Search
- Page Path
- HOME > Search
- Adrenal gland
- The Fascinating Interplay between Growth Hormone, Insulin-Like Growth Factor-1, and Insulin
- Eline C. Nijenhuis-Noort, Kirsten A. Berk, Sebastian J. C. M. M. Neggers, Aart J. van der Lely
- Endocrinol Metab. 2024;39(1):83-89. Published online January 9, 2024
- DOI: https://doi.org/10.3803/EnM.2024.101
- 1,455 View
- 109 Download
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
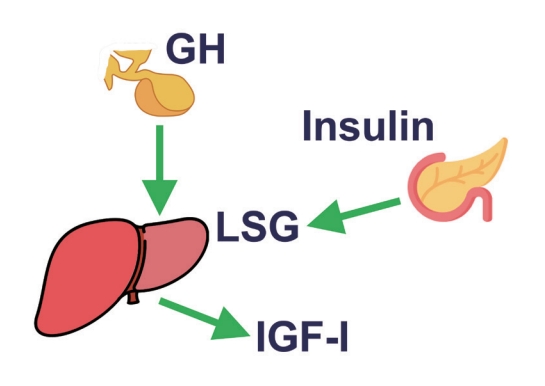
ePub - This review intends to provide the reader with a practical overview of several (patho)physiological conditions in which knowledge of the interplay between growth hormone (GH), insulin-like growth factor-1 (IGF-1), and insulin is important. This might help treating physicians in making the right decisions on how to intervene and improve metabolism for the benefit of patients, and to understand why and how metabolism responds in their specific cases. We will specifically address the interplay between GH, IGF-1, and insulin in type 1 and 2 diabetes mellitus, liver cirrhosis, and acromegaly as examples in which this knowledge is truly necessary.
-
Citations
Citations to this article as recorded by- IGF-1 and IGF-2 as Molecules Linked to Causes and Consequences of Obesity from Fetal Life to Adulthood: A Systematic Review
Justyna Szydlowska-Gladysz, Adrianna Edyta Gorecka, Julia Stepien, Izabela Rysz, Iwona Ben-Skowronek
International Journal of Molecular Sciences.2024; 25(7): 3966. CrossRef
- IGF-1 and IGF-2 as Molecules Linked to Causes and Consequences of Obesity from Fetal Life to Adulthood: A Systematic Review

- Calcium & bone metabolism
- Acromegaly and Bone: An Update
- Andrea Giustina
- Endocrinol Metab. 2023;38(6):655-666. Published online December 22, 2023
- DOI: https://doi.org/10.3803/EnM.2023.601
- 1,264 View
- 103 Download
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
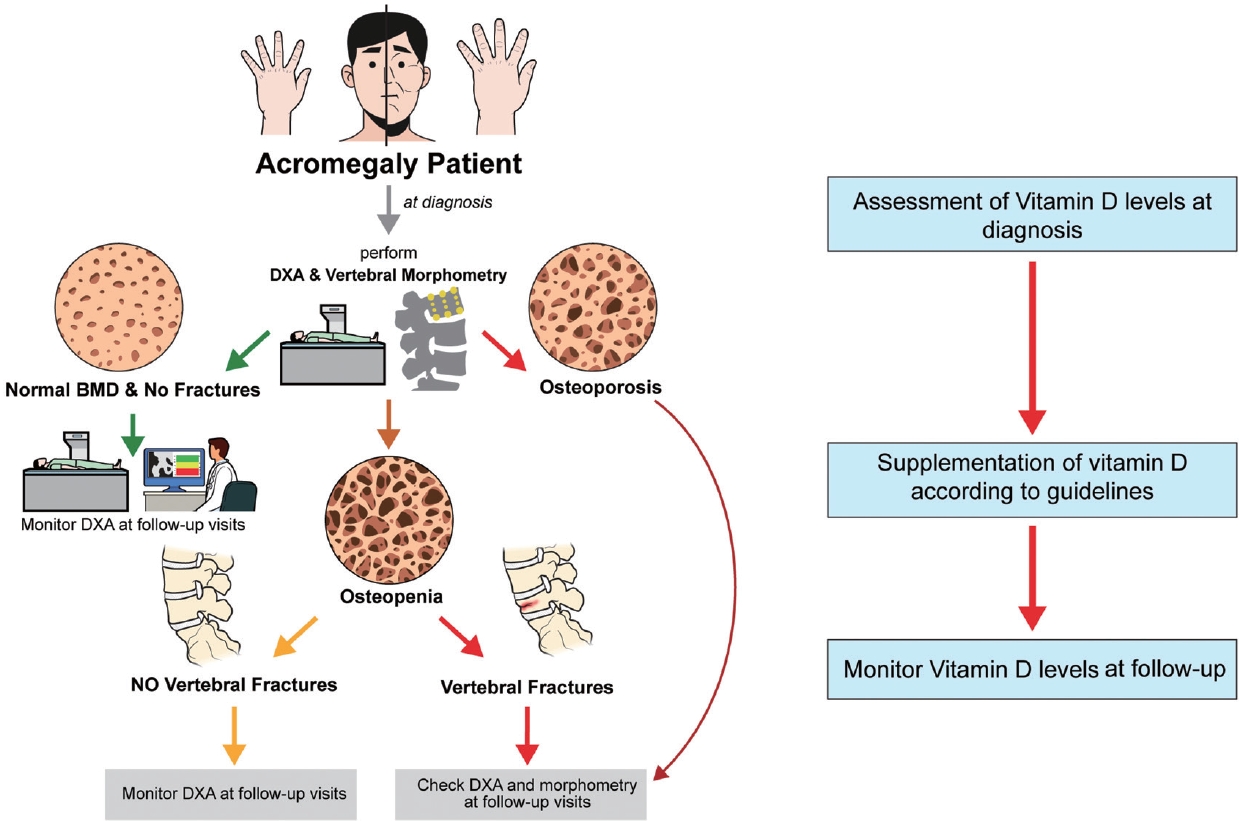
ePub - Since our discovery in 2006 that acromegaly is associated with an increased risk of vertebral fractures, many authors have confirmed this finding in both cross-sectional and prospective studies. Due to the high epidemiological and clinical impact of this newly discovered comorbidity of acromegaly, this topic has progressively become more important and prominent over the years, and the pertinent literature has been enriched by new findings on the pathophysiology and treatment. The aim of this narrative review was to discuss these novel findings, integrating them with the seminal observations, in order to give the reader an updated view of how the field of acromegaly and bone is developing, from strong clinical observations to a mechanistic understanding and possible prevention and treatment.

- Hypothalamus and pituitary gland
- Multiomics Approach to Acromegaly: Unveiling Translational Insights for Precision Medicine
- Kyungwon Kim, Cheol Ryong Ku, Eun Jig Lee
- Endocrinol Metab. 2023;38(5):463-471. Published online October 13, 2023
- DOI: https://doi.org/10.3803/EnM.2023.1820
- 1,653 View
- 117 Download
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
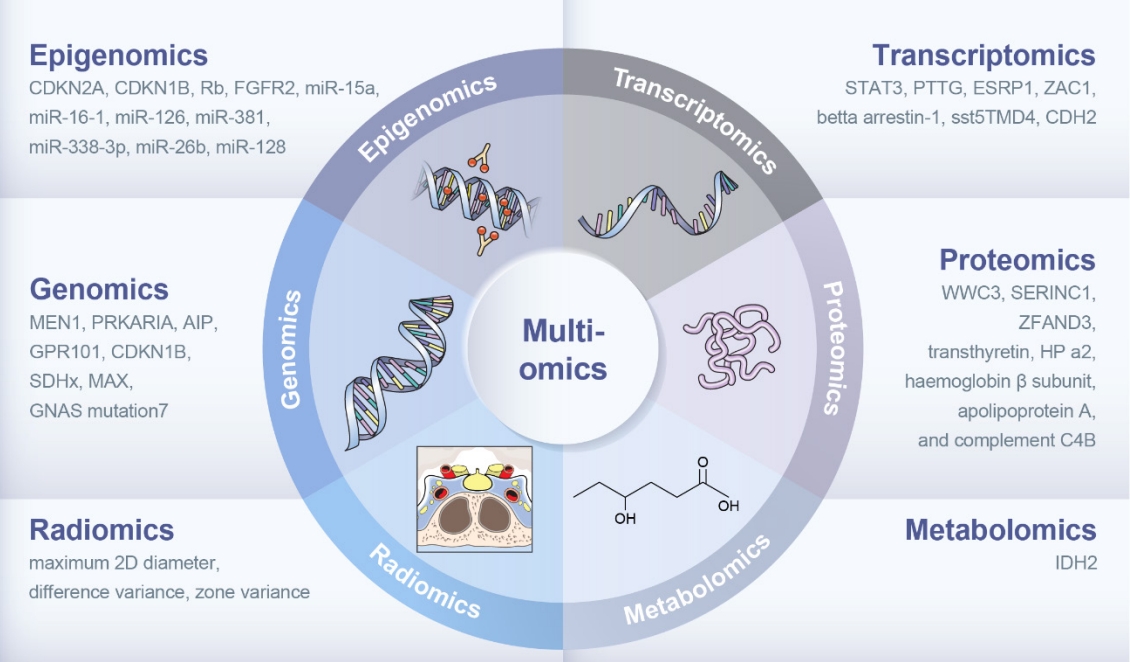
ePub - The clinical characteristics and prognoses of acromegaly vary among patients. Assessment of current and novel predictors can lead to multilevel categorization of patients, allowing integration into new clinical guidelines and a reduction in the increased morbidity and mortality associated with acromegaly. Despite advances in the diagnosis and treatment of acromegaly, its pathophysiology remains unclear. Recent advancements in multiomics technologies, including genomics, transcriptomics, proteomics, metabolomics, and radiomics, have offered new opportunities to unravel the complex pathophysiology of acromegaly. This review comprehensively explores the emerging role of multiomics approaches in elucidating the molecular landscape of acromegaly. We discuss the potential implications of multiomics data integration in the development of novel diagnostic tools, identification of therapeutic targets, and the prospects of precision medicine in acromegaly management. By integrating diverse omics datasets, these approaches can provide valuable insights into disease mechanisms, facilitate the identification of diagnostic biomarkers, and identify potential therapeutic targets for precision medicine in the management of acromegaly.
-
Citations
Citations to this article as recorded by- “Micromegaly”: Acromegaly with apparently normal GH, an entity on its own?
Lucio Vilar, Luciana Ansaneli Naves, Manoel Ricardo Alves Martins, Antônio Ribeiro-Oliveira Jr
Best Practice & Research Clinical Endocrinology & Metabolism.2024; : 101878. CrossRef
- “Micromegaly”: Acromegaly with apparently normal GH, an entity on its own?

- Hypothalamus and Pituitary Gland
- Independent Skeletal Actions of Pituitary Hormones
- Se-Min Kim, Farhath Sultana, Funda Korkmaz, Daria Lizneva, Tony Yuen, Mone Zaidi
- Endocrinol Metab. 2022;37(5):719-731. Published online September 28, 2022
- DOI: https://doi.org/10.3803/EnM.2022.1573
- 3,583 View
- 231 Download
- 4 Web of Science
- 4 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
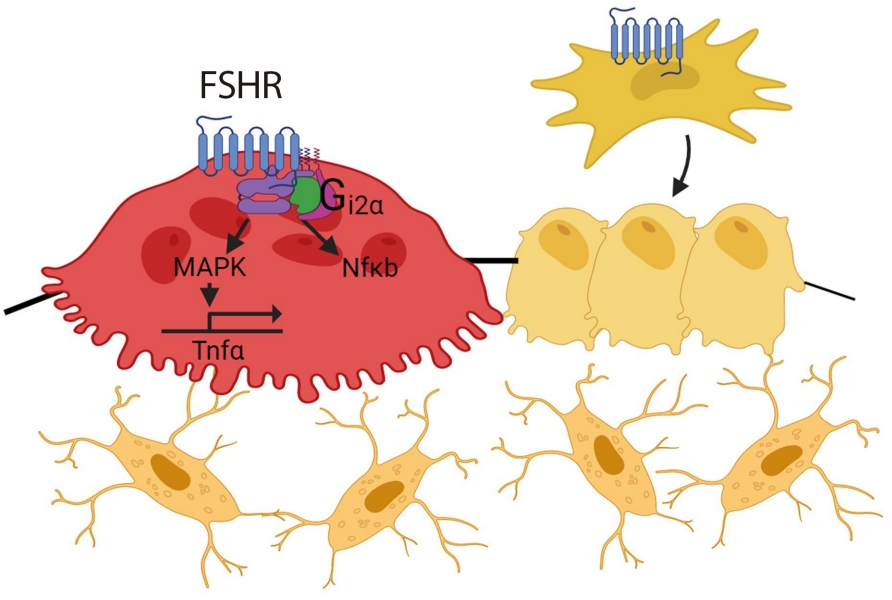
ePub - Over the past years, pituitary hormones and their receptors have been shown to have non-traditional actions that allow them to bypass the hypothalamus-pituitary-effector glands axis. Bone cells—osteoblasts and osteoclasts—express receptors for growth hormone, follicle stimulating hormone (FSH), thyroid stimulating hormone (TSH), adrenocorticotrophic hormone (ACTH), prolactin, oxytocin, and vasopressin. Independent skeletal actions of pituitary hormones on bone have been studied using genetically modified mice with haploinsufficiency and by activating or inactivating the receptors pharmacologically, without altering systemic effector hormone levels. On another front, the discovery of a TSH variant (TSH-βv) in immune cells in the bone marrow and skeletal action of FSHβ through tumor necrosis factor α provides new insights underscoring the integrated physiology of bone-immune-endocrine axis. Here we discuss the interaction of each pituitary hormone with bone and the potential it holds in understanding bone physiology and as a therapeutic target.
-
Citations
Citations to this article as recorded by- New tools for bone health assessment in secreting pituitary adenomas
Meliha Melin Uygur, Stefano Frara, Luigi di Filippo, Andrea Giustina
Trends in Endocrinology & Metabolism.2023; 34(4): 231. CrossRef - A Causality between Thyroid Function and Bone Mineral Density in Childhood: Abnormal Thyrotropin May Be Another Pediatric Predictor of Bone Fragility
Dongjin Lee, Moon Ahn
Metabolites.2023; 13(3): 372. CrossRef - The mechanism of oxytocin and its receptors in regulating cells in bone metabolism
Liu Feixiang, Feng Yanchen, Li Xiang, Zhang Yunke, Miao Jinxin, Wang Jianru, Lin Zixuan
Frontiers in Pharmacology.2023;[Epub] CrossRef - To investigate the mechanism of Yiwei Decoction in the treatment of premature ovarian insufficiency-related osteoporosis using transcriptomics, network pharmacology and molecular docking techniques
Weisen Fan, Yan Meng, Jing Zhang, Muzhen Li, Yingjie Zhang, Xintian Qu, Xin Xiu
Scientific Reports.2023;[Epub] CrossRef
- New tools for bone health assessment in secreting pituitary adenomas

- Hypothalamus and Pituitary Gland
- Metabolic Impacts of Discontinuation and Resumption of Recombinant Human Growth Hormone Treatment during the Transition Period in Patients with Childhood-Onset Growth Hormone Deficiency
- Yun Jeong Lee, Yunha Choi, Han-Wook Yoo, Young Ah Lee, Choong Ho Shin, Han Saem Choi, Ho-Seong Kim, Jae Hyun Kim, Jung Eun Moon, Cheol Woo Ko, Moon Bae Ahn, Byung-Kyu Suh, Jin-Ho Choi
- Endocrinol Metab. 2022;37(2):359-368. Published online April 25, 2022
- DOI: https://doi.org/10.3803/EnM.2021.1384
- 4,326 View
- 180 Download
- 3 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Discontinuing growth hormone (GH) treatment during the transition to adulthood has been associated with adverse health outcomes in patients with childhood-onset growth hormone deficiency (CO-GHD). This study investigated the metabolic changes associated with interrupting GH treatment in adolescents with CO-GHD during the transition period.
Methods
This study included 187 patients with CO-GHD who were confirmed to have adult GHD and were treated at six academic centers in Korea. Data on clinical parameters, including anthropometric measurements, metabolic profiles, and bone mineral density (BMD) at the end of childhood GH treatment, were collected at the time of re-evaluation for GHD and 1 year after treatment resumption.
Results
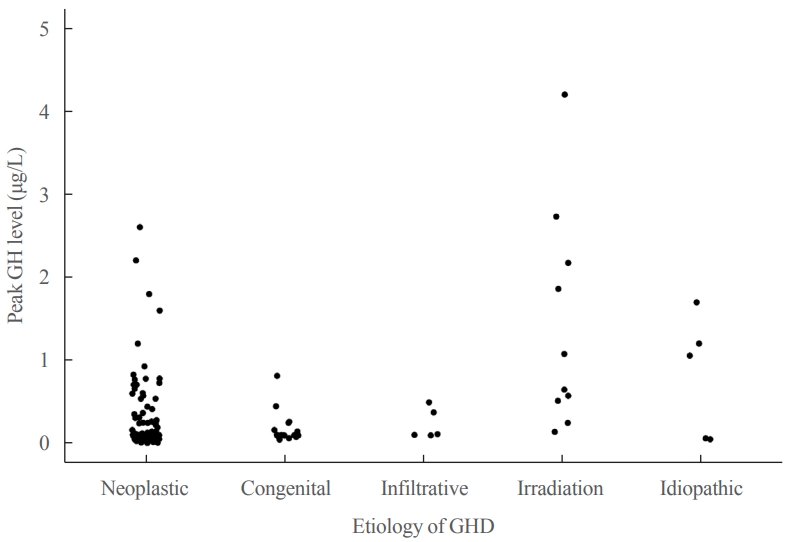
Most patients (n=182, 97.3%) had organic GHD. The median age at treatment discontinuation and re-evaluation was 15.6 and 18.7 years, respectively. The median duration of treatment interruption was 2.8 years. During treatment discontinuation, body mass index Z-scores and total cholesterol, low-density lipoprotein, and non-high-density lipoprotein (HDL) cholesterol levels increased, whereas fasting glucose levels decreased. One year after GH treatment resumption, fasting glucose levels, HDL cholesterol levels, and femoral neck BMD increased significantly. Longer GH interruption (>2 years, 60.4%) resulted in worse lipid profiles at re-evaluation. The duration of interruption was positively correlated with fasting glucose and non-HDL cholesterol levels after adjusting for covariates.
Conclusion
GH treatment interruption during the transition period resulted in worse metabolic parameters, and a longer interruption period was correlated with poorer outcomes. GH treatment should be resumed early in patients with CO-GHD during the transition period. -
Citations
Citations to this article as recorded by- Ghrelin regulating liver activity and its potential effects on liver fibrosis and Echinococcosis
Jiang Zhu, Tanfang Zhou, Meng Menggen, Kalibixiati Aimulajiang, Hao Wen
Frontiers in Cellular and Infection Microbiology.2024;[Epub] CrossRef - Relationship between the Stimulated Peak Growth Hormone Level and Metabolic Parameters in Children with Growth Hormone Deficiency
Seong Yong Lee
The Ewha Medical Journal.2023;[Epub] CrossRef - Dyslipidaemia and growth hormone deficiency – A comprehensive review
Matthias Hepprich, Fahim Ebrahimi, Emanuel Christ
Best Practice & Research Clinical Endocrinology & Metabolism.2023; 37(6): 101821. CrossRef
- Ghrelin regulating liver activity and its potential effects on liver fibrosis and Echinococcosis

- Miscellanenous
- Cushing Syndrome Associated Myopathy: It Is Time for a Change
- Martin Reincke
- Endocrinol Metab. 2021;36(3):564-571. Published online June 18, 2021
- DOI: https://doi.org/10.3803/EnM.2021.1069
- 4,719 View
- 170 Download
- 16 Web of Science
- 15 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
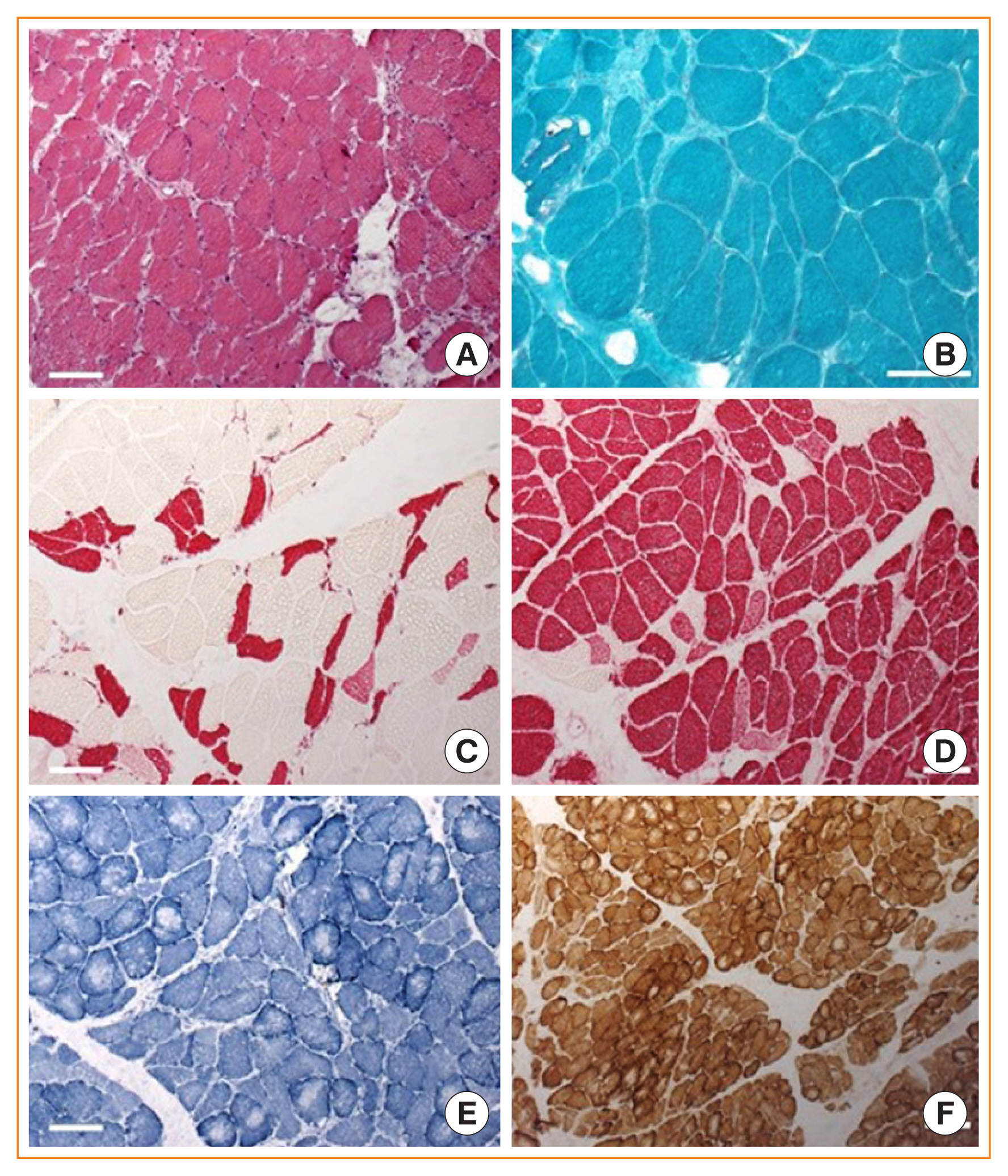
ePub - Cushing syndrome is the result of excessive levels of glucocorticoids. Endogenous Cushing syndrome is rare with an incidence of two to three cases per million per year. Clinically, the presentation consists of a characteristic phenotype including skin symptoms and metabolic manifestations. A frequent co-morbidity with high impact on quality of life is Cushing syndrome associated myopathy. It characteristically affects the proximal myopathy, impairing stair climbing and straightening up. The pathophysiology is complex and involves protein degradation via the forkhead box O3 (FOXO3) pathway, intramuscular fat accumulation, and inactivity-associated muscle atrophy. Surgical remission of Cushing syndrome is the most important step for recovery of muscle function. Restoration depends on age, co-morbidities and postoperative insulin-like growth factor concentrations. At average, functionality remains impaired during the long-term compared to age and sex matched control persons. Growth hormone therapy in individuals with impaired growth hormone secretion could be an option but has not been proved in a randomized trial.
-
Citations
Citations to this article as recorded by- Long-Term Consequences of Cushing Syndrome: A Systematic Literature Review
Soraya Puglisi, Anna Maria Elena Perini, Cristina Botto, Francesco Oliva, Massimo Terzolo
The Journal of Clinical Endocrinology & Metabolism.2024; 109(3): e901. CrossRef - The Link between Mitochondrial Dysfunction and Sarcopenia: An Update Focusing on the Role of Pyruvate Dehydrogenase Kinase 4
Min-Ji Kim, Ibotombi Singh Sinam, Zerwa Siddique, Jae-Han Jeon, In-Kyu Lee
Diabetes & Metabolism Journal.2023; 47(2): 153. CrossRef - Intelligent Systems for Muscle Tracking: A Review on Sensor‐Algorithm Synergy
Arjun Putcha, Tien Nguyen, Regina Smith, Rachel Choffin, Wubin Bai
Advanced Intelligent Systems.2023;[Epub] CrossRef - Sarcopenia in youth
Han Na Jung, Chang Hee Jung, You-Cheol Hwang
Metabolism.2023; 144: 155557. CrossRef - Cushing Syndrome
Martin Reincke, Maria Fleseriu
JAMA.2023; 330(2): 170. CrossRef - Musculoskeletal complications of Cushing syndrome
Dorota Leszczyńska, Alicja Szatko, Lucyna Papierska, Wojciech Zgliczyński, Piotr Glinicki
Rheumatology.2023; 61(4): 271. CrossRef - Circulating myomiRNAs as biomarkers in patients with Cushing’s syndrome
C. Pivonello, R. Patalano, C. Simeoli, T. Montò, M. Negri, F. Amatrudo, N. Di Paola, A. Larocca, E. M. Crescenzo, R. Pirchio, D. Solari, C. de Angelis, R. S. Auriemma, L. M. Cavallo, A. Colao, R. Pivonello
Journal of Endocrinological Investigation.2023; 47(3): 655. CrossRef - Low-grade inflammation during the glucocorticoid withdrawal phase in patients with Cushing's syndrome
Frederick Vogel, Leah Braun, Stephanie Zopp, Elisabeth Nowak, Jochen Schreiner, Irina Benz, German Rubinstein, Heike Künzel, Katrin Ritzel, Matthias Kroiss, Jürgen Honegger, Felix Beuschlein, Katharina Schilbach, Daniel Teupser, Martin Bidlingmaier, Marti
European Journal of Endocrinology.2023; 188(4): 375. CrossRef - An odd case of immune-mediated necrotizing myopathy, complicated with sagittal, transverse and sigmoid sinus thrombosis
Andreea Trandafir, Violeta Claudia Bonjincă, Delia Tulba, Gelu Onose
Balneo and PRM Research Journal.2023; 14(Vol.14, no): 630. CrossRef - Morbidität und Mortalität beim Cushing-Syndrom
Frederick Vogel, Leah Braun, Martin Reincke
Der Internist.2022; 63(1): 34. CrossRef - Pathophysiology of Mild Hypercortisolism: From the Bench to the Bedside
Vittoria Favero, Arianna Cremaschi, Chiara Parazzoli, Alberto Falchetti, Agostino Gaudio, Luigi Gennari, Alfredo Scillitani, Fabio Vescini, Valentina Morelli, Carmen Aresta, Iacopo Chiodini
International Journal of Molecular Sciences.2022; 23(2): 673. CrossRef - Long‐term morbidity and mortality in patients with Cushing's syndrome
Leah T. Braun, Frederick Vogel, Martin Reincke
Journal of Neuroendocrinology.2022;[Epub] CrossRef - Clinical presentation and etiology of Cushing's syndrome: Data from ERCUSYN
Elena Valassi
Journal of Neuroendocrinology.2022;[Epub] CrossRef - Pyruvate dehydrogenase kinase 4 promotes ubiquitin–proteasome system‐dependent muscle atrophy
Ibotombi Singh Sinam, Dipanjan Chanda, Themis Thoudam, Min‐Ji Kim, Byung‐Gyu Kim, Hyeon‐Ji Kang, Jung Yi Lee, Seung‐Hoon Baek, Shin‐Yoon Kim, Bum Jin Shim, Dongryeol Ryu, Jae‐Han Jeon, In‐Kyu Lee
Journal of Cachexia, Sarcopenia and Muscle.2022; 13(6): 3122. CrossRef - Consensus on diagnosis and management of Cushing's disease: a guideline update
Maria Fleseriu, Richard Auchus, Irina Bancos, Anat Ben-Shlomo, Jerome Bertherat, Nienke R Biermasz, Cesar L Boguszewski, Marcello D Bronstein, Michael Buchfelder, John D Carmichael, Felipe F Casanueva, Frederic Castinetti, Philippe Chanson, James Findling
The Lancet Diabetes & Endocrinology.2021; 9(12): 847. CrossRef
- Long-Term Consequences of Cushing Syndrome: A Systematic Literature Review

- Clinical Study
- Associations of GNAS Mutations with Surgical Outcomes in Patients with Growth Hormone-Secreting Pituitary Adenoma
- Hyein Jung, Kyungwon Kim, Daham Kim, Ju Hyung Moon, Eui Hyun Kim, Se Hoon Kim, Cheol Ryong Ku, Eun Jig Lee
- Endocrinol Metab. 2021;36(2):342-350. Published online March 23, 2021
- DOI: https://doi.org/10.3803/EnM.2020.875

- 4,271 View
- 142 Download
- 4 Web of Science
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
The guanine nucleotide-binding protein, alpha stimulating (GNAS) gene has been associated with growth hormone (GH)-secreting pituitary adenoma. We investigated the prevalence of GNAS mutations in Korean patients with acromegaly and assessed whether mutation status correlated with biochemical or clinical characteristics.
Methods
We studied 126 patients with acromegaly who underwent surgery between 2005 and 2014 at Severance Hospital. We performed GNAS gene analysis and evaluated age, sex, hormone levels, postoperative biochemical remission, and immunohistochemical staining results of the tumor.
Results
GNAS mutations were present in 75 patients (59.5%). Patients with and without GNAS mutations showed similar age distribution and Knosp classification. The proportion of female patients was 76.5% and 48.0% in the GNAS-negative and GNAS-mutation groups, respectively (P=0.006). In immunohistochemical staining, the GNAS-mutation group showed higher GH expression in pituitary tumor tissues than the mutation-negative group (98.7% vs. 92.2%, P=0.015). Patients with GNAS mutations had higher preoperative insulin-like growth factor-1 levels (791.3 ng/mL vs. 697.0 ng/mL, P=0.045) and lower immediate postoperative basal (0.9 ng/mL vs. 1.0 ng/mL, P=0.191) and nadir GH levels (0.3 ng/mL vs. 0.6 ng/mL, P=0.012) in oral glucose tolerance tests. Finally, the GNAS-mutation group showed significantly higher surgical remission rates than the mutation-negative group, both at 1 week and 6 months after surgical resection (70.7% vs. 54.9%, P=0.011; 85.3% vs. 82.4%, P=0.007, respectively).
Conclusion
GNAS mutations in GH-secreting pituitary tumors are associated with higher preoperative insulin-like growth factor-1 levels and surgical remission rates and lower immediate postoperative nadir GH levels. Thus, GNAS mutation status can predict surgical responsiveness in patients with acromegaly. -
Citations
Citations to this article as recorded by- Genetic diagnosis in acromegaly and gigantism: From research to clinical practice
Claudia Ramírez-Rentería, Laura C. Hernández-Ramírez
Best Practice & Research Clinical Endocrinology & Metabolism.2024; : 101892. CrossRef - CD8/PD-L1 immunohistochemical reactivity and gene alterations in cutaneous squamous cell carcinoma
Haruto Nishida, Yoshihiko Kondo, Takahiro Kusaba, Kazuhiro Kawamura, Yuzo Oyama, Tsutomu Daa, Avaniyapuram Kannan Murugan
PLOS ONE.2023; 18(2): e0281647. CrossRef - Dynamic monitoring of circulating tumor DNA to analyze genetic characteristics and resistance profile of lorlatinib in ALK positive previously treated NSCLC
Xiya Ma, Kun Zhang, Jing Xu, Hongjun Gao, Shaoxing Yang, Haifeng Qin, Hong Wang, Fang Gao, Xiaoqing Liu
Thoracic Cancer.2023; 14(20): 1980. CrossRef - Multiomics Approach to Acromegaly: Unveiling Translational Insights for Precision Medicine
Kyungwon Kim, Cheol Ryong Ku, Eun Jig Lee
Endocrinology and Metabolism.2023; 38(5): 463. CrossRef - Hotspots of Somatic Genetic Variation in Pituitary Neuroendocrine Tumors
Mariana Torres-Morán, Alexa L. Franco-Álvarez, Rosa G. Rebollar-Vega, Laura C. Hernández-Ramírez
Cancers.2023; 15(23): 5685. CrossRef
- Genetic diagnosis in acromegaly and gigantism: From research to clinical practice

- Hypothalamus and Pituitary gland
- Reference Ranges of Serum Insulin-Like Growth Factor-I and Insulin-Like Growth Factor Binding Protein-3: Results from a Multicenter Study in Healthy Korean Adults
- In-Kyung Jeong, Jong Kyu Byun, Junghyun Noh, Sang Wan Kim, Yoon-Sok Chung, Tae Sun Park, Sung-Woon Kim
- Endocrinol Metab. 2020;35(4):954-959. Published online December 1, 2020
- DOI: https://doi.org/10.3803/EnM.2020.785
- 4,352 View
- 124 Download
- 2 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
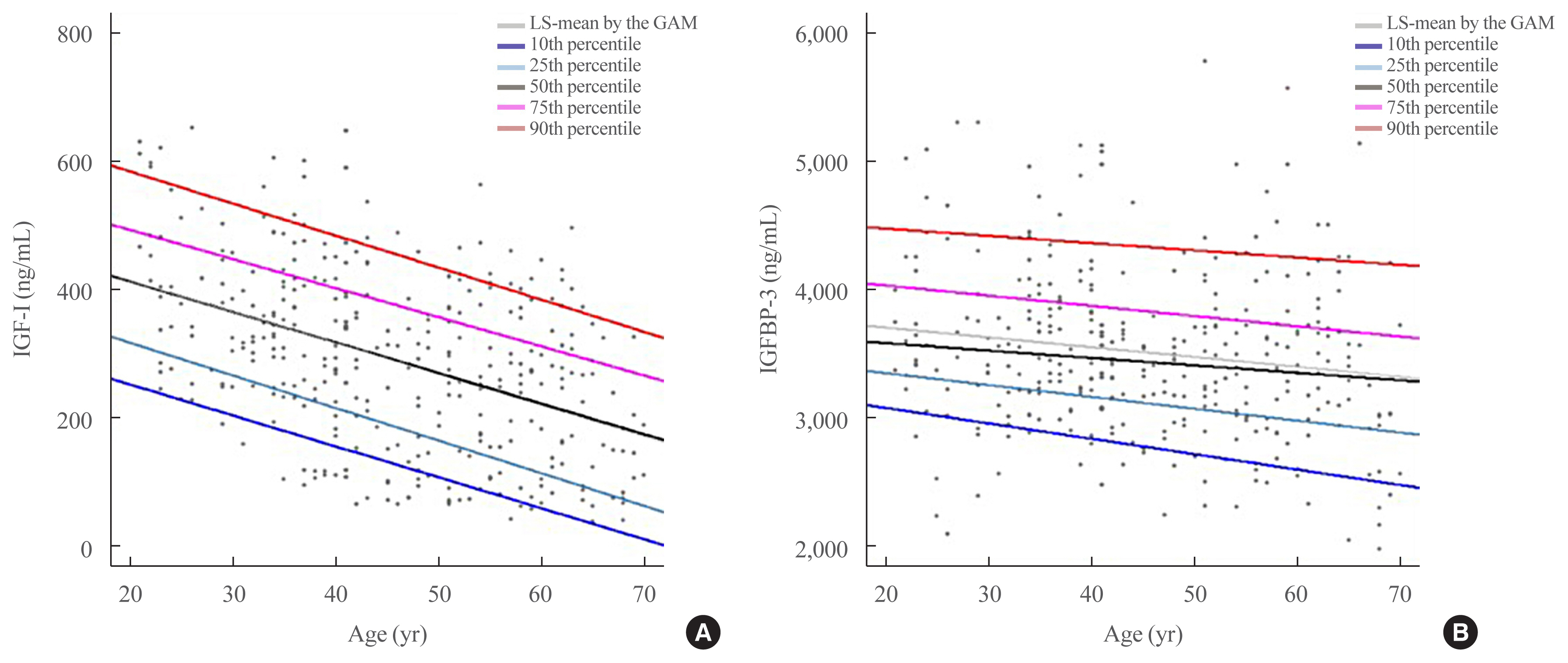
ePub - Insulin-like growth factor-I (IGF-I) plays a pivotal role in the diagnosis and treatment of growth hormone (GH) excess or deficiency. The GH study group of the Korean Endocrine Society aims to establish the Korean reference ranges of serum IGF-I and insulin-like growth factor binding protein-3 (IGFBP-3) and assess the relationship between IGF-I and IGFBP-3 and clinical parameters. Fasting serum was collected from healthy Korean adults at health promotion centers of five hospitals nationwide. Serum IGF-I and IGFBP-3 were measured via an immunoradiometric assay using a DSL kit (Diagnostic Systems Laboratories). Serum samples from 354 subjects (180 male, 174 female) were analyzed based on sex at 10-year intervals from 21 to 70 years. IGF-I levels were inversely correlated with age. After adjustment of age, the IGF-I/IGFBP-3 ratio was significantly negatively associated with blood pressure and free thyroxine and positively associated with weight, hemoglobin, creatinine, alanine transferase, fasting glucose, and thyroid stimulating hormone. Therefore, age- and sex-specific reference ranges of serum IGF-I and IGFBP-3 can be efficient in evaluating GH excess or deficiency in Korean population.
-
Citations
Citations to this article as recorded by- Capillary blood as a complementary matrix for doping control purposes. Application to the definition of the individual longitudinal profile of IGF-1
Carlotta Stacchini, Francesco Botrè, Xavier de la Torre, Monica Mazzarino
Journal of Pharmaceutical and Biomedical Analysis.2023; 227: 115274. CrossRef
- Capillary blood as a complementary matrix for doping control purposes. Application to the definition of the individual longitudinal profile of IGF-1

- Miscellaneous
- Diagnosis and Treatment of Growth Hormone Deficiency: A Position Statement from Korean Endocrine Society and Korean Society of Pediatric Endocrinology
- Jung Hee Kim, Hyun Wook Chae, Sang Ouk Chin, Cheol Ryong Ku, Kyeong Hye Park, Dong Jun Lim, Kwang Joon Kim, Jung Soo Lim, Gyuri Kim, Yun Mi Choi, Seong Hee Ahn, Min Ji Jeon, Yul Hwangbo, Ju Hee Lee, Bu Kyung Kim, Yong Jun Choi, Kyung Ae Lee, Seong-Su Moon, Hwa Young Ahn, Hoon Sung Choi, Sang Mo Hong, Dong Yeob Shin, Ji A Seo, Se Hwa Kim, Seungjoon Oh, Sung Hoon Yu, Byung Joon Kim, Choong Ho Shin, Sung-Woon Kim, Chong Hwa Kim, Eun Jig Lee
- Endocrinol Metab. 2020;35(2):272-287. Published online June 24, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.2.272
- 9,352 View
- 425 Download
- 13 Web of Science
- 15 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
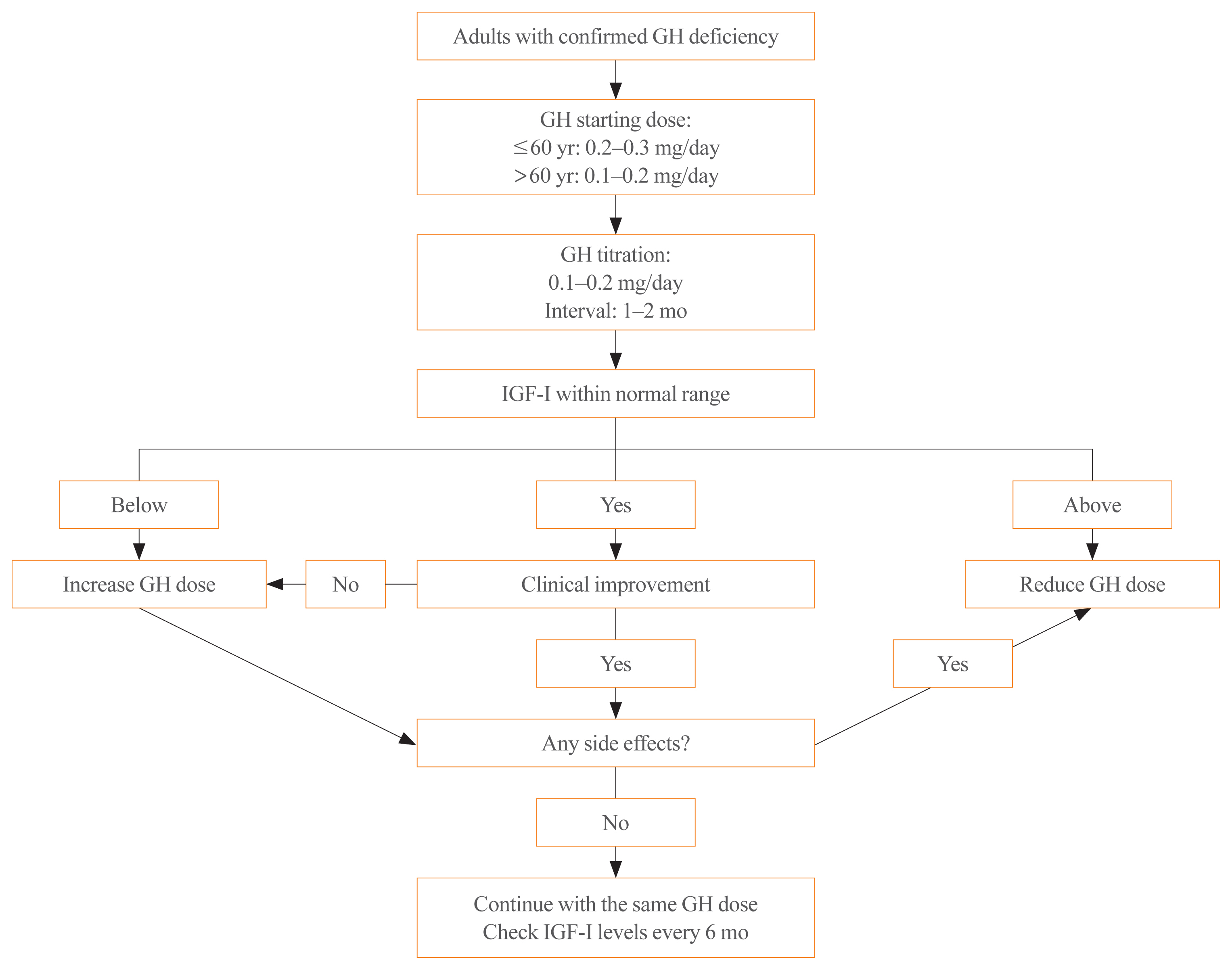
ePub - Growth hormone (GH) deficiency is caused by congenital or acquired causes and occurs in childhood or adulthood. GH replacement therapy brings benefits to body composition, exercise capacity, skeletal health, cardiovascular outcomes, and quality of life. Before initiating GH replacement, GH deficiency should be confirmed through proper stimulation tests, and in cases with proven genetic causes or structural lesions, repeated GH stimulation testing is not necessary. The dosing regimen of GH replacement therapy should be individualized, with the goal of minimizing side effects and maximizing clinical improvements. The Korean Endocrine Society and the Korean Society of Pediatric Endocrinology have developed a position statement on the diagnosis and treatment of GH deficiency. This position statement is based on a systematic review of evidence and expert opinions.
-
Citations
Citations to this article as recorded by- Once-Weekly Somapacitan as an Alternative Management of Growth Hormone Deficiency in Prepubertal Children: A Systematic Review and Meta-Analysis of Randomized Controlled Trial
Ghina Tsurayya, Cut Alifiya Nazhifah, Muhammad Rahmat Pirwanja, Putri Oktaviani Zulfa, Muhammad Raihan Ramadhan Tatroman, Fajar Fakri, Muhammad Iqhrammullah
Children.2024; 11(2): 227. CrossRef - Evaluation of Adult Height in Patients with Non-Permanent Idiopathic GH Deficiency
Agnese Murianni, Anna Lussu, Chiara Guzzetti, Anastasia Ibba, Letizia Casula, Mariacarolina Salerno, Marco Cappa, Sandro Loche
Endocrines.2023; 4(1): 169. CrossRef - The effect of hypothalamic involvement and growth hormone treatment on cardiovascular risk factors during the transition period in patients with childhood-onset craniopharyngioma
Sang Hee Park, Yun Jeong Lee, Jung-Eun Cheon, Choong Ho Shin, Hae Woon Jung, Young Ah Lee
Annals of Pediatric Endocrinology & Metabolism.2023; 28(2): 107. CrossRef - Continuous Glucose Monitoring: A Possible Aid for Detecting Hypoglycemic Events during Insulin Tolerance Tests
Soo Yeun Sim, Moon Bae Ahn
Sensors.2023; 23(15): 6892. CrossRef - The risk patients with AGHD have of developing CVD
Eisha Javed, Maha Zehra, Naz Elahi
International Journal of Cardiology Cardiovascular Risk and Prevention.2023; 19: 200221. CrossRef - Diagnosis of GH Deficiency Without GH Stimulation Tests
Anastasia Ibba, Sandro Loche
Frontiers in Endocrinology.2022;[Epub] CrossRef - Metabolic Impacts of Discontinuation and Resumption of Recombinant Human Growth Hormone Treatment during the Transition Period in Patients with Childhood-Onset Growth Hormone Deficiency
Yun Jeong Lee, Yunha Choi, Han-Wook Yoo, Young Ah Lee, Choong Ho Shin, Han Saem Choi, Ho-Seong Kim, Jae Hyun Kim, Jung Eun Moon, Cheol Woo Ko, Moon Bae Ahn, Byung-Kyu Suh, Jin-Ho Choi
Endocrinology and Metabolism.2022; 37(2): 359. CrossRef - A Radiomics-Based Model with the Potential to Differentiate Growth Hormone Deficiency and Idiopathic Short Stature on Sella MRI
Taeyoun Lee, Kyungchul Song, Beomseok Sohn, Jihwan Eom, Sung Soo Ahn, Ho-Seong Kim, Seung-Koo Lee
Yonsei Medical Journal.2022; 63(9): 856. CrossRef - Phenotypic spectrum of patients with mutations in CHD7: clinical implications of endocrinological findings
Ja Hye Kim, Yunha Choi, Soojin Hwang, Gu-Hwan Kim, Han-Wook Yoo, Jin-Ho Choi
Endocrine Connections.2022;[Epub] CrossRef - Immune Checkpoint Inhibitors and Endocrine Disorders: A Position Statement from the Korean Endocrine Society
Hyemi Kwon, Eun Roh, Chang Ho Ahn, Hee Kyung Kim, Cheol Ryong Ku, Kyong Yeun Jung, Ju Hee Lee, Eun Heui Kim, Sunghwan Suh, Sangmo Hong, Jeonghoon Ha, Jun Sung Moon, Jin Hwa Kim, Mi-kyung Kim
Endocrinology and Metabolism.2022; 37(6): 839. CrossRef - Laron syndrome: clinic, diagnostics (а clinical case)
P.M. Lіashuk, R.P. Lіashuk, N.I. Stankova, M.B. Kudina
INTERNATIONAL JOURNAL OF ENDOCRINOLOGY (Ukraine).2022; 18(3): 193. CrossRef - Diagnosis for Pheochromocytoma and Paraganglioma: A Joint Position Statement of the Korean Pheochromocytoma and Paraganglioma Task Force
Eu Jeong Ku, Kyoung Jin Kim, Jung Hee Kim, Mi Kyung Kim, Chang Ho Ahn, Kyung Ae Lee, Seung Hun Lee, You-Bin Lee, Kyeong Hye Park, Yun Mi Choi, Namki Hong, A Ram Hong, Sang-Wook Kang, Byung Kwan Park, Moon-Woo Seong, Myungshin Kim, Kyeong Cheon Jung, Chan
Endocrinology and Metabolism.2021; 36(2): 322. CrossRef - Asian Conference on Tumor Ablation Guidelines for Adrenal Tumor Ablation
Byung Kwan Park, Masashi Fujimori, Shu-Huei Shen, Uei Pua
Endocrinology and Metabolism.2021; 36(3): 553. CrossRef - Asian Conference on Tumor Ablation guidelines for renal cell carcinoma
Byung Kwan Park, Shu-Huei Shen, Masashi Fujimori, Yi Wang
Investigative and Clinical Urology.2021; 62(4): 378. CrossRef - Diagnosis and Treatment of Adult Growth Hormone Deficiency
Jung Hee Kim
The Korean Journal of Medicine.2021; 96(5): 400. CrossRef
- Once-Weekly Somapacitan as an Alternative Management of Growth Hormone Deficiency in Prepubertal Children: A Systematic Review and Meta-Analysis of Randomized Controlled Trial

- Hypothalamus and Pituitary gland
- Precision Therapy in Acromegaly Caused by Pituitary Tumors: How Close Is It to Reality?
- Cheol Ryong Ku, Vladimir Melnikov, Zhaoyun Zhang, Eun Jig Lee
- Endocrinol Metab. 2020;35(2):206-216. Published online June 24, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.2.206
- 6,648 View
- 250 Download
- 8 Web of Science
- 8 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
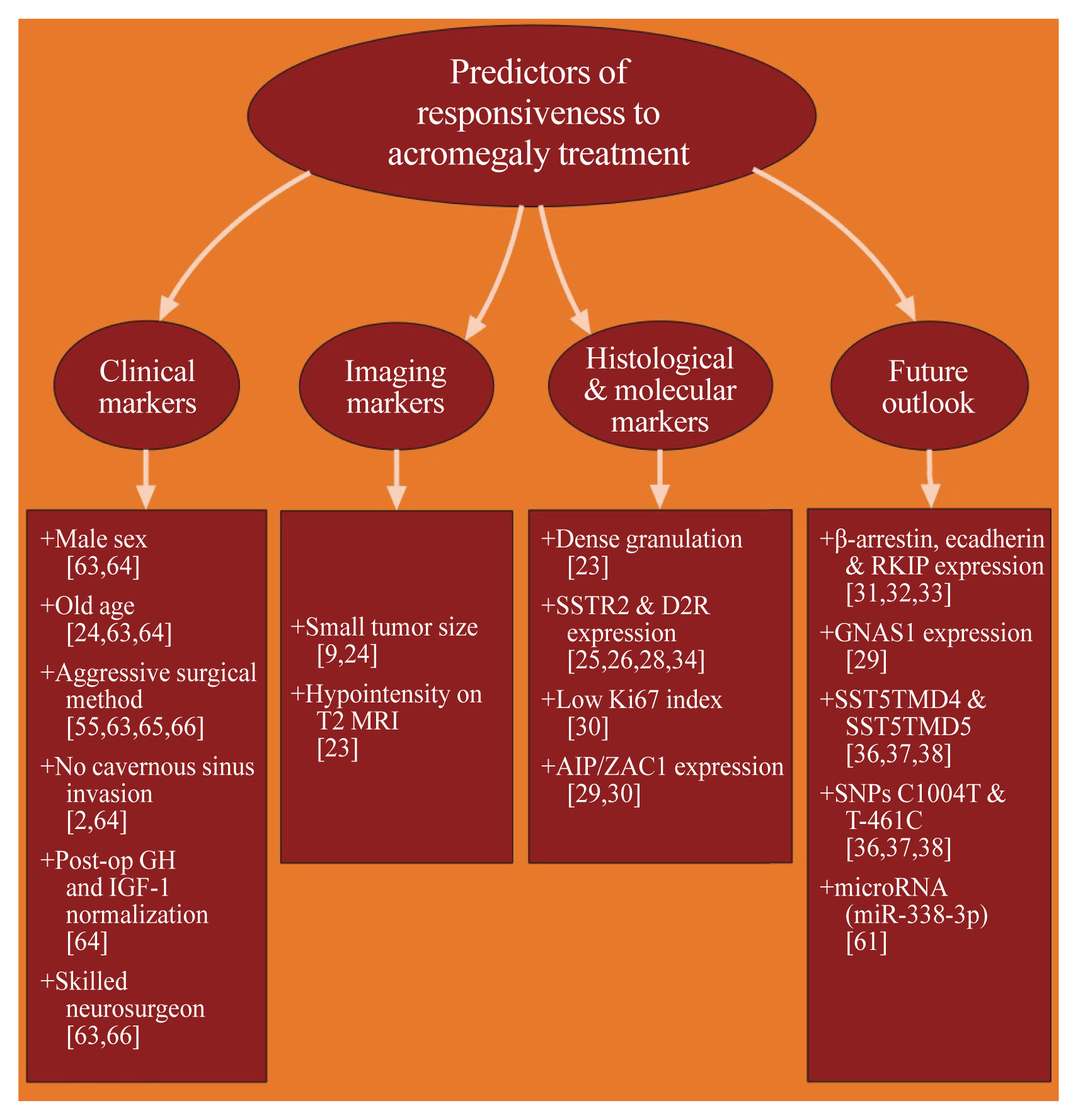
ePub - Acromegaly presents with an enigmatic range of symptoms and comorbidities caused by chronic and progressive growth hormone elevations, commonly due to endocrinologic hypersecretion from a pituitary gland tumor. Comprehensive national acromegaly databases have been appearing over the years, allowing for international comparisons of data, although still presenting varying prevalence and incidence rates. Lack of large-scale analysis in geographical and ethnic differences in clinical presentation and management requires further research. Assessment of current and novel predictors of responsiveness to distinct therapy can lead to multilevel categorization of patients, allowing integration into new clinical guidelines and reduction of increased morbidity and mortality associated with acromegaly. This review compares current data from epidemiological studies and assesses the present-day application of prognostic factors in medical practice, the reality of precision therapy, as well as its future prospects in acromegaly, with a special focus on its relevance to the South Korean population.
-
Citations
Citations to this article as recorded by- Biomarkers of response to treatment in acromegaly
Leandro Kasuki, Elisa Lamback, Ximene Antunes, Mônica R. Gadelha
Expert Review of Endocrinology & Metabolism.2024; 19(1): 71. CrossRef - Multiomics Approach to Acromegaly: Unveiling Translational Insights for Precision Medicine
Kyungwon Kim, Cheol Ryong Ku, Eun Jig Lee
Endocrinology and Metabolism.2023; 38(5): 463. CrossRef - Risk of depression in patients with acromegaly in Korea (2006-2016): a nationwide population-based study
Shinje Moon, Sangmo Hong, Kyungdo Han, Cheol-Young Park
European Journal of Endocrinology.2023; 189(3): 363. CrossRef - The Future of Somatostatin Receptor Ligands in Acromegaly
Monica R Gadelha, Luiz Eduardo Wildemberg, Leandro Kasuki
The Journal of Clinical Endocrinology & Metabolism.2022; 107(2): 297. CrossRef - Innovative therapeutics in acromegaly
Leandro Kasuki, Mônica R. Gadelha
Best Practice & Research Clinical Endocrinology & Metabolism.2022; 36(6): 101679. CrossRef - Risk of Neurodegenerative Diseases in Patients With Acromegaly
Sangmo Hong, Kyungdo Han, Kyung-Soo Kim, Cheol-Young Park
Neurology.2022;[Epub] CrossRef - Machine Learning-based Prediction Model for Treatment of Acromegaly With First-generation Somatostatin Receptor Ligands
Luiz Eduardo Wildemberg, Aline Helen da Silva Camacho, Renan Lyra Miranda, Paula C L Elias, Nina R de Castro Musolino, Debora Nazato, Raquel Jallad, Martha K P Huayllas, Jose Italo S Mota, Tobias Almeida, Evandro Portes, Antonio Ribeiro-Oliveira, Lucio Vi
The Journal of Clinical Endocrinology & Metabolism.2021; 106(7): 2047. CrossRef - Skin anomalies in acromegalic patients (Review of the practical aspects)
Florica Sandru, Adelina Popa, Dan Paduraru, Alexandru Filipescu, Mara Carsote, Adina Ghemigian
Experimental and Therapeutic Medicine.2021;[Epub] CrossRef
- Biomarkers of response to treatment in acromegaly

- Endocrine Research
- Therapeutic Effect of a Novel Chimeric Molecule Targeting Both Somatostatin and Dopamine Receptors on Growth Hormone-Secreting Pituitary Adenomas
- Jean Kim, Ju Hun Oh, Heather Harlem, Michael D. Culler, Cheol Ryong Ku, Eun Jig Lee
- Endocrinol Metab. 2020;35(1):177-187. Published online March 19, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.1.177
- 4,423 View
- 104 Download
- 4 Web of Science
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
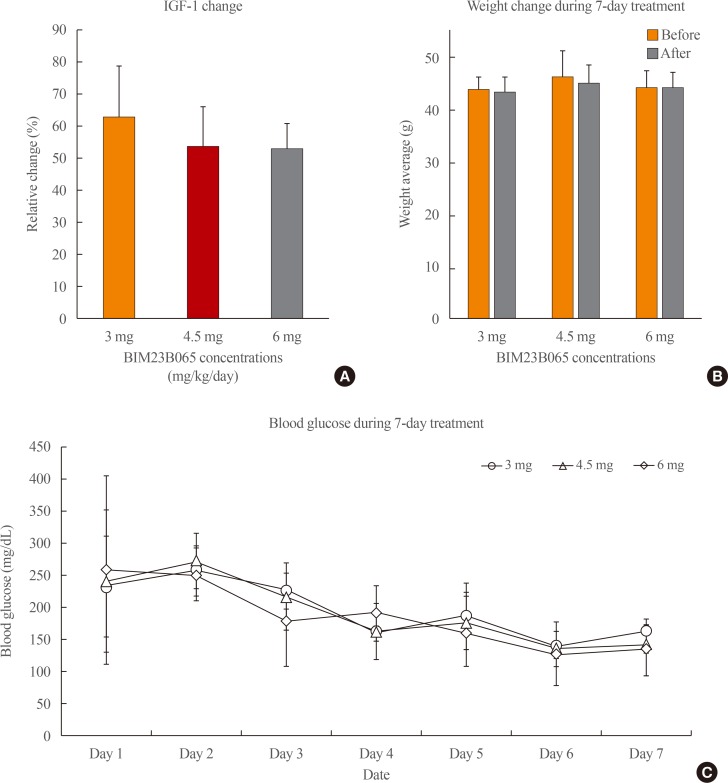
ePub Background Acromegaly is a rare disease primarily caused by growth hormone (GH)-secreting pituitary adenomas, and its treatment is costly. Moreover, some patients are unresponsive to treatment. Hence, there are increasing efforts to develop new drugs with improved effectiveness for this disease. BIM23B065 is a novel chimeric molecule that acts on both somatostatin and dopamine receptors. This study aimed to investigate the effects of BIM23B065 compared with those of a somatostatin receptor analog and a dopamine agonist.
Methods The effects of BIM23B065 on the proliferation, GH and insulin-like growth factor-1 (IGF-1) levels, and extracellular signal-regulated kinase (ERK) 1/2 and cyclic AMP response element binding (CREB) phosphorylation of GH3 cells were investigated with MTS assay, enzyme-linked immunosorbent assay, and Western blotting, respectively. The dosage and treatment duration of BIM23B065 were tested in animal models of GH-secreting pituitary adenoma. The effect of BIM23B065 (3 mg/kg/day) on changes in IGF-1 levels before and after treatment was further investigated.
Results In vitro , BIM23B065 treatment decreased GH release in the culture media and downregulated ERK 1/2 and CREB phosphorylation to 22% and 26%, respectively.In vivo , IGF-1 expression decreased to 50 % after 4 weeks of treatment with BIM23B065 using an osmotic pump implant. Moreover, magnetic resonance imaging results showed that the tumor size decreased significantly following treatment with BIM23B065 for 4 weeks.Conclusion The novel chimeric molecule was effective in decreasing IGF-1 and GH levels and may serve as an effective therapeutic agent for acromegaly.
-
Citations
Citations to this article as recorded by- Pituitary Tumorigenesis—Implications for Management
Rodanthi Vamvoukaki, Maria Chrysoulaki, Grigoria Betsi, Paraskevi Xekouki
Medicina.2023; 59(4): 812. CrossRef - Current and Emerging Medical Therapies in Pituitary Tumors
Nicolas Sahakian, Frédéric Castinetti, Thierry Brue, Thomas Cuny
Journal of Clinical Medicine.2022; 11(4): 955. CrossRef - Characterization of the ability of a, second-generation SST-DA chimeric molecule, TBR-065, to suppress GH secretion from human GH-secreting adenoma cells
Thomas Cuny, Thomas Graillon, Célines Defilles, Rakesh Datta, Shengwen Zhang, Dominique Figarella-Branger, Henry Dufour, Grégory Mougel, Thierry Brue, Tanya Landsman, Heather A. Halem, Michael D. Culler, Anne Barlier, Alexandru Saveanu
Pituitary.2021; 24(3): 351. CrossRef - Efficacy of a Novel Second-Generation Somatostatin-Dopamine Chimera (TBR-065) in Human Medullary Thyroid Cancer: A Preclinical Study
Alessandra Dicitore, Maria Celeste Cantone, Germano Gaudenzi, Davide Saronni, Silvia Carra, Maria Orietta Borghi, Manuela Albertelli, Diego Ferone, Leo J. Hofland, Luca Persani, Giovanni Vitale
Neuroendocrinology.2021; 111(10): 937. CrossRef - Emerging drugs for the treatment of acromegaly
Claudia Campana, Giuliana Corica, Federica Nista, Francesco Cocchiara, Giulia Graziani, Keyvan Khorrami, Marta Franco, Mara Boschetti, Diego Ferone, Federico Gatto
Expert Opinion on Emerging Drugs.2020; 25(4): 409. CrossRef
- Pituitary Tumorigenesis—Implications for Management

- Clinical Effects of E. coli Derived Recombinant Human Growth Hormone (DA - 3001) in Patients with Growth Hormone Deficiency.
- Sei Won Yang, Jeh Hoon Shin, Duk Hi Kim, Byung Churl Lee, Hyung Ro Moon
- J Korean Endocr Soc. 1995;10(4):377-385. Published online November 7, 2019
- 1,282 View
- 31 Download
-
 Abstract
Abstract
 PDF
PDF - Recently, methionyl-hGH was produced in the E. coil K-12, W3110 by recombinant DNA technology in Korea. In this paper, the clinical efficacy and immunogenicity of this GH were studied in 43 patients with growth hormone deficency.The subjects of this study were aged 4.3-18.5 years and each patient received GH 0.5-0.71U/kg week subcutaneously, 6-7 times a week for 1 year. During treatment, height, body weight and bone age were checked. Blood count, urinalysis, blood chemistry and thyroid hormonal concentrations were checked before and every 3 months. The measurement of IGF-1 was performed and assay of antibody against hGH was performed before and every 6 months.The height velocities significantly increased from 3.7+-3.0 cm/year to 11.0+-4.2 cm/year and 9.9+-3.2 cm/year at 6 and 12 months after GH therapy, respectively. The Height SDS were significantly improved after GH therapy with increasing ratio of bone age to chronological age from 0.60+-0.19 at pretreatment to 0.68+-0.16 at 6 month, 0.69+-0.16 at 12 month of therapy. The plasma IGF-1 levels significantly increased during treatment. Three out of 35 patients(8.3%) showed antibody against hGH after 1 year of treatment. Thoughout study, we could not observe any remarkable side effect with GH treatment.These results indicate that this E. coli derived methionyl recombinant growth hormone is effective in improving the index of linear growth in the children with growth hormone deficiency without significant side effect.

- Correlation between Urinary Growth Hormone Level and Peak Serum Growth Hormone Level in Growth Hormone Provocation Test Using Insulin and L - dopa.
- Woo Yeong Chung, Byung Kiu Park, Chul Ho Kim
- J Korean Endocr Soc. 1995;10(4):370-376. Published online November 7, 2019
- 1,005 View
- 21 Download
-
 Abstract
Abstract
 PDF
PDF - To investigate the correlation between urinary growth hormone(GH) level and peak serum GH level, urinary GH value measured by overnight collection of urine for 10 hours and serum GH value in response to GH provocation test using insulin and L-dopa were measured in 9 cases of GH complete deficiency(GCD), 19 cases of GH partial deficiency(GPD) and 40 cases of GH normal short stature(GHN). Urinary GH values were measured by the EIA method using PICOIA HGH plate(Joo Woo Pharmaceutical Co., Japan). Urinary GH was expressed in terms of nanograms per gm creatinine(ng/gCr). Serum GH was measured by immunoradiometric assay using "Daiichi kit"(Je Il Pharmaceutical Co., Japan). Wilcoxon ranked sum test and student's t-test were used to assess the significance of differences between the groups of the patients. The correlation between urinary GH level and peak serum GH level was assessed by the parametric Pearson correlation test. The correlation between peak serum GH level in GH provocation test using insulin and urinary GH level measured by overnight 10 hours collection method showed statistically significant results in all the patients(Y=0.464072X +9.208044, r=0.48987, p=0.0001) and in the GH deficiency groups(GCD+GPD) (Y=0.924659X +9.2385509, r=0.80437, p=0.0001). In case of L-dopa stimulation test, urinary GH values were also positively correlated with peak serum GH level when all the patients were participated(Y=0.572988X +8.312993, r=0.58212, p=0.0001). In contrast, no correlation was found when patients were confined to GH deficiency group(GCD+GPD)(Y=0.127712X +8.3129939, r=0.08044, p=0.6841).

- Changes of Bone Turnover Markers after Treatment with Growth Hormone Therapy in Children with Growth Retardation.
- In Myung Yang, Jeong Taek Woo, Sung Woon Kim, Jin Woo Kim, Young Seol Kim, Young Kil Choi, Ki Oak han, Duk Yoon Kim, Hyung In Yang
- J Korean Endocr Soc. 1994;9(4):344-349. Published online November 6, 2019
- 1,157 View
- 24 Download
-
 Abstract
Abstract
 PDF
PDF - The effects of growth hormone(GH) deficiency and recombinant human GH replacement(0.5IU/kg per week) on bone mineral metabolism in 21GH-deficiency children were studied. All children had significantly reduction of bone density(Z score;-1.4+-0.71). After 1 month of therapy, the levels of serum insulin-like growth factor 1(IGF-1), osteocalcin(OC) and carboxyterminal propeptede of type 1 procollagen(PICP) were significantly elevated. But IGFBP-3 were not shown to change significantly. The changes in serum levels of PICP during the first month of recombinant human GH treatment were positively related to growth velocity, whereas the changes in IGF-1 and OC during the first month of therapy were not. We conclude that the recombinant human GH treatment caused significant modifications of mineral metablism and that the measurement of the changes of biochemical markers of bone metablism espacially PICP may be a useful tool in prediction improved growth velocity during long term GH replacement therpy.

- In Situ Hybridization Analysis of Human Growth Hormone and Prolactin Secreting Pitultary Adenomas.
- Jae Wha Jo, Eun Jig Lee, Moon Suk Nam, Kyung Rae Kim, Sung Kil Lim, Hyun Chul Lee, Kap Bum Huh, Tae Seung Kim, Sun Ho Kim, Joong Uhn Choi, Kyu Chang Lee, Hyun Joo Jung, Sang Seop Chung
- J Korean Endocr Soc. 1994;9(2):82-92. Published online November 6, 2019
- 1,245 View
- 29 Download
-
 Abstract
Abstract
 PDF
PDF - A non-isotopic in situ hybridization method with biotin-labelled oligonucleotide probes was used to examine growth hormone(GH) and prolactin(PRL) gene expression in 32 patients with pituitary adenomas; 13 were prolactinomas, 8 GH secreting adenomas, and 11 mixed GH and PRL secreting adenomas.Positive immunostaining for GH was found in all patients with GH secreting adenomas, and mixed GH and PRL secreting adenomas. Positive immunostaining for PRL was found in all patients with prolactinomas and 9(81.8%) of 11 mixed GH and PRL secreting adenomas, 5(62.5%) of 8 GH secreting adenomas. Immunohistochemistry revealed that 13 were lactotrope adenomas, 5 somatotrope adenomas, and 14 GH and PRL cell adenomas.In situ hybridization revealed that GH mRNA expression was found in all the patients with somatotrope adenomas and GH and PRL cell adenomas, and 6(46.1%) of 13 lactotrope adenomas. PRL mRNA expression was 100% in lactotrope and GH and PRL cell adenomas, and 4(80.0%) of 5 somatotrope adenomas.The patients with a clinical diagnosis of acromegaly had detectable PRL mRNA in their neoplasm and it is suggested that the PRL cells in the adenomas did not result from dedifferentiation, but from the neoplastic stimulus for some mixed tumors probably occurred in cells previously committed to produce PRL and GH. In lactotrope adenomas, the PRL cells of the patients without expression of GH mRNA may be arised from cells programmed to secrete PRL or precussor PRL cells rather than from mixed GH-PRL cells. The finding that some patients produced mRNA detectable by in situ hybridization, but no hormone detectable by immunohistochemistry within tumor was suggested of a silent adenoma.These observations indicated that in situ hybridization studies may improve the classification of pituitary adenomas and may provide a precise knowledge of the biology of these neoplasms.


 KES
KES

 First
First Prev
Prev



