Articles
- Page Path
- HOME > Endocrinol Metab > Volume 30(2); 2015 > Article
-
Original ArticleComparison of Serum Adipocytokine Levels according to Metabolic Health and Obesity Status
- Tae Hoon Lee1*, Won Seon Jeon1*, Ki Joong Han1, Shin Yeoung Lee1, Nam Hee Kim1, Hyun Beom Chae1, Choel Min Jang1, Kyung Mo Yoo1, Hae Jung Park1, Min Kyung Lee1, Se Eun Park1, Hyung Geun Oh2, Cheol-Young Park1, Won-Young Lee1, Ki-Won Oh1, Sung-Woo Park1, Eun-Jung Rhee1
-
Endocrinology and Metabolism 2015;30(2):185-194.
DOI: https://doi.org/10.3803/EnM.2015.30.2.185
Published online: June 30, 2015
1Division of Endocrinology and Metabolism, Department of Internal Medicine, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul, Korea.
2Department of Neurology, Soonchunhyang University College of Medicine, Cheonan, Korea.
- Corresponding author: Eun-Jung Rhee. Division of Endocrinology and Metabolism, Department of Internal Medicine, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, 29 Saemunan-ro, Jongno-gu, Seoul 110-746, Korea. Tel: +82-2-2001-2485, Fax: +82-2011-1588, hongsiri@hanmail.net
- *These authors contributed equally to this work.
Copyright © 2015 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Metabolic health is an emerging concept that is highly correlated with various metabolic complications, and adipocytokines have been causally linked to a wide range of metabolic diseases. Thus, this study compared serum adipocytokine levels according to metabolic health and obesity status.
-
Methods
- Four hundred and fifty-six nondiabetic subjects (mean age, 40.5 years) were categorized into four groups according to metabolic health and obesity status: metabolically healthy nonobese (MHNO), metabolically healthy obese (MHO), metabolically unhealthy nonobese (MUHNO), and metabolically unhealthy obese (MUHO). Being metabolically healthy was defined as the presence of fewer than two of the following five metabolic abnormalities: high blood pressure, high fasting blood glucose, high triglyceride, low high density lipoprotein cholesterol, and being in the highest decile of the homeostatic model assessment of insulin resistance index. Obesity status was assessed using body mass index (BMI), with obesity defined as a BMI higher than 25 kg/m2. Levels of serum interleukin-6 (IL-6), monocyte chemoattractant protein-1 (MCP-1), tumor necrosis factor α (TNF-α), and adipocyte fatty acid binding protein (A-FABP) were also evaluated.
-
Results
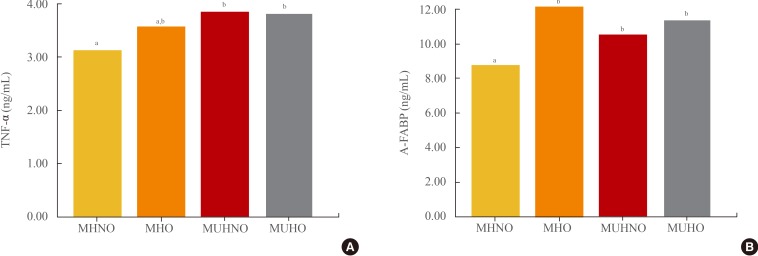
- Of the 456 subjects, 247 (54.2%) were in the MHNO group, 66 (14.5%) were in the MHO group, 66 (14.5%) were in the MUHNO group, and 77 (16.9%) were in the MUHO group. There were no significant differences in IL-6 or MCP-1 levels among the groups, but levels of TNF-α and A-FABP were significantly higher in the MUHNO group compared to the MHNO group.
-
Conclusion
- High TNF-α and A-FABP levels are significantly associated with metabolically unhealthiness in nonobese Korean individuals.
- The prevalence of obesity continues to increase worldwide, and it has been projected that more than 1 billion people will be afflicted with this condition by 2030 [1]. Obesity is a risk factor for comorbid conditions such as type 2 diabetes mellitus (T2DM), hypertension, cardiovascular disease, stroke, and several types of cancer [23], which are all associated with an increased risk for premature death.
- The term metabolically healthy obese (MHO) is now widely used to describe a subtype of obesity that appears to be protected against obesity-related metabolic comorbidities and mortality [456]. Compared to metabolically unhealthy individuals, metabolically healthy individuals possess a favorable metabolic profile that is characterized by a high degree of insulin sensitivity [7], a lesser state of inflammation [8], and favorable lipid and hepatic enzyme profiles [910].
- The discovery of adipocytokines, which are products of adipose tissue, was a turning point in the understanding of metabolic disease. Adipocytokines play an important role in the metabolic homeostasis of healthy subjects, and changes in the levels and activity of these factors in obese subjects contribute to metabolic complications [11]. Accordingly, the measurement of serum adipocytokine levels could be helpful for assessing subclinical metabolic disturbances. Although underlying mechanisms that could explain favorable metabolic profile of MHO individuals are largely unknown, adipocytokines might be key factors of metabolic health in view of the fact that they are significantly associated with metabolic diseases [12].
- Relatively few studies have examined the relationships among adipocytokines, metabolic health, and obesity [913]. Thus, the present study compared serum adipocytokine levels according to metabolic health and obesity status in an Asian population.
INTRODUCTION
- Subjects
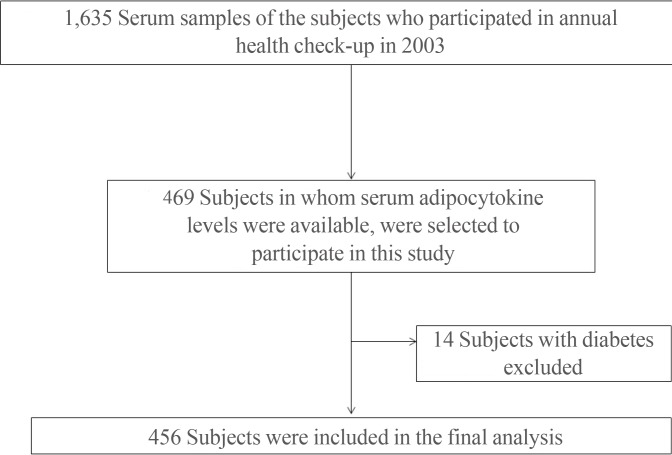
- This study was performed as a substudy in the Kangbuk Samsung Medical Center-adipokine study, which is a longitudinal study performed in subjects who received annual health check-ups at the Health Promotion Center at Kangbuk Samsung Hospital in Seoul, Korea, between 2003 and 2007 [14]. One thousand six hundred and thirty-five serum samples of the subjects who participated in annual health check-up in 2003, 469 subjects in whom serum adipocytokine levels were available, were selected to participate in this study. Among these, 456 subjects (303 men and 153 women) after exclusion of 14 subjects with diabetes were enrolled in final analysis as a cross-sectional study (mean age, 40.5 years) (Fig. 1).
- The study protocol conformed to ethical guidelines of the 1975 Declaration of Helsinki, and accordingly the Kangbuk Samsung Hospital Human Research Committee approved it. The Institutional Review Board of Kangbuk Samsung Hospital also approved this study and no specific informed consent was obtained. The informed consent requirement for this study was exempted by the Institutional Review Board at the time the study was in the planning phase because researchers only accessed the database for analysis purposes, which was free of identifying personal information.
- Anthropometric and laboratory measurements
- Height and weight were measured twice using automated instruments and then averaged. The body mass index (BMI) was calculated as by dividing the weight in kilogram by the square of the height in meters. Blood pressure (BP) was measured using a standardized sphygmomanometer after 5 minutes of rest. The waist circumference was measured in the standing position, at the middle point between anterior iliac crest and lower border of rib by a single examiner.
- Body composition measurements of the subjects were carried out by segmental bioelectric impedance, using eight tractile electrodes according to the manufacturer's instructions (InBody 3.0, Biospace, Seoul, Korea). Lean mass (kg), fat mass (kg), percent fat mass (%), and waist-hip ratio as a marker of abdominal obesity, were measured.
- All of the subjects were examined after an overnight fast. The hexokinase method was used to test fasting glucose concentrations (Hitachi Modular D2400, Roche, Tokyo, Japan). Fasting insulin concentrations were determined by electrochemiluminescence immunoassay (Hitachi Modular E170, Roche). An enzymatic calorimetric test was used to measure the total cholesterol (TC) and triglyceride (TG) concentrations. The selective inhibition method was used to measure the level of high density lipoprotein cholesterol (HDL-C), and a homogeneous enzymatic calorimetric test was used to measure the level of low density lipoprotein cholesterol (LDL-C). Samples for measurement of interleukin-6 (IL-6), monocyte chemoattractant protein-1 (MCP-1), tumor necrosis factor α (TNF-α), and adipocyte fatty acid binding protein (A-FABP) were distributed to tubes and stored at -80℃ prior to measurement of serum levels by an enzyme-linked immunosorbent assay method (BioVendor Laboratory Medicine, Modrice, The Czech Republic). Hemoglobin A1c was measured by immunoturbidimetric assay with a Cobra Integra 800 automatic analyzer (Roche Diagnostics, Basel, Switzerland) with a reference value of 4.4% to 6.4%. The methodology was aligned with the Diabetes Control and Complications Trial and National Glycohemoglobin Standardization Program (NGSP) standards [15]. The intra-assay coefficient of variation (CV) was 2.3% and interassay CV was 2.4%, both within the NGSP acceptable limits [16].
- The subjects with underlying diabetes were excluded from the study. The presence of impaired fasting glucose and diabetes mellitus (DM) was determined according to the self-questionnaire of the participants and the diagnostic criteria of American Diabetes Association [17]. Insulin resistance was measured using the homeostatic model assessment of insulin resistance (HOMA-IR) and was obtained by applying the following formula: HOMA-IR=fasting insulin (IU/mL)×fasting blood glucose (mmol/L)/22.5 [18].
- Definition of metabolic health and obesity status
- Obesity status was defined based on the combined consideration of obesity status by BMI category (nonobese <25 kg/m2, obese ≥25 kg/m2). In 2000, the World Health Organization Western Pacific Region suggested revised Asia-Pacific criteria of obesity in Asian populations using reduced values for BMI ≥25 kg/m2 in both sexes [19].
- Being metabolically healthy was defined by having less than two metabolic abnormalities among the four components of metabolic syndrome besides waist circumference criteria plus insulin resistance status defined by HOMA-IR, which was modified from the criteria by Wildman et al. [2021]:
- (1) Systolic BP ≥130 mm Hg and/or diastolic BP ≥85 mm Hg or on antihypertensive treatment
- (2) TG ≥150 mg/dL
- (3) Fasting glucose ≥100 mg/dL
- (4) HDL-C <40 mg/dL in men, <50 mg/dL in women
- (5) HOMA-IR ≥90th percentile (≥2.28)
- According to the above criteria, participants were divided into four groups:
- (1) Metabolically healthy nonobese (MHNO): BMI <25 kg/m2 and <2 metabolic risk factor
- (2) MHO: BMI ≥25 kg/m2 and <2 metabolic risk factor
- (3) Metabolically unhealthy nonobese (MUHNO): BMI <25 kg/m2 and ≥2 metabolic risk factor
- (4) Metabolically unhealthy obese (MUHO): BMI ≥25 kg/m2 and ≥2 metabolic risk factor
- Statistical analysis
- All data were analyzed using SPSS version 18.0 (SPSS Inc., Chicago, IL, USA). Comparisons of the mean values and the prevalence of metabolic variables among the four groups divided by metabolic health and obesity status were performed with one-way analysis of variance test and chi-square test, and data that do not follow normal distribution were analyzed after logarithmic transformation. Bivariate correlation analysis was performed to evaluate relation between metabolic parameters and adipocytokines. Statistical significance was defined as P<0.05.
METHODS
- The general characteristics of the participants are presented in Table 1. For all participants, the mean age was 40.5 years, and the proportion of males was 66.4% (303/456). Of the 456 subjects, 247 (54.2%) were in the MHNO group, 66 (14.5%) were in the MHO group, 66 (14.5%) were in the MUHNO group, and 77 (16.9%) were in the MUHO group (Table 2).
- A bivariate correlation analysis, which included metabolic variables with adipocytokines, TNF-α and A-FABP showed significant correlation with TC, TG, HDL-C, LDL-C, BMI, BP, and body fat mass (Table 3).
- MUHNO group had a higher proportion of males, higher average BMI and BP; and higher levels of serum fasting glucose and TG; and lower level of serum HDL-C compared with MHNO group. MUHO group had a higher proportion of males, higher BP; and higher levels of serum fasting glucose, TG, and fasting insulin compared with MHO group. Among measured adipocytokines, IL-6 and MCP-1 levels did not differ significantly among four groups. MUHNO group showed significantly higher levels of TNF-α and A-FABP than those of MHNO group (Fig. 2). Meanwhile, there was no significant difference in levels of TNF-α and A-FABP between MUHO and MHO groups.
RESULTS
- The principal finding of this study is that metabolically unhealthy individuals exhibited significantly higher levels of several adipocytokines compared to metabolically healthy individuals in the nonobese groups (MHNO and MUHNO). Although this result was not observed in the obese groups, the present data indicate that there is an association between metabolic health and serum levels of adipocytokines.
- Metabolic health has been highlighted as an important determinant of favorable clinical outcomes in recent literatures [122223]. Previously, our retrospective longitudinal study reported that metabolically unhealthy subjects showed significantly increased risk for diabetes development in a median 48.7 months of follow-up period compared with metabolically healthy subjects, regardless of obesity status assessed by BMI [24]. Moreover, a cross-sectional study conducted by our group that included 24,063 participants in a health screening program found that metabolically unhealthy subjects are more prone to coronary artery calcification (as assessed by coronary artery calcium scores) than metabolic healthy subjects [25].
- Numerous studies have demonstrated that MHO individuals display a greater degree of sensitivity and lower fasting glucose and insulin levels compared to MUHO individuals [78262728]. Furthermore, MHO individuals have higher HDL-C levels but lower TG and hepatic enzyme levels compared to MUHO individuals [78132728]. Additional clinical characteristics of MHO individuals include lower free fatty acid concentrations [726], higher circulating levels of natural killer cells and cytotoxic T lymphocytes [29], and significantly lower levels of high-sensitivity C-reactive protein and alpha-1 antitrypsin [8]. Although the present study did not observe any significant differences between the MHO and MUHO groups in terms of adipocytokine levels, it still provides meaningful data. However, the reasons why only nonobese subjects showed significant differences according to metabolic health in terms of adipocytokines in the present study is a matter of debate. This discrepancy may be explained by the use of BMI to assess obesity rather than waist circumference, which specifically evaluates abdominal obesity. Further studies should be conducted using nonobese groups of individuals with an unhealthy metabolic status to clarify this issue.
- The measurement of circulating adipocytokines could be useful to evaluate metabolic health. There are many studies that suggested the significant association between adipocytokines and metabolic complications such as DM microangiopathy [30], coronary atherosclerosis [3132], nonalcoholic fatty liver disease [33]. However, only a few studies reported about the relationship between adipocytokines and metabolic health and obesity. One cross-sectional analysis in 716 subjects reported that MHO individuals have adiponectin concentrations similar to those found in normal body weight subjects [12]. In this study, MHO was defined as a BMI above 30 kg/m2 plus HDL-C of at least 40 mg/dL in the absence of T2DM and hypertension. Another cross-sectional study evaluated 60 morbidly obese patients and found that serum concentrations of several adipocytokines were quite different between insulin-sensitive and insulin-resistant obese groups [9]. In contrast to the above studies, the present study defined metabolic health according to the presence or absence of various metabolic health-related components, such as, BP, fasting glucose, TG, HDL-C, and HOMA-IR [1920].
- Of the four serum adipocytokines measured, TNF-α and A-FABP showed an association with metabolic health in nonobese individuals. TNF-α was the first proinflammatory cytokine identified in the adipose tissue of obese mice that plays a role as a functional link between inflammation and insulin resistance, marking the start of the metabolic inflammation concept [34]. Mice lacking TNF-α or its receptors maintained insulin sensitivity in both dietary and genetic models of obesity [35]. There is also a strong association between circulating TNF-α and insulin resistance or other metabolic complications in humans [3637]. Although few studies have been performed on TNF-α levels in metabolically unhealthy lean subjects, normal-weight women with polycystic ovarian syndrome, which is known to be caused by insulin resistance, showed significantly increased TNF-α levels compared with control subjects, suggesting increased TNF-α levels in lean, metabolically unhealthy subjects [38]. Also known as FABP4 or aP2, A-FABP is one of nine FABPs with tissue-specific function and patterns of expression [39]. In mouse models, A-FABP deficiency was resulted in reduced adiposity, enhanced insulin sensitivity and reduced hepatosteatosis [40]. Recently, A-FABP is considered as an independent marker of metabolic syndrome and atherosclerosis [414243].
- In the present study, IL-6 and MCP-1 did not differ significantly among the groups. IL-6 is one of major proinflammatory cytokine that induces insulin resistance via an interruption of insulin-signaling pathways [44]. However, there is contradictory evidence from recent studies regarding the influence of IL-6 on insulin action and glucose metabolism [4546]. The release of IL-6 from skeletal muscle may have beneficial effects, including increased glucose uptake and fatty acid oxidation [47], and it appears that the activation of AMP-kinase by IL-6 mediates these effects [48]. The fact that IL-6 is produced by several organs might contribute to its complex effects on metabolic regulation. MCP-1 decreases insulin-stimulated glucose uptake in adipocytes [49], and plays a pivotal role in the development of atherosclerosis via the enhancement of adhesion molecules on leukocytes and endothelial cells [50]. However, recent studies investigating the role of MCP-1 on insulin resistance have produced inconsistent results [5152]. These inconsistent results regarding the association of IL-6 and MCP-1 serum levels with metabolic properties might be in line with the present findings.
- Our study has several limitations. First, we used a cross-sectional study design and therefore could not establish a cause-and-effect relationship between adipocytokines and metabolic health. Second, as most of the participants were only slightly obese Asians (BMI <30 kg/m2), our data might not be applicable to the global population with higher grades of obesity. Third, among 1,635 subjects in whom serum samples were collected, serum adipocytokine levels were available only in 469 subjects. This might act as a confounding factor or a selection bias. Finally, since there is no unified definition of metabolic health, there might be considerable differences in data according to which definition is applied [53]. Despite limitations of the study, this study has significance in that it was the first study to examine the role of adipocytokines in metabolic health and obesity status.
- In conclusion, the present study found that MUHNO subjects showed significantly higher levels of serum TNF-α and A-FABP compared to MHNO subjects. This suggests that adipocytokines may be significant determinants of metabolic health, but further longitudinal studies are needed to assess whether circulating adipocytokines represent a biomarker for metabolic health or whether these factors result in metabolic complications. Furthermore, it is necessary to determine whether the manipulation of adipocytokines could be a future therapy for metabolic abnormalities. If there is a causal relationship between adipocytokines and metabolic health, early lifestyle modifications or pharmacological interventions will likely provide beneficial effects for metabolically high-risk individuals.
DISCUSSION
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
Article information
- 1. Kelly T, Yang W, Chen CS, Reynolds K, He J. Global burden of obesity in 2005 and projections to 2030. Int J Obes (Lond) 2008;32:1431–1437. ArticlePubMedPDF
- 2. Mokdad AH, Ford ES, Bowman BA, Dietz WH, Vinicor F, Bales VS, Marks JS. Prevalence of obesity, diabetes, and obesity-related health risk factors, 2001. JAMA 2003;289:76–79. ArticlePubMedPDF
- 3. Van Gaal LF, Mertens IL, De Block CE. Mechanisms linking obesity with cardiovascular disease. Nature 2006;444:875–880. ArticlePubMedPDF
- 4. Calori G, Lattuada G, Piemonti L, Garancini MP, Ragogna F, Villa M, Mannino S, Crosignani P, Bosi E, Luzi L, Ruotolo G, Perseghin G. Prevalence, metabolic features, and prognosis of metabolically healthy obese Italian individuals: the Cremona Study. Diabetes Care 2011;34:210–215. ArticlePubMed
- 5. Ortega FB, Lee DC, Katzmarzyk PT, Ruiz JR, Sui X, Church TS, Blair SN. The intriguing metabolically healthy but obese phenotype: cardiovascular prognosis and role of fitness. Eur Heart J 2013;34:389–397. ArticlePubMedPDF
- 6. Hamer M, Stamatakis E. Metabolically healthy obesity and risk of all-cause and cardiovascular disease mortality. J Clin Endocrinol Metab 2012;97:2482–2488. ArticlePubMedPMC
- 7. Succurro E, Marini MA, Frontoni S, Hribal ML, Andreozzi F, Lauro R, Perticone F, Sesti G. Insulin secretion in metabolically obese, but normal weight, and in metabolically healthy but obese individuals. Obesity (Silver Spring) 2008;16:1881–1886. ArticlePubMed
- 8. Karelis AD, Faraj M, Bastard JP, St-Pierre DH, Brochu M, Prud'homme D, Rabasa-Lhoret R. The metabolically healthy but obese individual presents a favorable inflammation profile. J Clin Endocrinol Metab 2005;90:4145–4150. ArticlePubMed
- 9. Kloting N, Fasshauer M, Dietrich A, Kovacs P, Schon MR, Kern M, Stumvoll M, Bluher M. Insulin-sensitive obesity. Am J Physiol Endocrinol Metab 2010;299:E506–E515. ArticlePubMed
- 10. Messier V, Karelis AD, Robillard ME, Bellefeuille P, Brochu M, Lavoie JM, Rabasa-Lhoret R. Metabolically healthy but obese individuals: relationship with hepatic enzymes. Metabolism 2010;59:20–24. ArticlePubMed
- 11. Cao H. Adipocytokines in obesity and metabolic disease. J Endocrinol 2014;220:T47–T59. ArticlePubMedPMC
- 12. Bluher M. Are there still healthy obese patients? Curr Opin Endocrinol Diabetes Obes 2012;19:341–346. ArticlePubMed
- 13. Aguilar-Salinas CA, Garcia EG, Robles L, Riano D, Ruiz-Gomez DG, Garcia-Ulloa AC, Melgarejo MA, Zamora M, Guillen-Pineda LE, Mehta R, Canizales-Quinteros S, Tusie Luna MT, Gomez-Perez FJ. High adiponectin concentrations are associated with the metabolically healthy obese phenotype. J Clin Endocrinol Metab 2008;93:4075–4079. ArticlePubMedPDF
- 14. Rhee EJ, Seo MH, Jeon WS, Won HK, Choi JH, Park SE, Park CY, Kwag HJ, Oh KW, Park SW, Kim SW, Lee WY. The association of baseline adipocytokine levels with glycemic progression in nondiabetic Korean adults in 4 years of follow-up. Diabetes Res Clin Pract 2012;98:501–507. ArticlePubMed
- 15. National Glycohemoglobin Standardization Program. List of NGSP certified methods [Internet]; NGSP; c2010. cited 2012 Nov 26. Available from: http://www.ngsp.org/docs/methods.pdf.
- 16. Schwartz KL, Monsur JC, Bartoces MG, West PA, Neale AV. Correlation of same-visit HbA1c test with laboratory-based measurements: a MetroNet study. BMC Fam Pract 2005;6:28ArticlePubMedPMCPDF
- 17. American Diabetes Association. Standards of medical care in diabetes: 2014. Diabetes Care 2014;37(Suppl 1):S14–S80. ArticlePubMed
- 18. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412–419. ArticlePubMedPDF
- 19. World Health Organization Western Pacific Region. The Asia-Pacific perspective: redefining obesity and its treatment; Geneva: WHO; 2000.
- 20. Wildman RP, Muntner P, Reynolds K, McGinn AP, Rajpathak S, Wylie-Rosett J, Sowers MR. The obese without cardiometabolic risk factor clustering and the normal weight with cardiometabolic risk factor clustering: prevalence and correlates of 2 phenotypes among the US population (NHANES 1999-2004). Arch Intern Med 2008;168:1617–1624. ArticlePubMed
- 21. Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, Fruchart JC, James WP, Loria CM, Smith SC Jr. International Diabetes Federation Task Force on Epidemiology and Prevention. Hational Heart, Lung, and Blood Institute. American Heart Association. World Heart Federation. International Atherosclerosis Society. International Association for the Study of Obesity. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009;120:1640–1645. ArticlePubMed
- 22. Primeau V, Coderre L, Karelis AD, Brochu M, Lavoie ME, Messier V, Sladek R, Rabasa-Lhoret R. Characterizing the profile of obese patients who are metabolically healthy. Int J Obes (Lond) 2011;35:971–981. ArticlePubMedPDF
- 23. Appleton SL, Seaborn CJ, Visvanathan R, Hill CL, Gill TK, Taylor AW, Adams RJ. North West Adelaide Health Study Team. Diabetes and cardiovascular disease outcomes in the metabolically healthy obese phenotype: a cohort study. Diabetes Care 2013;36:2388–2394. ArticlePubMedPMC
- 24. Rhee EJ, Lee MK, Kim JD, Jeon WS, Bae JC, Park SE, Park CY, Oh KW, Park SW, Lee WY. Metabolic health is a more important determinant for diabetes development than simple obesity: A 4-year retrospective longitudinal study. PLoS One 2014;9:e98369ArticlePubMedPMC
- 25. Rhee EJ, Seo MH, Kim JD, Jeon WS, Park SE, Park CY, Oh KW, Park SW, Lee WY. Metabolic health is more closely associated with coronary artery calcification than obesity. PLoS One 2013;8:e74564ArticlePubMedPMC
- 26. Stefan N, Kantartzis K, Machann J, Schick F, Thamer C, Rittig K, Balletshofer B, Machicao F, Fritsche A, Haring HU. Identification and characterization of metabolically benign obesity in humans. Arch Intern Med 2008;168:1609–1616. ArticlePubMed
- 27. Brochu M, Tchernof A, Dionne IJ, Sites CK, Eltabbakh GH, Sims EA, Poehlman ET. What are the physical characteristics associated with a normal metabolic profile despite a high level of obesity in postmenopausal women? J Clin Endocrinol Metab 2001;86:1020–1025. ArticlePubMed
- 28. Marini MA, Succurro E, Frontoni S, Hribal ML, Andreozzi F, Lauro R, Perticone F, Sesti G. Metabolically healthy but obese women have an intermediate cardiovascular risk profile between healthy nonobese women and obese insulin-resistant women. Diabetes Care 2007;30:2145–2147. ArticlePubMed
- 29. Lynch LA, O'Connell JM, Kwasnik AK, Cawood TJ, O'Farrelly C, O'Shea DB. Are natural killer cells protecting the metabolically healthy obese patient? Obesity (Silver Spring) 2009;17:601–605. ArticlePubMed
- 30. Jung CH, Kim BY, Mok JO, Kang SK, Kim CH. Association between serum adipocytokine levels and microangiopathies in patients with type 2 diabetes mellitus. J Diabetes Investig 2014;5:333–339.ArticlePubMed
- 31. Shimabukuro M, Hirata Y, Tabata M, Dagvasumberel M, Sato H, Kurobe H, Fukuda D, Soeki T, Kitagawa T, Takanashi S, Sata M. Epicardial adipose tissue volume and adipocytokine imbalance are strongly linked to human coronary atherosclerosis. Arterioscler Thromb Vasc Biol 2013;33:1077–1084. ArticlePubMed
- 32. Lee KK, Suh YS, Yum KS. The effect of visceral fat area and adipocytokines on acute myocardial infarction: a case-control study in adult Korean population. Korean J Obes 2012;21:57–64.ArticlePDF
- 33. Seo YY, Cho YK, Bae JC, Seo MH, Park SE, Rhee EJ, Park CY, Oh KW, Park SW, Lee WY. Tumor necrosis factor-alpha as a predictor for the development of nonalcoholic fatty liver disease: a 4-year follow-up study. Endocrinol Metab (Seoul) 2013;28:41–45. ArticlePubMedPMC
- 34. Hotamisligil GS, Shargill NS, Spiegelman BM. Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance. Science 1993;259:87–91. ArticlePubMed
- 35. Uysal KT, Wiesbrock SM, Marino MW, Hotamisligil GS. Protection from obesity-induced insulin resistance in mice lacking TNF-alpha function. Nature 1997;389:610–614. ArticlePubMedPDF
- 36. Hivert MF, Sullivan LM, Fox CS, Nathan DM, D'Agostino RB Sr, Wilson PW, Meigs JB. Associations of adiponectin, resistin, and tumor necrosis factor-alpha with insulin resistance. J Clin Endocrinol Metab 2008;93:3165–3172. ArticlePubMedPMCPDF
- 37. Berg AH, Scherer PE. Adipose tissue, inflammation, and cardiovascular disease. Circ Res 2005;96:939–949. ArticlePubMed
- 38. Tarkun I, Cetinarslan B, Turemen E, Canturk Z, Biyikli M. Association between circulating tumor necrosis factor-alpha, interleukin-6, and insulin resistance in normal-weight women with polycystic ovary syndrome. Metab Syndr Relat Disord 2006;4:122–128. ArticlePubMed
- 39. Chmurzynska A. The multigene family of fatty acid-binding proteins (FABPs): function, structure and polymorphism. J Appl Genet 2006;47:39–48. ArticlePubMedPDF
- 40. Maeda K, Cao H, Kono K, Gorgun CZ, Furuhashi M, Uysal KT, Cao Q, Atsumi G, Malone H, Krishnan B, Minokoshi Y, Kahn BB, Parker RA, Hotamisligil GS. Adipocyte/macrophage fatty acid binding proteins control integrated metabolic responses in obesity and diabetes. Cell Metab 2005;1:107–119. ArticlePubMed
- 41. Karakas SE, Almario RU, Kim K. Serum fatty acid binding protein 4, free fatty acids, and metabolic risk markers. Metabolism 2009;58:1002–1007. ArticlePubMedPMC
- 42. Bagheri R, Qasim AN, Mehta NN, Terembula K, Kapoor S, Braunstein S, Schutta M, Iqbal N, Lehrke M, Reilly MP. Relation of plasma fatty acid binding proteins 4 and 5 with the metabolic syndrome, inflammation and coronary calcium in patients with type-2 diabetes mellitus. Am J Cardiol 2010;106:1118–1123. ArticlePubMedPMC
- 43. Krusinova E, Pelikanova T. Fatty acid binding proteins in adipose tissue: a promising link between metabolic syndrome and atherosclerosis? Diabetes Res Clin Pract 2008;82(Suppl 2):S127–S134. ArticlePubMed
- 44. Kim JH, Bachmann RA, Chen J. Interleukin-6 and insulin resistance. Vitam Horm 2009;80:613–633. ArticlePubMed
- 45. Chan KH, Brennan K, You NC, Lu X, Song Y, Hsu YH, Chaudhuri G, Nathan L, Tinker L, Liu S. Common variations in the genes encoding C-reactive protein, tumor necrosis factor-alpha, and interleukin-6, and the risk of clinical diabetes in the Women's Health Initiative Observational Study. Clin Chem 2011;57:317–325. ArticlePubMedPDF
- 46. Suzuki T, Imai J, Yamada T, Ishigaki Y, Kaneko K, Uno K, Hasegawa Y, Ishihara H, Oka Y, Katagiri H. Interleukin-6 enhances glucose-stimulated insulin secretion from pancreatic beta-cells: potential involvement of the PLC-IP3-dependent pathway. Diabetes 2011;60:537–547. ArticlePubMedPMC
- 47. Febbraio MA, Pedersen BK. Muscle-derived interleukin-6: mechanisms for activation and possible biological roles. FASEB J 2002;16:1335–1347. ArticlePubMed
- 48. Al-Khalili L, Bouzakri K, Glund S, Lonnqvist F, Koistinen HA, Krook A. Signaling specificity of interleukin-6 action on glucose and lipid metabolism in skeletal muscle. Mol Endocrinol 2006;20:3364–3375. ArticlePubMedPDF
- 49. Sartipy P, Loskutoff DJ. Monocyte chemoattractant protein 1 in obesity and insulin resistance. Proc Natl Acad Sci U S A 2003;100:7265–7270. ArticlePubMedPMC
- 50. Yu R, Kim CS, Kawada T, Kwon TW, Lim TH, Kim YW, Kwon BS. Involvement of leukotactin-1, a novel CC chemokine, in human atherosclerosis. Atherosclerosis 2004;174:35–42. ArticlePubMed
- 51. Murdolo G, Nowotny B, Celi F, Donati M, Bini V, Papi F, Gornitzka G, Castellani S, Roden M, Falorni A, Herder C, Falorni A. Inflammatory adipokines, high molecular weight adiponectin, and insulin resistance: a population-based survey in prepubertal schoolchildren. PLoS One 2011;6:e17264ArticlePubMedPMC
- 52. Zhang Y, Zhang J, Tian C, Narenqimuge , Deng Y, Zhao Y, Fan H, Huang J. The -2518A/G polymorphism in the monocyte chemoattractant protein-1 (MCP-1) gene and diabetes risk: a meta-analysis. Diabetes Res Clin Pract 2011;94:e89–e92. ArticlePubMed
- 53. Hinnouho GM, Czernichow S, Dugravot A, Batty GD, Kivimaki M, Singh-Manoux A. Metabolically healthy obesity and risk of mortality: does the definition of metabolic health matter? Diabetes Care 2013;36:2294–2300. ArticlePubMedPMC
References
Comparison of mean (A) tumor necrosis factor α (TNF-α) and (B) adipocyte fatty acid binding protein (A-FABP) levels according to four groups divided by metabolic health and obesity. MHNO, metabolically healthy nonobese; MHO, metabolically healthy obese; MUHNO, metabolically unhealthy nonobese; MUHO, metabolically unhealthy obese. a,bDifferent letters denote significant differences between the designated groups in post hoc analyses. Otherwise, groups showed no significant differences between each groups with post hoc analyses.
General Characteristics of the Participants (n=456)
Values are expressed as mean±SD or number (%).
SBP, systolic blood pressure; DBP, diastolic blood pressure; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; HOMA-IR, homeostatic model assessment of insulin resistance; IL-6, interleukin-6; MCP-1, monocyte chemoattractant protein-1; TNF-α, tumor necrosis factor α; A-FABP, adipocyte fatty acid binding protein.
Comparison of Metabolic Parameters according to Metabolic Health and Obesity
Values are expressed as mean±SD or number (%).
MHNO, metabolically healthy nonobese; MHO, metabolically healthy obese; MUHNO, metabolically unhealthy nonobese; MUHO, metabolically unhealthy obese; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; SBP, systolic blood pressure; DBP, diastolic blood pressure; HOMA-IR, homeostatic model assessment of insulin resistance; IL-6, interleukin-6; MCP-1, monocyte chemoattractant protein-1; TNF-α, tumor necrosis factor α; A-FABP, adipocyte fatty acid binding protein.
aP values for one-way analysis of variance test;b,c,d,eDifferent letters denote significant differences between the designated groups in post hoc analyses. Otherwise, groups showed no significant differences between each groups with post hoc analyses.
Bivariate Correlation Analyses of Metabolic Parameters and Adipocytokines
IL-6, interleukin-6; MCP-1, monocyte chemoattractant protein-1; TNF-α, tumor necrosis factor α; A-FABP, adipocyte fatty acid binding protein; HDL-C, high density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; SBP, systolic blood pressure; DBP, diastolic blood pressure; HOMA-IR, homeostatic model assessment of insulin resistance.
aP<0.01 for the bivariate correlation analysis; bP<0.05 for the bivariate correlation analysis.
Figure & Data
References
Citations

- Differences in the levels of inflammatory markers between metabolically healthy obese and other obesity phenotypes in adults: A systematic review and meta-analysis
Zhouli Su, Ljupcho Efremov, Rafael Mikolajczyk
Nutrition, Metabolism and Cardiovascular Diseases.2024; 34(2): 251. CrossRef - Relationship between the thrombospondin-1/Toll-like receptor 4 (TSP1/TLR4) pathway and vitamin D levels in obese and normal weight subjects with different metabolic phenotypes
Eman Y. Khairy, Azza Saad
The Journal of Physiological Sciences.2023;[Epub] CrossRef - Pattern of Adiponectin, Osteocalcin, Irisin, FGF-21, and MCP-1 According to the Body Size Phenotype: Could They Be Markers of Metabolic Health in Mexican-Mestizo Middle-Aged Women?
Lourdes Balcázar-Hernandez, Lourdes Basurto, Leticia Manuel-Apolinar, Sara Vega-García, Norma Basurto-Acevedo, Carlos Martínez-Murillo, Rosalinda Sánchez-Arenas
Metabolites.2021; 11(11): 771. CrossRef - Exploring Therapeutic Targets to Reverse or Prevent the Transition from Metabolically Healthy to Unhealthy Obesity
Tenzin D. Dagpo, Christopher J. Nolan, Viviane Delghingaro-Augusto
Cells.2020; 9(7): 1596. CrossRef - Poor Vitamin D Status in Active Pulmonary Tuberculosis Patients and Its Correlation with Leptin and TNF-α
Qiuzhen WANG, Aiguo MA, Tianlin GAO, Yufeng LIU, Lisheng REN, Lei HAN, Boyang WEI, Qian LIU, Chunjiang DONG, Yuze MU, Duo LI, Frans J KOK, Evert G SCHOUTEN
Journal of Nutritional Science and Vitaminology.2019; 65(5): 390. CrossRef - Metabolic Health—The Role of Adipo-Myokines
Christine Graf, Nina Ferrari
International Journal of Molecular Sciences.2019; 20(24): 6159. CrossRef - Does the Metabolically Healthy Obese Phenotype Protect Adults with Class III Obesity from Biochemical Alterations Related to Bone Metabolism?
Ligiane Marques Loureiro, Suzane Lessa, Rodrigo Mendes, Sílvia Pereira, Carlos José Saboya, Andrea Ramalho
Nutrients.2019; 11(9): 2125. CrossRef - Being Metabolically Healthy, the Most Responsible Factor for Vascular Health
Eun-Jung Rhee
Diabetes & Metabolism Journal.2018; 42(1): 19. CrossRef - Association between Serum Dipeptidyl Peptidase-4 Concentration and Obesity-related Factors in Health Screen Examinees
Ji Yeon Lee, Byoung Kuk Jang, Min Kyung Song, Hye Soon Kim, Mi-Kyung Kim
Journal of Obesity & Metabolic Syndrome.2017; 26(3): 188. CrossRef - Kofaktoren und Komorbiditäten bei Necrobiosis lipoidica – Analyse der deutschen DRG‐Daten von 2012
Finja Jockenhöfer, Knut Kröger, Joachim Klode, Regina Renner, Cornelia Erfurt‐Berge, Joachim Dissemond
JDDG: Journal der Deutschen Dermatologischen Gesellschaft.2016; 14(3): 277. CrossRef - The relationship between serum fatty-acid binding protein 4 level and lung function in Korean subjects with normal ventilatory function
Hye-Jeong Park, Se Eun Park, Cheol-Young Park, Seong Yong Lim, Won-Young Lee, Ki-Won Oh, Sung-Woo Park, Eun-Jung Rhee
BMC Pulmonary Medicine.2016;[Epub] CrossRef - Cofactors and comorbidities of necrobiosis lipoidica: analysis of the German DRG data from 2012
Finja Jockenhöfer, Knut Kröger, Joachim Klode, Regina Renner, Cornelia Erfurt‐Berge, Joachim Dissemond
JDDG: Journal der Deutschen Dermatologischen Gesellschaft.2016; 14(3): 277. CrossRef - Response: Comparison of Serum Adipocytokine Levels according to Metabolic Health and Obesity Status (Endocrinol Metab2015;30:185-94, Tae Hoon Lee et al.)
Eun-Jung Rhee
Endocrinology and Metabolism.2015; 30(3): 416. CrossRef - Letter: Comparison of Serum Adipocytokine Levels according to Metabolic Health and Obesity Status (Endocrinol Metab2015;30:185-94, Tae Hoon Lee et al.)
Mikyung Kim
Endocrinology and Metabolism.2015; 30(3): 414. CrossRef - Adipokine Profiles and Metabolic Health
Seung-Hwan Lee
Endocrinology and Metabolism.2015; 30(2): 175. CrossRef

 KES
KES
 PubReader
PubReader Cite
Cite