Receptor-Mediated Muscle Homeostasis as a Target for Sarcopenia Therapeutics
Article information
Abstract
Sarcopenia is a disease characterized by age-related decline of skeletal muscle mass and function. The molecular mechanisms of the pathophysiology of sarcopenia form a complex network due to the involvement of multiple interconnected signaling pathways. Therefore, signaling receptors are major targets in pharmacological strategies in general. To provide a rationale for pharmacological interventions for sarcopenia, we herein describe several druggable signaling receptors based on their role in skeletal muscle homeostasis and changes in their activity with aging. A brief overview is presented of the efficacy of corresponding drug candidates under clinical trials. Strategies targeting the androgen receptor, vitamin D receptor, Insulin-like growth factor-1 receptor, and ghrelin receptor primarily focus on promoting anabolic action using natural ligands or mimetics. Strategies involving activin receptors and angiotensin receptors focus on inhibiting catabolic action. This review may help to select specific targets or combinations of targets in the future.
INTRODUCTION
Sarcopenia is an age-related skeletal muscle disorder. The term “sarcopenia” was first introduced in the 1980s to refer to an agerelated loss of lean body mass that affects mobility, independence, and mortality [1]. In the past, there was a misconception that muscle loss with advancing age was an inevitable consequence of normal aging. Very recently, sarcopenia has been assigned to a new disease entity in the International Classification of Diseases, 10th and 11th revisions, although no drug has yet been approved for sarcopenia [2,3].
In 2010, the European Working Group on Sarcopenia in Older People (EWGSOP) published a working definition of sarcopenia. The EWGSOP2 recommends a 4-step diagnostic approach, Find-Assess-Confirm-Severity (F-A-C-S), with the measurement of three parameters (muscle strength, muscle quantity/quality, and physical performance) as an indicator of severity [4,5]. The Asian Working Group has provided diagnostic strategies and cutoff values for Asian populations to facilitate the study of sarcopenia in Asian countries [6]. The International Clinical Practice Guidelines for Sarcopenia published in 2018 suggest strong recommendations for combination treatment involving physical training and protein supplementation as the primary treatment for sarcopenia [7].
Several clinical studies have shown the possibility of pharmacological interventions for sarcopenia [8,9]. For example, vitamin D supplementation had the beneficial effect of increasing muscle fiber size by 10% in elderly women [10]. Testosterone improved walking distance in men, although it had a risk of cardiovascular side effects [11]. Treatment with the myostatin inhibitor LY2495655 increased muscle mass and partially improved physical performance in elderly patients with weakened muscle strength [12]. In elderly individuals with sarcopenia, treatment with bimagrumab (BYM-338), an inhibitor of activin type II receptors, increased skeletal muscle mass [13,14].
In order to design an effective strategy for the treatment and prevention of sarcopenia, it is necessary to understand its mechanisms and signaling pathways [15]. Herein, we provide insights into potential targets to support the study of sarcopenia with a focus on pharmacological strategies.
CELLULAR SIGNALING PATHWAYS AND MUSCLE AGING
Most age-related factors contribute to the development of sarcopenia, and senile disorders such as cancer, arthritis, and chronic obstructive pulmonary disease are associated with a higher risk of the sarcopenic phenotype [16,17]. Aging impairs the balance between muscle anabolic and catabolic pathways, which leads to a gradual loss of muscle mass and function [18]. Insulin-like growth factor-1 (IGF-1), testosterone, exercise, β2-adrenergic agents, and branched-chain amino acids are the major triggers of the anabolic pathway and are known to cause muscle hypertrophy through increased protein synthesis [19]. AKT/mammalian target of the rapamycin (mTOR) signaling activity induces phosphorylation of S6K and 4E-BP, leading to an increase in protein synthesis. AKT also regulates the initiation of translation through the glycogen synthase kinase 3β (GSK3β)-eukaryotic translation initiation factor 2B (eIF2B) pathway [20-22]. Conversely, myostatin, glucocorticoids, oxidative stress (reactive oxygen species and nitric oxide), inflammatory cytokines (tumor necrosis factor alpha and interleukin 6 [IL-6]), and metabolic stress induce catabolic pathways, leading to muscle atrophy. The forkhead box O (FOXO) and nuclear factor-κB pathways are crucial regulators of the expression of E3 ubiquitin ligases atrogin-1 and muscle RING-finger protein 1 (MuRF1), which play a dominant role in muscle atrophy. The activity of the FOXO transcription factor is regulated by AKT signaling [23,24].
Muscular homeostasis involves a very complex network comprising multiple interconnected signaling pathways [25,26]. Unlike genetically induced or disease-related muscle disorders, the key cause of sarcopenia is aging. Therefore, understanding the molecular events caused by aging guides the direction of pharmacological interventions and helps to avoid unwanted adverse events.
PHARMACOLOGICAL STRATEGIES FOR SARCOPENIA IN CLINICAL TRIALS
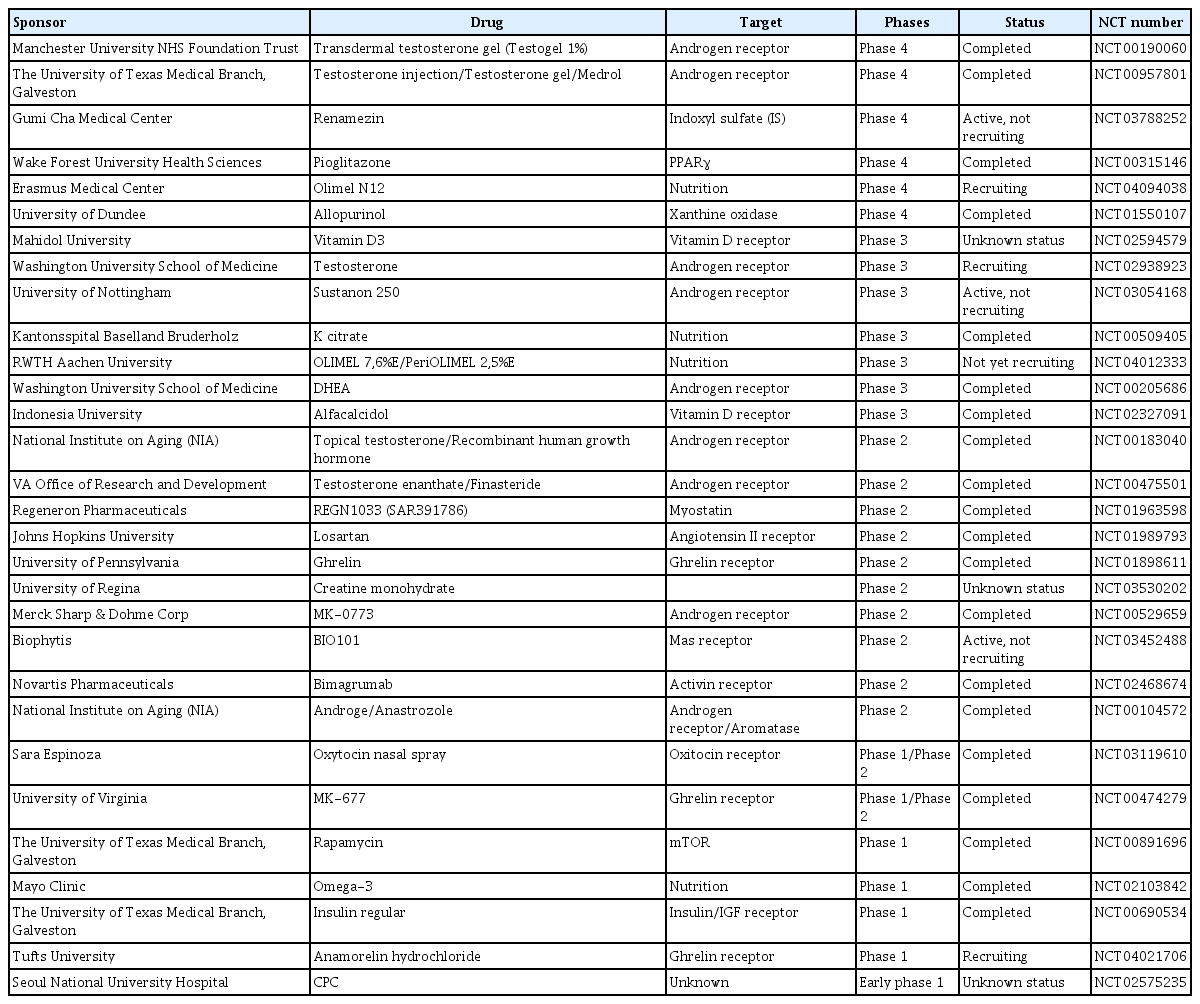
Over the course of decades, dozens of clinical studies have been conducted on pharmacotherapy for sarcopenia, and the relevant information is easily accessible through ClinicalTrials.gov (Table 1, Fig. 1). There are several pharmacologically validated strategies, including anabolic steroids, exercise mimetics, nutrient supplements, myokines, and monoclonal antibody modulation. Numerous drugs target biological signaling pathways, and most of them are signaling receptors (Fig. 2). Systematic explorations and statistical analyses have often shown representative signal receptors that are likely to be targets for pharmacological interventions (Fig. 3).

Cell signaling pathways and pharmacological strategies for sarcopenia. GHSR, growth hormone secretagogues receptor or ghrelin receptor; IGF-1R, insulin-like growth factor 1 receptor; ACE, angiotensin-converting enzyme; ActRIIB, activin type II receptor; Ang1, angiotensin I; Ang2, angiotensin II; AGTR1, angiotensin II receptor type 1; SARM, selective androgen receptor modulator; AR, androgen receptor; VDR, vitamin D receptor; mTOR, mammalian target of the rapamycin; FOXO, forkhead box O.

Distribution of target families of small-molecule drugs in sarcopenia. Related data are described on ClinicalTrials.gov, and the figure includes data up to March 2021. IS, indoxyl sulfate; XO, xanthine oxidase.
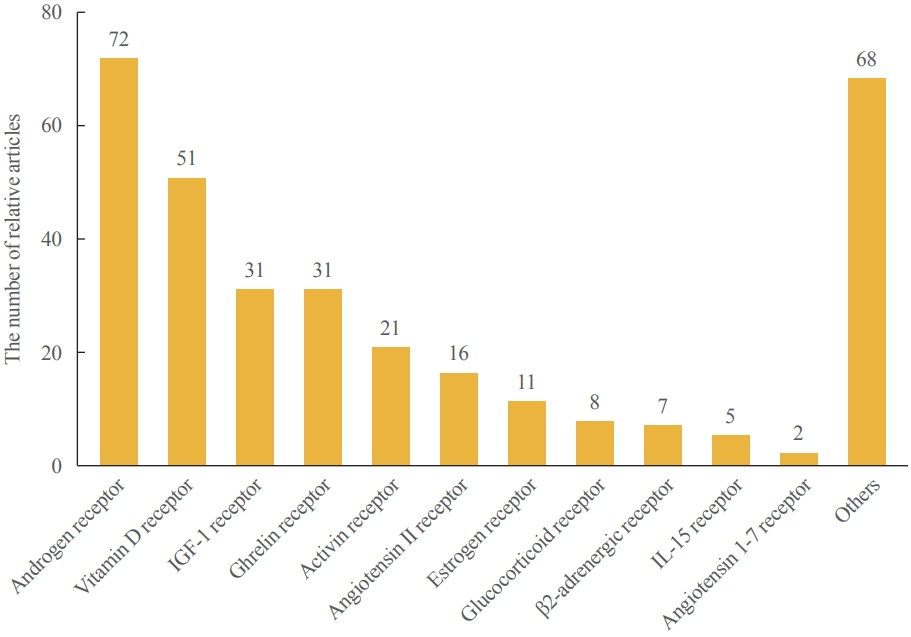
The current status of articles on sarcopenia and cell signaling receptors. A literature search was conducted in PubMed (keyword: sarcopenia, receptor, and/or related-term) and the relevant data extend up to March 2021. IGF-1, insulin-like growth factor 1; IL-15, interleukin 15.
Cellular signaling is usually transduced through four major types of receptors classified on the basis of their molecular structures and transduction mechanisms; ligand-gated ion channels, G-protein-coupled receptors (GPCRs), nuclear receptors, and receptor kinases [27]. These four major families of signaling proteins account for 44% of all human drug targets in the ChEMBL database. Moreover, 77% of small-molecule drugs target major signaling protein families (GPCRs 33%, ion channels 18%, kinases 3%, and nuclear receptors 16%) [28].
Intracellular receptors account for 10% to 15% of drugs on the market, including drugs that act on cytoplasmic receptors such as androgen receptors (ARs), estrogen receptors, progesterone receptors, and glucocorticoid receptors, and other drugs that act on nuclear receptors such as vitamin D receptor (VDR), thyroid hormone receptors, and peroxisome proliferator-activated receptors [27-30]. Ligands of intracellular receptors include lipophilic vitamins, steroid hormones, and small chemicals such as hydrogen peroxide and nitric oxide, which require membrane permeability for intracellular delivery [30,31]. There are several barriers to the intracellular delivery of therapeutic drugs, such as lysosome degradation and active efflux out of the cell. Lowmolecular-weight lipophilic compounds can diffuse directly into cells, whereas high-molecular-weight compounds usually need membrane transporters or endocytosis [32,33]. Proper entry into the cell and subsequent contact with the exact target lead to better therapeutic effects and reduce undesirable adverse effects [34].
The etiology of sarcopenia is not completely understood. Therefore, understanding the pathogenic mechanisms and collecting molecular targets should precede attempting pharmacological interventions as a therapeutic strategy. The following section provides a brief overview of the current knowledge of membrane or intracellular signaling receptors used as pharmacological interventions for sarcopenia.
Intracellular receptors
Androgen receptor
AR is a nuclear receptor, and its activity is regulated through the binding of androgenic steroids such as testosterone. Activated AR is translocated from the cytoplasm into the nucleus [35]. The AR gene is widely expressed in cells and tissues in the skeletal muscle environment, including myoblasts, myofibers, and satellite cells [36]. In normal men, androgens increase the level of circulating IGF-1, which is a powerful anabolic agent in muscles [37].
Testosterone is a major androgen secreted endogenously that interacts with skeletal muscle cells through binding to ARs. Testosterone levels gradually decrease after 30 years of age, and this decrease is associated with a decline in muscle mass and strength [38,39]. Furthermore, testosterone promotes regeneration by activating satellite cells [40]. Although there are side effects such as erythrocytosis and cardiovascular risk [41,42], few thromboembolic events have been reported in the literature, and the European Medicines Agency announced that “there is no consistent evidence that testosterone products have increased cardiovascular risk” [43,44]. Thus, testosterone therapy may be beneficial for those who have symptoms of low muscle mass and function caused by testosterone deficiency.
Selective androgen receptor modulators (SARMs) are a type of non-steroidal ligands that have an affinity for AR and exhibit anabolic properties specifically in muscles. SARMs were developed to avoid the side effects of androgens while taking advantage of the anabolic effects of AR agonists [45-47]. GTx-024 and MK-0773 are non-steroidal SARMs. Although not specifically targeted for sarcopenia, GTx-024 was evaluated as a potential treatment for muscle wasting and cachexia in completed phase 2 and 3 trials (NCT00467844; 2007, NCT01355497; 2011) [48,49]. The GTx-024 group exhibited a significant increase in total lean mass. In a phase 2 trial, treatment with MK-0773 in patients with sarcopenia showed a statistically significant improvement in total lean mass [50]. Therefore, SARMs have the potential to be applied as a treatment for sarcopenia.
Vitamin D receptor
VDR is a member of the nuclear receptor family and acts as a ligand-dependent transcription factor. When binding to calcitriol, an active metabolite of vitamin D, VDR moves to the nucleus and interacts with retinoid X receptor (RXR) to form heterodimers. The VDR/RXR complex binds to the vitamin D response element and regulates the expression of target genes such as the FOXO1 transcription factor and inhibitor of DNA-binding/differentiation (ID) gene, which is responsible for muscle growth, metabolism, and myoblast differentiation [51,52].
Human muscle immunohistochemistry showed that VDR expression decreased with age [53]. VDR plays a regulatory role in calcium homeostasis [54]. In rats, a diet without vitamin D causes hypocalcemia and muscle protein degradation, which can be partially corrected with calcium supplementation alone [55]. Vitamin D deficiency is associated with a variety of myopathies, including myalgia [56]. The muscle fibers of VDR-null mice were smaller than those of the control group, and the expression of early markers of muscle differentiation, such as myogenic factor 5 (myf5), myogenin, E2A, and early myosin heavy chain isoforms, was aberrantly high and persistent [57]. Myocyte-specific VDR-null mice showed a proportional decrease in lean mass, voluntary wheel-running distance, average running speed, and grip strength [58]. A statistical analysis of randomized controlled trials conducted between 1966 and January 2014 revealed that vitamin D supplementation significantly increased muscle strength, but did not affect muscle mass or muscle power [59].
International clinical practice guidelines for sarcopenia do not recommend vitamin D supplementation to patients with sarcopenia because of the lack of robust evidence [7], and there are no studies showing the results of using vitamin D as a strategy for sarcopenia on ClinicalTrials.gov. Further research on vitamin D and sarcopenia would be useful for formulating a pharmacological strategy.
Membrane receptors
IGF-1 receptor
The IGF-1 receptor (IGF1R) is a transmembrane receptor that is activated by IGF-1 (a growth hormone) and belongs to the large class of tyrosine kinase receptors. Activation of the IGF1R by IGF-1 binding induces multiple signaling pathways, including the phosphoinositide 3-kinase (PI3K)/Akt and mitogen-activated protein kinase (MAPK)/extracellular signal-regulated kinase (ERK) pathways, and leads to muscle hypertrophy through the promotion of anabolic effects [60]. Muscle-specific IGF-1 transgenic mice consistently showed persistent muscle hypertrophy [61]. Serum IGF-1 levels decrease during aging, and this decrease is associated with a variety of pathological conditions, including chronic disease, inflammation, and malnutrition [62,63]. In experimental muscle atrophy models such as dexamethasone-injected mice, muscular dystrophy, and hind limb suspension, local overexpression of IGF-1 was able to restore the weakened muscle [64-66]. In addition, adeno-associated virus-mediated IGF-1 gene transfer blocked aging-related muscle atrophy in old mice [67]. Because IGF-1 production is promoted by growth hormone, the side effects of IGF-1 may be similar to those from the abuse of other growth hormones. Lipohypertrophy and erythema at the injection site are very frequent side effects [68,69]. Other negative symptoms reported after administration of recombinant human IGF-I include myalgia, edema, hypoglycemia, seizures, jaw pain, headaches, altered liver function, and increased liver and kidney mass [69-72].
In the clinical stage, a phase 2 randomized clinical trial evaluating the efficacy of IGF-1 involved administering subcutaneous injections once daily for 6 months to boys with Duchenne muscular dystrophy [73]. The 6-minute walking distance change increased by 3.4 m in the IGF-1 treatment group and decreased by 5.1 m in the control group. The height velocity in the IGF-1 treatment group was 2.6 cm/year, and that in the control group was –0.06 cm/year (NCT01207908). More clinical studies are needed to determine whether IGF-1 administration could be a safe and effective therapeutic strategy for sarcopenia.
Ghrelin receptor
The ghrelin receptor is a seven-transmembrane GPCR that binds to growth hormone secretagogues (GHS) such as ghrelin. Ghrelin receptor expression occurs mainly in the brain, but lower levels are also found in the skeletal muscle and heart, as well as in peripheral organs such as the pancreas, spleen, kidneys, and adrenal glands [74,75]. Ghrelin is important for food intake, weight control, and glucose homeostasis. Ghrelin administration promotes the synthesis of nitric oxide and central regulation of food intake in the hypothalamus [76].
Although the level of muscle GHS receptors (GHSRs) is low, ghrelin exhibits anabolic properties in skeletal muscle. Ghrelin prevents muscle atrophy induced by cisplatin treatment or tumor implantation by upregulating AKT, MyoD, and myogenin and by inhibiting inflammatory cytokines, p38-CCAAT/enhancer binding proteins-β (C/EBP-β), and myostatin [77]. Ghrelin levels are generally lower in elderly adults than in middleaged adults. Elderly individuals with sarcopenia showed significantly lower ghrelin levels than those without sarcopenia [78]. In old mice, genetic deficiency of ghrelin showed an acceleration of fasting-induced muscle atrophy and a decrease in the percentage of lean mass and wheel running distance [79]. In healthy elderly individuals, the oral administration of ibutamoren (MK-677), a non-peptide agonist of ghrelin receptors, increased serum growth hormone and IGF-1 levels without serious side effects, but failed to induce a body weight increase [80]. A phase 2 study showed that anamorelin, a GHSR agonist, improved lean body mass, performance status, and quality of life in patients with non-small cell lung cancer [81].
Overall, ghrelin and other small molecule agonists of the ghrelin receptor increase food intake and stimulate muscle anabolic action. Therefore, these could be attractive candidates for the treatment of sarcopenia.
Activin type II receptors
The activin type II receptors (ActRIIA or ActRIIB) belong to the larger transforming growth factor-β (TGF-β) receptor family. These receptors are involved in a variety of physiological and cellular processes including embryonic development and cellular homeostasis [82]. The myostatin/activin type IIB receptor pathway is a major signaling pathway that regulates muscle growth and maintenance [83,84]. In the membrane, the binding of ActRIIB to its natural ligands such as activin, myostatin, and growth differentiation factor 11 (GDF11) activates the activin receptor-like kinase (ALK4 or ALK5), and then leads to the phosphorylation of SMAD2/3. Phosphorylated SMAD2/3 forms a heterotrimeric complex with SMAD4 to regulate gene expression and intracellular signaling [85,86]. Activation of SMAD2/3 signaling leads to inactivation of AKT signaling and subsequent dephosphorylation of FOXO3, which not only inhibits protein synthesis, but also promotes proteolysis through the ubiquitin-proteasome system [87,88].
Myostatin, known as GDF8, is a member of the TGF-β superfamily and is a molecular target that has been intensively studied for muscle wasting disorders. Myostatin is mainly expressed in cells of the skeletal muscle lineage and inhibits the growth and differentiation of muscle cells [89-91]. Overexpression of the myostatin gene in mice caused skeletal muscle atrophy [92], while pharmacological and genetic blockade of ActRIIB induced muscle hypertrophy in mice [93]. Patients without mature myostatin caused by genetic mutations have been shown to have increased muscle mass and strength [94].
The first human trial of myostatin inhibitors was to evaluate the efficacy of stamulumab (MYO-029) in patients with muscular dystrophy. The phase 2 clinical trial failed to show efficacy for increasing muscle strength and the study was discontinued [95]. Bimagrumab (BYM-338) is a human monoclonal antibody with a high affinity for ActIIRB and inhibits downstream signaling by preventing ligand-receptor binding. A phase 2 study to determine the effectiveness of BYM-338 in elderly with sarcopenia and mobility restrictions showed that 16 weeks of BYM-338 treatment increased skeletal muscle mass and strength and improved mobility (NCT01601600) [13]. Since then, Novartis conducted a phase 2 clinical trial to confirm the safety and efficacy of bimagrumab with adequate nutrition and light exercise (NCT02333331). Six months of bimagrumab treatment showed good safety, increased lean body mass, and reduced fat mass, but there was no significant difference in physical performance [14].
In the meantime, numerous myostatin inhibitory agents have been developed (domagrozumab [PF-06252616], landogrozumab [LY-2495655], trevogrumab [REGN-1033], ramatercept [ACE-031], and ACE-083). Many of them have progressed to human trials [96]. Inhibition of ActRIIB-related ligands has been reported to induce beneficial hypertrophy in muscles, but it can cause serious side effects on other tissues. Overexpression of follistatin (FST), an myostatin (MSTN)/GDF11 inhibitor, reduced bone mineral density and induced tibia fracture in mice [97]. Administration of ACE-031 to boys with Duchenne muscular dystrophy has shown a potential risk of epistaxis and telangiectasia [98].
Activin and bone morphogenetic protein (BMP) are expressed in a variety of tissues, including muscle, and regulate the development of many cell types. These members of the TGF-β family, including myostatin, have high similarities in their recognition sites, indicating that inhibition of one of these ligands may cause unwanted side effects in other tissues [96]. In fact, several myostatin inhibitors have cross-reactivity with activin A, BMPs, and GDF11 [99]. Therefore, the study of drug delivery systems for precise targeting would be helpful for the development of myostatin-related drugs.
Angiotensin II receptor
Angiotensin II receptor type 1 (AGTR1 or AT1) is a member of the G protein-coupled receptor family, and angiotensin II, a ligand of AGTR1, is an important bioactive molecule of the renin-angiotensin system, which manages blood pressure, fluid retention, and electrolyte balance [100]. Disruption of the Agtr1a gene, which encodes AT1A, prolongs the lifespan of mice through the prevention of cardiac hypertrophy and fibrosis [101], and pharmacological blockade of AGTR1 improves muscle repair and regeneration by inhibiting the activity of the C1q-Wnt/β-catenin signaling pathway and canonical TGF-β signaling pathway [102-104]. However, persistent and excessive activation of AGTR1 causes a variety of age-related diseases such as cardiovascular disease [105].
Inhibitors of angiotensin-converting enzyme (ACE), which plays a role in converting angiotensin I to angiotensin II, have been mainly used as primary and secondary prophylactic treatments for cardiovascular disease [106]. ACE inhibitors are known to increase the level of IGF-I, a hormone that stimulates muscle growth, and the number of mitochondria [107], suggesting beneficial effects of ACE inhibitors on sarcopenia. Perindopril, a long-acting ACE inhibitor, improved walking distance and reduced hip fracture in elderly individuals with vascular diseases [108,109]. In addition, treatment of losartan, a selective and competitive AGTR1 antagonist, resulted in a reduction of serum TGF-β1 levels and subsequent inhibition of SMAD2/3 activity, leading to muscle remodeling and functional recovery in sarcopenic mice [104,110]. In humans, a phase 2 trial of losartan for the prevention of sarcopenia has been completed (NCT01989793). However, the results have not yet been reported. Additional evidence should be provided before recommending ACE inhibitors or ATR antagonists to prevent sarcopenia.
CONCLUSIONS
Sarcopenia patients have health problems related to physical activity and comorbidities such as osteoporosis, obesity, diabetes, and cognitive disorders [8,111]. There is no U.S. Food and Drug Administration-approved drug for sarcopenia to date, and nonpharmacological interventions such as resistance training and nutritional supplementation are common and applicable alternative strategies.
Pharmacological interventions such as myostatin inhibitors, testosterone, and SARMs clinically show the potential to prevent sarcopenia. However, they have not been successful in terms of safety due to erythrocytosis, cardiovascular risk, epistaxis, and telangiectasis. As sarcopenia treatment requires longterm therapy, safety issues should be considered very carefully. Although not covered in this paper, targeting the Mas receptors and ryanodine receptors could be a potential pharmacological strategy. Angiotensin 1-7 peptide, a ligand of the Mas receptor, has a preventive effect on immobilization-induced muscle atrophy [112]. Sarconeos (BIO101), the Mas receptor activator, is undergoing a phase 2 clinical trial (NCT03452488, 2018). Treatment with ARM210 (S48168), a RyR calcium channel stabilizer, improved muscle function and histology in an Mdx mouse model without side effects, and a clinical trial has been done but has not been reported yet [113].
Drug delivery systems are an emerging field in sarcopenia drug development. This strategy aims to properly expose the drug to the site of action of skeletal muscle, while sparing unwanted organs [114,115]. Adeno-associated virus, muscle-targeting delivery systems, nanoparticles, and extracellular vesicles are current promising drug delivery systems [116]. Gold nanoparticles conjugated to the IL-4 cytokine are more stable than soluble IL-4 in vitro and exhibit faster muscle regeneration in vivo [117]. Exosome-mediated delivery of myostatin inhibitor showed increased serum stability, delivery efficiency, and regenerative efficacy in Mdx mice [118].
The molecular mechanisms of sarcopenia form a very complex network due to simultaneous reactions of multiple risk factors and interactions between elements of the network. Therefore, patient typing with a variety of biomarkers will facilitate drug efficacy, thereby enabling personalized medicine in sarcopenia. Diversified strategies involving combinations of drugs targeting different pathways may also increase the success of interventions for sarcopenia.
Notes
CONFLICTS OF INTEREST
Ki-Sun Kwon also serves as the chief executive officer of Aventi Inc., but does not have any conflicts of interest. Jong-Hyeon Yoon does not have any conflicts of interest to declare.
Acknowledgements
This work was supported by grants from the Bio & Medical Technology Development Program (2017M3A9D8048708, Ki-Sun Kwon) of the National Research Foundation (NRF) funded by the Korean government (Ministry of Science and ICT) and the KRIBB Research Initiative Program.