41]. Additionally, according to some guidelines, GC users with prior fragility fractures or elderly patients are eligible for the initiation of anti-osteoporosis treatment, regardless of the presence of other risk factors.
A T-score of −2.5 in BMD is usually applied according to the guidelines for postmenopausal osteoporosis treatment; however, some guidelines suggest less stringent T-score values (≤−1.5). However, there is no established T-score threshold for bone-protective therapy in patients on GCs.
For FRAX, the intervention thresholds are divided into age-dependent thresholds and fixed thresholds, according to the guidelines. The International Osteoporosis Foundation and the European Calcified Tissue Society, the Bone Section of the French Society for Rheumatology (SFR)/Osteoporosis Research and Information Group (GRIO), and NOGG guidelines recommend treatment for postmenopausal women and men over 50 years of age whose 10-year probability of fracture estimated by the FRAX exceeds the intervention threshold at a given age. However, a fixed-probability intervention threshold was used for the ACR, KSBMR/KCR, and SER. The ACR and KSBMR/KCR guidelines recommend a 10% probability of a major osteoporotic fracture and a 1% probability of a hip fracture.
TREATMENT
Oral bisphosphonates, such as alendronate and risedronate, have been shown to be effective as first line therapies in preventing fractures in GIOP, in addition to adequate calcium and vitamin D supplementation [12]. However, long-term use of bisphosphonates is associated with an increased risk of adverse events such as jaw necrosis or atypical femur fractures [44].
For postmenopausal women, selective estrogen receptor modulators prevent bone loss and lower the risk of fracture by decreasing osteoblast and osteoclast bone turnover [45]. One controlled trial demonstrated that raloxifene is an option for the prevention of BMD loss in postmenopausal women receiving long-term GC treatment [46]. Recently, we performed an RCT to evaluate the effectiveness of bazedoxifene in postmenopausal women with RA receiving GCs. Although bazedoxifene improved BMD and reduced bone turnover markers, it failed to prove a significant BMD change exceeding the least significant change [47].
In recent years, more potent intravenous antiresorptive agents, zoledronic acid [48], and denosumab have been shown to be superior to risedronate in facilitating a BMD increase in patients with GIOP [49]. However, evidence for the effectiveness of denosumab is limited to increasing BMD; therefore, further observational studies are needed to evaluate the effectiveness of denosumab in preventing fractures. Previous studies have shown that denosumab has the potential to prevent joint destruction in patients with early RA [50,51]. Given that RA patients frequently use GCs at an early stage, which results in an increased risk of fracture due to GIOP, denosumab may be a potentially useful treatment option for early RA patients to protect both joint destruction and fracture occurrence. However, when using denosumab, it is important to recognize the rapid offset of the antiresorptive effect if the 6-monthly injection is delayed [52]. Short-term or intermittent use of denosumab probably has no net benefit, as discontinuation of treatment is followed by rapid bone loss and an increased risk of multiple vertebral fractures [52]. Both patients and doctors need to understand this phenomenon before therapy is initiated and have a strategy for transitioning to another therapy, such as a bisphosphonate, if denosumab is discontinued [53].
However, antiresorptive agents may not be sufficient for osteoporosis caused by long-term GC use, particularly within the low-turnover state of the disease [54]. The effect of GCs on bones is also mediated by a decrease in bone formation due to a decrease in osteoblast production and activity [55].
Anabolic therapies protect against bone degradation by promoting new bone growth, rather than by reducing remodeling [55]. Teriparatide, a subcutaneous recombinant human parathyroid hormone, is the first anabolic agent approved for the treatment of osteoporosis. Teriparatide was approved by the U.S. Food and Drug Administration for postmenopausal women and men with osteoporosis in 2002. Subsequently, its preventive effect on vertebral fractures in GIOP was presented, but not in nonvertebral fractures [56,57]. Abaloparatide, a daily subcutaneous parathyroid hormone-related protein analog, has demonstrated significantly increased BMD at the lumbar spine and hip and reduced fracture incidence compared to placebo in postmenopausal women [55]. However, their use in GIOP has not yet been established. Romosozumab is a monoclonal antibody against sclerostin, a protein secreted by osteocytes that inhibits bone formation through the regulation of osteoblasts [58]. Although it has shown benefits in postmenopausal women and men [54], it has not yet been studied in patients on chronic GC treatment.
CONCLUSIONS
Since the introduction of the FRAX, a FRAX-based assessment of fracture risk has been used. However, high-quality data on treatment thresholds to prevent GIOP are limited. Along with efforts to identify potential biomarkers to predict the risk of individual osteoporotic fractures [59], further delicate criteria for individual patients are needed. In addition, many osteoporotic fractures occur in patients with osteopenia, because even though the risk of fracture is lower in osteopenia than in osteoporosis, the number of subjects at risk is much higher in the osteopenic range [60]. Therefore, early intervention in patients receiving long-term GC therapy is crucial to prevent the progression of osteopenia to osteoporosis.
Notes
CONFLICTS OF INTEREST
Yoon-Kyoung Sung has received research grants from Bristol-Myers Squibb, Eisai, Pfizer, and JW Pharmaceutical. Soo-Kyung Cho has no conflict of interest.
