Sex Factors in the Metabolic Syndrome as a Predictor of Cardiovascular Disease
Article information
Abstract
Background
Metabolic syndrome (MetS) is a condition characterized by a cluster of metabolic disorders and is associated with increased risk of cardiovascular disease (CVD). This study analyzed data from the Korean Health and Genome Study to examine the impact of MetS on CVD.
Methods
A total of 8,898 subjects (4,241 males and 4,657 females), 40 to 69 years of age, were enrolled and evaluated for the development of new onset CVD from 2001 to 2012 (median 8.1 years of follow-up).
Results
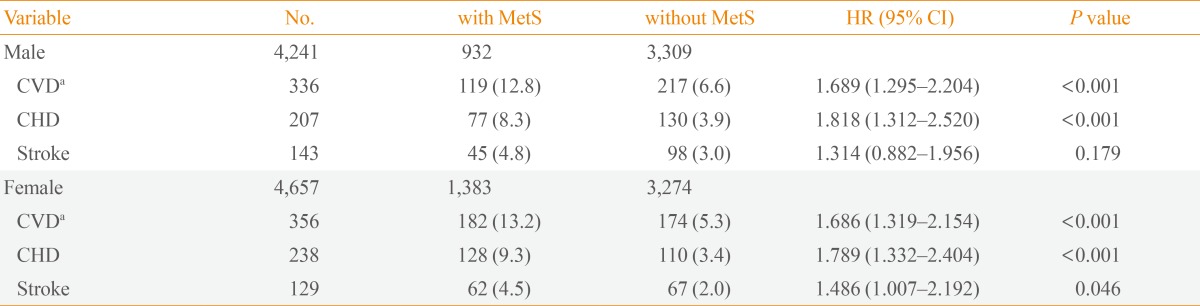
The prevalence of MetS at baseline was 22.0% (932/4,241) and 29.7% (1,383/4,657) in males and females, respectively. MetS was associated with increased risk of coronary heart disease (CHD; hazard ratio [HR], 1.818; 95% confidence interval [CI], 1.312 to 2.520 in males; HR, 1.789; 95% CI, 1.332 to 2.404 in females) and CVD (HR, 1.689; 95% CI, 1.295 to 2.204 in males; HR, 1.686; 95% CI, 1.007 to 2.192 in females). Specifically, MetS was associated with risk of future stroke in females only (HR, 1.486; 95% CI, 1.007 to 2.192). Among MetS components, abdominal obesity and hypertension were independent predictors of both CHD and CVD. In addition, a higher number of MetS components correlated with higher CVD risk.
Conclusion
MetS is a significant risk factor for the development of CVD although its impact varies between sexes.
INTRODUCTION
Metabolic syndrome (MetS) is a condition characterized by a cluster of metabolic disorders including abdominal obesity, insulin resistance/glucose intolerance, dyslipidemia and hypertension. The concept of MetS was introduced by Reaven [1] in 1988, as a clustering of cardiovascular risks, and MetS prevalence is increasing worldwide. The significance of MetS has unraveled gradually in recent years. According to data from the National Health and Nutrition Examination Survey (NHANES) III and NHANESs 1999 to 2006, the age-adjusted prevalence of MetS increased from 29.2% to 34.2% in the United States [2], and a similar trend has been observed in Asian countries [3]. The age-adjusted prevalence of MetS in Korea increased rapidly from 24.9% in 1998 to 31.3% in 2007 [4]. MetS also increases the risk of developing diabetes mellitus (DM) because its components represent major risk factors for impaired glucose metabolism [5]. Accordingly, the prevalence of DM and obesity in Korea has steadily increased [6,7] becoming one of the main targets of public health intervention as well as a social and economic problem in the near future. It is well documented that MetS is associated with increased risk of all-cause mortality and cardiovascular disease (CVD) [8,9,10]. However, the role of MetS in the development of CVD in Korea has only been shown in studies with small sample sizes or cross-sectional designs [11,12,13]. Therefore, this study analyzed data from the largest-scale Korean community based prospective cohort study to examine the impact of MetS on CVD over a 10-year follow-up period.
METHODS
Study population
The design and baseline characteristics of the Ansung-Ansan cohort study have been published previously [14]. Briefly, it is an ongoing prospective, community-based cohort study that is part of the Korean Health and Genome Study (KHGS), a community-based epidemiological survey to investigate trends in DM and associated risk factors. The baseline examination was performed in 2001 to 2002, and biennial follow-up examinations were continued through 2012. To be eligible, subjects must be 40 to 69 years old, have resided within the borders of the survey area for at least 6 months before testing, and be mentally and physically able to participate. Participants were recruited from two Korean communities within 60 km of Seoul. Ansung is a representative rural farming community that had a population of 132,906 in 2000 [15]. Ansan is a representative urban community that had a population of 554,998 in 2000 [15]. A total of 10,038 subjects (5,018 from Ansung and 5,020 from Ansan, respectively) were enrolled in KHGS. Throughout the study, the same trained researchers and instruments were used to collect data. Anthropometric parameters and blood pressure were measured by standard methods. Fasting plasma glucose, lipid profiles, insulin and proteinuria were measured in a central laboratory. Social factors (smoking, exercise, and alcohol intake) were assessed by questionnaire. Current smokers were defined as those who smoked at least one cigarette per day for at least the prior year. Excessive alcohol intake was defined as consumption of ≥30 g of alcohol per day. Informed written consent was obtained from all participants. The study protocol was approved by the ethics committee of the Korean Center for Disease Control and the Institutional Review Board of Ajou University School of Medicine.
Definition of the MetS
Subjects were diagnosed with MetS if they met at least three of the following revised National Cholesterol Education Program Adult Treatment Panel III criteria [16]: (1) abdominal obesity; (2) triglycerides 150 mg/dL or greater; (3) high density lipoprotein cholesterol (HDL-C) less than 40 mg/dL in males or less than 50 mg/dL in females; (4) blood pressure 130/85 mm Hg or greater, or current use of antihypertensive medications; or (5) fasting plasma glucose 100 mg/dL or greater, or previously diagnosed type 2 DM or on oral antidiabetic agents or insulin. Abdominal obesity was defined as a waist circumference ≥90 cm for males and ≥85 cm for females, which are the proposed ethnically appropriate abdominal obesity criteria for Korean people by the Korean Society for the Study of Obesity [17].
Determination of incident CVD
Coronary heart disease (CHD) was defined as definite myocardial infarction confirmed by electrocardiogram and/or enzyme changes or any angina diagnosis that required intervention after confirmation of coronary artery stenosis by coronary angiography. Stroke included cerebral infarction, hemorrhagic stroke, transient ischemic attack, and vertebrobasilar insufficiency as demonstrated by diagnostic work-ups, such as, computed tomography, magnetic resonance imaging studies and accompanying neurologic symptoms and/or signs. CVD was defined as the occurrence of CHD and/or stroke. Persons with medical events reported by the patient himself or found during routine follow-up were asked to bring their medical records. Each event was confirmed and documented by the relevant clinical information (death certificates, postmortem reports, medical records, hospital discharge summaries, laboratory results, etc.). Initial data were obtained from 10,038 subjects who participated in KHGS. Among these subjects, 1,140 were excluded for the following reasons: (1) previous history of CVD (n=230); (2) lack of follow-up examinations after baseline examination (n=908); and (3) missing data (n=2). After applying the above exclusion criteria, a total of 8,898 subjects were eligible for the study. Among them, 692 developed CVD events during the 10-year follow-up period (mean follow-up of 96.8 months). The CVD group comprised all those who developed CVD during the follow-up period.
Statistical analyses
Statistical analyses were carried out using PASW version 21.0 (IBM Co., Armonk, NY, USA). Descriptive statistics for continuous data are expressed as the mean±standard deviation if normally distributed. Discrete data were summarized as numbers with percentages. Independent t test and Mann-Whitney U test were used to compare differences in continuous variables between the two groups. Chi-squared tests were used to analyze dichotomous variables between the groups. Age- and multivariate-adjusted hazard ratios (HR) and 95% confidence intervals (CIs) were estimated with the use of the Cox proportional hazards model to examine MetS baseline status and determine whether the variables played a role in the prediction of incident CHD, stroke, or CVD. For all statistical analyses, a two-sided P<0.05 was considered statistically significant.
RESULTS
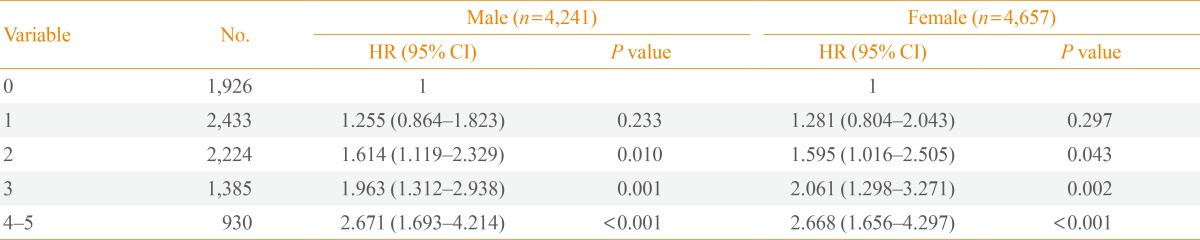
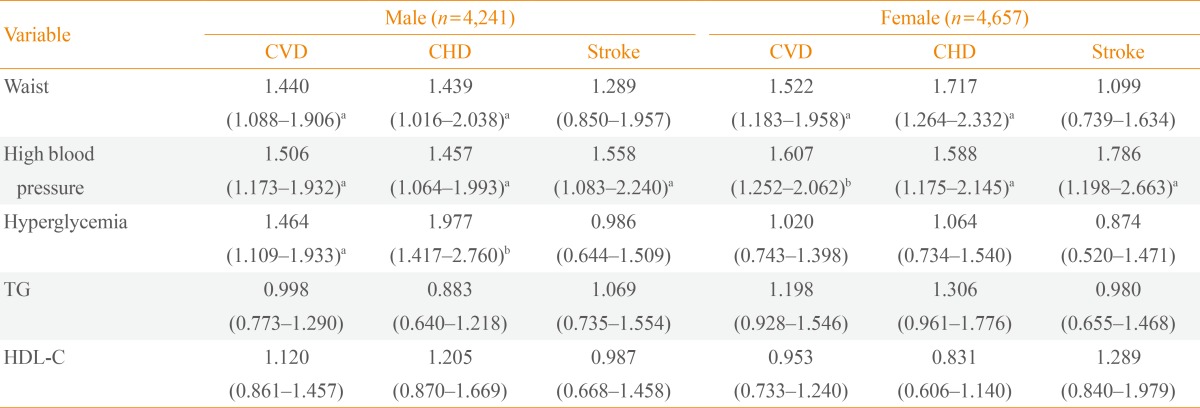
Baseline characteristics of the study subjects are shown in Table 1. Mean follow-up duration was 8.1 years. The overall prevalence of MetS at baseline was 26.0% (22.0% in males and 29.7% in females). Males had significantly higher waist circumference, fasting plasma glucose, diastolic blood pressure, and triglycerides. A greater proportion of males were also current smokers and reported excessive alcohol intake. Compared to males, females were older and had higher body mass index, HDL-C level and homeostasis model of assessment-insulin resistance (HOMA-IR). Females also had a greater number of MetS components (1.8 vs. 1.5) compared to males. MetS was associated with CVD in both males and females despite controlling for other well-documented cardiovascular risk factors. Specifically, MetS was found to be associated with an increased risk of future CHD (HR, 1.818; 95% CI, 1.312 to 2.520; P<0.001) and CVD (HR, 1.689; 95% CI, 1.295 to 2.204; P<0.001) in males. In females, MetS was significantly correlated with the development of CHD (HR, 1.789; 95% CI, 1.332 to 2.404; P<0.001), stroke (HR, 1.486; 95% CI, 1.007 to 2.192; P=0.046), and CVD (HR, 1.686; 95% CI, 1.319 to 2.154; P<0.001) after a mean follow-up of 8.1 years (Table 2). In addition to MetS itself, some individual components of the MetS were also significantly associated with the risk of developing CHD, stroke or CVD. This was independent of known cardiovascular risk factors such as age, current smoking, low density lipoprotein cholesterol (LDL-C), and HOMA-IR (Table 3). Abdominal obesity was an independent predictor of both CHD and CVD in both sexes. Hypertension was a significant risk factor in all outcome parameters including CHD, stroke, and CVD in both males and females. Hyperglycemia was significantly associated with CHD and CVD only in males. However, lipid parameters (i.e., triglycerides and HDL-C) were not significantly correlated with the development of CVD. The HR for developing CVD increased with increasing numbers of MetS components, after adjusting for confounding factors (Table 4).
Individual Metabolic Syndrome Components and the Risk of Cardiovascular Disease during Follow-Up (n=8,898)
DISCUSSION
In the present study, MetS was a significant risk factor for the development of CVD although its impact varies between sexes. More specifically, MetS was correlated with the development of stroke only in females. In addition, the number of MetS components was linearly associated with risk of CVD or CHD.
The overall prevalence rate of CVD in KHGS during the 10-year follow-up period was 7.7% (692/8,898). Overall, MetS prevalence was 26.0% (2,315/8,898) and a higher proportion of females had MetS compared to men, which is similar to previously reported values [4]. A meta-analysis by Gami et al. [18] that included 36 different prospective studies found that the overall relative risk for incident CVD for individuals with MetS was 1.78 (95% CI, 1.58 to 2.00). In this study, the HR for development of CVD was 1.689 (95% CI, 1.295 to 2.204) in males and 1.686 (95% CI, 1.319 to 2.154) in females. This HR is also close to the estimate obtained from Asia Pacific Cohort Study Collaboration, which gathered individual data from 35 cohorts from the Republic of Korea, China, Hong Kong, Japan, Taiwan, Thailand, and Singapore [19]. In the present study, MetS was associated with risk of stroke in females, which has been reported previously [11,20]. Northern Manhattan Study [21] and The Atherosclerosis Risk in Communities data [22] also found that the effect of MetS on stroke risk was greater among women. They suggested that there may be risk differentials according to sex for the MetS due to disparities in the prevalence and potency of vascular risk factors. Sex differences in MetS components (higher proportion of abdominal obesity and lower HDL-C levels in females; data not shown) in this study could explain this phenomenon. Alternative explanations for possible sex differences include a greater impact of the MetS among postmenopausal women. CVD development rate was much higher in females age ≥50 compared to the others (data not shown). However, we do not have data on menopause and hormone use in KHGS. Longitudinal studies based on stroke registries suggested that women are more likely than men to have a stroke and the natural course of stroke is worse in women having a higher probability to be functionally dependent and institutionalized [23]. Recently, meta-analysis found that the excess risk of stroke associated with DM is significantly higher in women than men [24]. These data support the idea that that women's metabolic and vascular risk factor profile has to deteriorate to a greater extent than men to transition from normal to a deranged metabolic state. Moreover, it has been suggested that gender difference in arterial structure and function may lead to gender difference in the relationship of risk factors with stroke [25].
Among the five MetS components, blood pressure and abdominal obesity components were the key predictors of CVD in this study. Hypertension itself is a known cardiovascular risk factor [26,27] and data from 57,237 subjects who came to regular health examinations at a single center in Korea also confirmed this association [28]. Moreover, abdominal obesity is significantly associated with risk of CHD [29] and insulin resistance, which is the underlying etiology of MetS [1]. Abdominal obesity is a major factor in the increased prevalence of MetS in Koreans over the past 10 years [4]. It was previously reported that MetS is associated with abnormal left ventricular structure and function [30,31]. This cardiac dysfunction induced by insulin resistance and MetS could explain the development of CVD [32]. Hyperglycemia was associated with CHD and CVD only in males, although components of hyperglycemia were predictors of CHD and CVD in total subjects including both males and females (data not shown). Lipid parameters did not predict the development of CVD, though previous reports are inconsistent regarding which MetS components predict CVD [10,11,13,28]. These discrepancies may be explained in part by the different study populations and different follow-up periods of each study. Aside from MetS components, age, smoking status, LDL-C, and HOMA-IR are independent risk factors of CVD (data not shown) which are well-known risk factors of CVD [27].
The present study showed that the risk of incident CVD was found to increase with the number of MetS components, and increased by 2.6-fold in those with four or more MetS components compared to those without any MetS components. This synergistic association is noteworthy because some individual components such as hypertriglyceridemia and low HDL-C levels were not significant predictors of CVD. Frequency of CHD, stroke, and CVD were significantly correlated with an increasing number of MetS components (data not shown). This linear synergistic correlation has been reported previously [13,20] and increases the likelihood of CVD to an extent greater than the likelihood conferred by any individual MetS component [33]. This study also found that the risk of CVD increased incrementally as the number of MetS components increased. A similar relationship was found when CVD was sub-classified as CHD or stroke (data not shown). Two or more MetS components were predictive of CVD. This suggests that subjects with two MetS components (especially those with abdominal obesity and hypertension) who do not yet have MetS, should be alert for the development of CVD. Furthermore, it is important to determine ethnicity-specific cutoff points for individual components of MetS based on cardiovascular outcomes.
To the best of the authors' knowledge, this study is the largest prospective cohort study of a Korean population with a long follow-up period that reports the association of MetS with incident CVD. The strengths of this study include its longitudinal population-based study design, long duration of follow-up and sufficient number of CVD events, data analysis according to sex and exclusion of patients with CVD at baseline. One limitation of this study is that MetS diagnosis was based on a single measurement of MetS components at baseline. During follow-up, risk factors may have changed due to medication or lifestyle modifications, and thus MetS diagnosis may have changed during the course of the study. This would weaken the associations found in this study, biasing the results toward the null hypothesis. Therefore, the associations reported here may be stronger than the results indicate.
In summary, MetS was found to be associated with future risk of CHD and CVD in a prospective study with 10 years of follow-up. More specifically, MetS predicted stroke in females and CHD/CVD in both sexes. Risk of incident CVD increased with the number of MetS components. Therefore, individualized lifestyle modifications according to sex and number of MetS components, with or without pharmacological interventions, should be conducted to reduce the burden and negative consequences of MetS.
ACKNOWLEDGMENTS
The authors are grateful to Bong Deok Kim and Hyun Kyu Kim, staff of the Center for Clinical Epidemiology, Ajou University School of Medicine, for their effort in managing the Korean Health and Genome Study. The epidemiological study was supported by the National Genome Research Institute, the Korean Center for Disease Control and Prevention (contract #2001~2003-348-6111-221, 2004-347-6111-213 and 2005-347-2400-2440-215). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Notes
No potential conflict of interest relevant to this article was reported.