Epidemiology of Childhood Obesity in Korea
Article information
Abstract
Over the past several decades, the prevalence of obesity has increased dramatically worldwide and is increasing not only in developed countries, but also in developing countries. This increase may lead to an increase in the incidence of chronic diseases throughout the lifespan. In Korean children and adolescents, the prevalence of obesity increased from 6.8% in 1998 to 10.0% in 2013. Obesity is a state that more commonly influences children and adolescents of lower socioeconomic status (SES) than those with a higher SES. However, the prevalence of metabolic syndrome in a nationally representative sample of Korean adolescents decreased from 1998 to 2012. According to the Diabetes Fact Sheet of the Korean Diabetes Association, the prevalence of type 2 diabetes among children aged 18 years or younger was 153.5 per 100,000 in 2006 and 205.0 per 100,000 in 2013. Obesity is a complex disease influenced by many interacting factors, such as adipocytokines, lipopolysaccharide-binding protein, adenovirus 36 infection, birth weight, lifestyle, and endocrine-disrupting chemicals. Obesity in youth can adversely impact practically every organ system and lead to serious consequences, such as metabolic, gastrointestinal, pulmonary, cardiovascular, and psychosocial complications. Therefore, coordinated efforts by governments, organizations, communities, and individuals are needed to prevent and treat childhood obesity. In particular, a long-term policy to improve the social environment will also be necessary.
INTRODUCTION
The global prevalence of obesity has increased dramatically over the past several decades. Overweight and obesity are among the leading causes of all-cause mortality and disability-adjusted life years (DALYs); an estimated 3.4 million people died due to overweight and obesity in 2010, which accounted for 4% of the DALYs that year [1]. In 2008, the burden of obesity was estimated to be 1.46 billion overweight adults and 502 million obese adults [2]. The prevalence of child overweight and obesity has also increased not only in developed countries, but also in developing countries [3]. Overweight and obesity are particularly well documented as contributors to chronic diseases in adults [4]. A similar rise in the prevalence of childhood overweight and obesity might also lead to an increase in the incidence of chronic diseases throughout the lifespan.
OBESITY, METABOLIC SYNDROME, AND TYPE 2 DIABETES
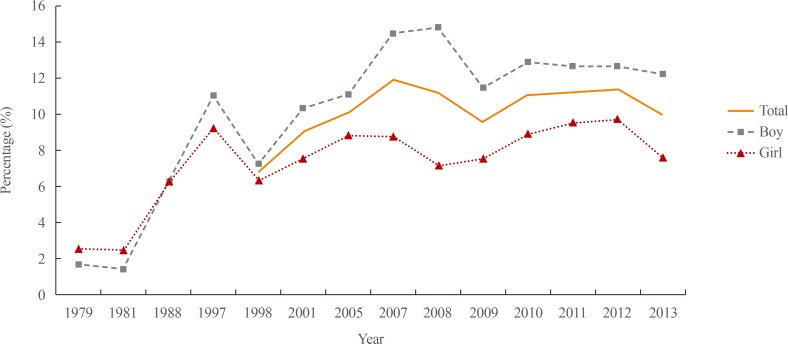
Overall, the prevalence of obesity increased from 6.8% to 10.0% of Korean children and adolescents during the period 1998 to 2013. The prevalence of obesity has increased dramatically during the past 35 years, climbing 6-fold in boys and 3-fold in girls (Table 1, Fig. 1) [56]. Obesity is a state that has a greater influence on children and adolescents of lower socioeconomic status (SES) compared with those of higher SES. From 1998 to 2009, the prevalence of obesity in Korean children and adolescents aged 2 to 18 years in the low-SES groups (lowest income quartile) increased from 5.0% to 9.7%. However, in those with a higher SES (highest income quartile), the prevalence of obesity decreased from 6.6% to 5.5% in that same time period [7]. Many studies have reported that SES, which can influence lifestyle such as food choice and physical activity, is associated with a risk of obesity in both adults and children [89]. In Korea, Kang et al. [10] have reported that a low paternal education level and the presence of non-parental caregivers were associated with a risk of childhood obesity. Oh and Hong [11] have indicated that the rates of household food insecurity were high in low-income urban families, and household food insecurity is related to childhood obesity.
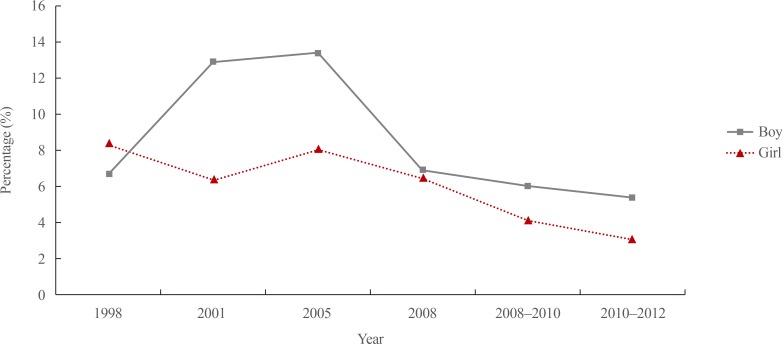
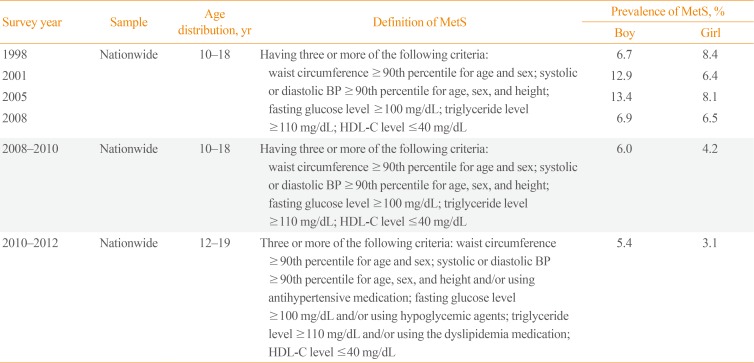
Obesity is principally responsible to the growing worldwide prevalence of metabolic syndrome (MetS), which consists of a cluster of metabolic disorders that include central obesity, glucose intolerance, insulin resistance, dyslipidemia, and hypertension. A nationally representative sample of Korean adolescents revealed that the rate of MetS decreased from 1998 to 2012 (5.4% of boys and 3.1% of girls had MetS in 2012, which was down from 6.7% of boys and 8.4% of girls in 1998) (Table 2, Fig. 2); previous studies have indicated that obesity is associated with an increased risk of MetS in children [1213141516]. The rate of MetS increased directly with the grade of obesity [17]. In Korea, overweight adolescents aged 12 to 13 years were also found to have a higher prevalence of MetS compared with their normal-weight peers [18]. Kim et al. [19] have reported that overweight children and adolescents aged 10 to 18 years in Korea had elevated blood pressure, abnormal lipid profiles, and an increasing number of MetS components compared with normal-weight children and adolescents.
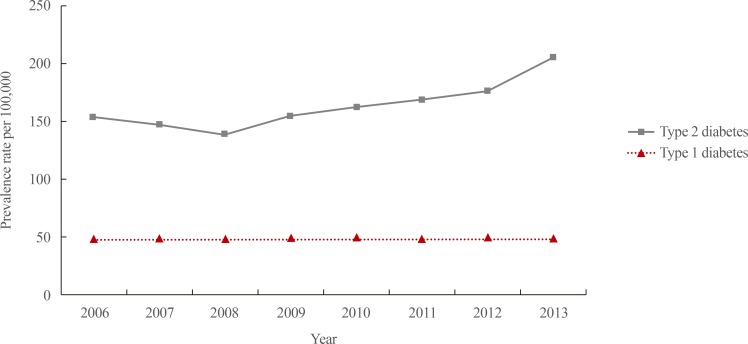
Obesity is the most important risk factor of the development of type 2 diabetes in children and adolescents [20]. A previous study has indicated that an increase in the frequency of obesity induced an increase in type 2 diabetes in youth [21]. The increased amount of adipose tissue in obese individuals produces esterified fatty acids, glycerol, hormones, proinflammatory cytokines, and other factors that contribute to insulin resistance, an important factor in the development of type 2 diabetes [2022]. According to the Diabetes Fact Sheet of the Korean Diabetes Association, the prevalence of type 2 diabetes among children 18 years of age or younger was 153.5 per 100,000 in 2006 and 205.0 per 100,000 in 2013, representing a relative increase of 33.6%. Although the prevalence of type 2 diabetes might be overestimated, it is apparent that the rate of this condition in children and adolescents is dramatically increasing in Korea. However, the prevalence rates of type 1 diabetes in children 18 years of age or younger in 2006 and 2013 were nearly identical: 47.7 per 100,000 and 48.1 per 100,000, respectively (Fig. 3) [23].
RISK FACTORS OF OBESITY IN CHILDREN AND ADOLESCENTS
Obesity is a complex disease that is influenced by many interacting genetic and non-genetic factors [24]. Adipocytokines, such as leptin, adiponectin, visfatin, resistin, and adipocyte fatty acid-binding protein, are a marker of obesity. In Korean children and adolescents, the visfatin level was correlated with the amount of visceral fat, while the resistin level has also been associated with obesity indicators [2526]. In Korean children nine years of age, adipocyte fatty acid-binding protein was found to be related to obesity and abdominal obesity [27]. Previous studies have indicated an inverse relationship between adiponectin concentration and obesity in Korean children and adolescents [28293031].
Recently, obesity has been associated with increased microbial translocation [32]. Lipopolysaccharide-binding protein, the endogenous reactive biomarker produced in response to microbial translocation, was correlated with the risk of an elevated body mass index (BMI) and obesity-related insulin resistance in adolescents aged 12 to 13 years [33]. The role of bacteria and viruses has been suggested to be a risk factor of development of obesity. In Korea, the prevalence of adenovirus 36 infection was higher in obese children than in their peers of normal weight [34].
Birth weight has also been associated with risk of later obesity in children and adults. In Korea, among adolescents aged 12 to 15 years, low birth weight increased the risk of insulin resistance [35]. Another study has indicated that, although insulin resistance was unrelated to birth weight, currently being overweight was associated with insulin resistance [36]. Kim et al. [37] have reported that birth weight didn't influence adiponectin levels in adolescence.
Generally, lifestyle factors, such as inadequate intakes of vegetables, fruit and milk; eating too many high-calorie, low-nutrient foods; skipping breakfast; low levels of physical activity; daily TV viewing; and insufficient sleep, are well-recognized risk factors of weight gain and obesity [38]. In Korea, the traditional lifestyle has given way to a highly modernized lifestyle involving less physical exertion and more unhealthy eating habits. This change has contributed to the increase in obesity among children and adolescents [39]. In addition, fat and carbohydrate intake levels decreased among the adolescent population from 1998 to 2005. In addition, those with a Western dietary pattern of flour and bread, pizza and hamburgers, meat and meat products, dairy products, and soft drinks were found to face a higher risk of being overweight and having elevated serum triglycerides compared with those with a more traditional dietary pattern, including higher consumption of white rice, kimchi, vegetables, and fish [40]. Previous studies among children and adolescents have suggested an overall positive association of sugar-sweetened beverage consumption with obesity [41]. However, sugar intake from milk and fruits was inversely related to overweight and obesity in girls [42]. Sugar intake from different food sources might play an important role in the risk of obesity in children and adolescents [43].
In children aged 7 to 18 years, high sodium was associated with increased risk of obesity and abdominal obesity independent of calorie intake [44]. A randomized controlled trial study has suggested that physical activity enhanced insulin sensitivity in overweight adolescents [45]. Physical fitness, which is the ability to carry out daily tasks without fatigue, was also found to be lower in overweight/obese children than in the normal-weight group [46].
A previous study has reported an inverse relationship between sleep duration and BMI in children. Shorter sleep duration in children was identified in those with a higher BMI, which was calculated using Korean age- and sex-specific pediatric values for height and weight [47]. A shared familial environment is also an important risk factor. Park et al. [48] reported that maternal anthropometry and dietary behavior had a greater effect on the obesity of children than their paternal contributions. Lee et al. [49] demonstrated that children living with overweight parents or grandparents showed an association with a greater risk of childhood overweight and obesity.
Recently, exposure to endocrine-disrupting chemicals (EDCs) through the ingestion of contaminated food or water and the inhalation of polluted air or from dermal exposure has received attention as a key contributor to obesity. EDCs lead to obesity by changing the function of thyroid and steroid hormones and activating peroxisome proliferator-activated receptors, which play a major role in adipocyte differentiation and energy storage [50]. A previous study has suggested that bisphenol A, which is used to manufacture polycarbonate resin and is a breakdown product of coatings, was related to obesity in United States children and adolescents [51]. Persistent organic pollutants (POPs), which are lipophilic compounds that travel with lipids and accumulate mainly in the adipose tissue, are associated with an increased risk of obesity and type 2 diabetes [52]. In Korean children, POPs level has been shown to negatively affect the chance of metabolic components, such as blood pressure and triglyceride level [53]. Phthalate exposure has also increased after widespread use of plastic products and due to their use as vehicles for fragrance. Several studies have additionally reported an association between phthalates and the risk of obesity development in children [54]. In Korea, phthalate presence in urine and serum are associated with childhood obesity. Diethylhexyl phthalate exposure in particular was positively related to increase BMI during the first 3 months after birth [55]. The level of dehydroepiandrosterone, an endogenous steroid, has demonstrated a significant difference between obese and control groups [56].
PHYSICAL AND PSYCHOLOGICAL COMORBIDITIES
Obesity in youth can adversely impact almost every organ system and commonly leads to serious consequences, such as metabolic, gastrointestinal, pulmonary, cardiovascular, and psychosocial complications [57].
Childhood obesity is associated with an increased risk of metabolic complications in youth. In Korea, the prevalence of MetS was higher in overweight adolescents than in normal-weight adolescents [18]. The risk of dyslipidemia was also increased in obese children and adolescents compared to the normal-weight counterparts [19]. Lee et al. [58] have indicated that the apolipoprotein B/apolipoprotein A1 ratio, which reflects lipid disturbance, was higher in a patient with abdominal obesity; their findings were confirmed and have been associated with increased rates of MetS. Choi et al. [59] have suggested that low density lipoprotein particle size negatively correlates with BMI. Remnant lipoprotein cholesterol, which is the product of the lipolytic degradation of triglyceride-rich lipoproteins produced by the liver and intestine, was previously shown to be higher in obese children and adolescents [6061]. In addition, obese children and adolescents had higher blood pressure readings than their peers of a normal weight [62].
Nonalcoholic fatty liver disease (NAFLD), which is characterized by the accumulation of macrovascular fat in hepatocytes, can also affect children. Although the mechanism of action in pediatric NAFLD remains unclear, obesity and hyperinsulinemia along with insulin resistance play an important role [63]. In Korea, Kim et al. [64] have reported that the prevalence of NAFLD was higher in overweight than in normal-weight children. Ko et al. [65] have indicated that children with nonalcoholic steatohepatitis have a higher BMI than those with simple steatosis. Furthermore, in obese Korean children ranging in age from 9 to 12 years, insulin resistance was associated with the risk of NAFLD [66].
Pulmonary disorders are also related to obesity in children. Lung function was measured using the forced expiratory volume during one second to calculate the forced vital capacity ratio in obese children and was found to be significantly lower than in normal-weight children [67]. Hong et al. [68] have reported that high BMI is associated with an increased risk of asthma symptoms in boys. In addition, several studies have indicated an association of obesity with allergic rhinitis and atopic dermatitis in children and adolescents [6970].
Obesity in children and adolescents is also associated with psychosocial problems. Obese Korean children are often troubled with body dissatisfaction, which can lead to lower self-esteem and depression [71]. The psychological stress and depression caused by obesity are also associated with a poor health-related quality of life [72]. Furthermore, body dissatisfaction contributes to suicidal ideation in adolescents based upon a large and nationally representative sample of Korean adolescents [73].
CONCLUSIONS
Obesity in children and adolescents is a major public health issue worldwide and is conditioned by socioeconomic environment as well as individual behavior. Therefore, coordinated efforts by governments, organizations, communities, and individuals are needed to prevent and treat child obesity. In particular, a long-term policy to improve social environment is necessary. In addition, surveillance of childhood obesity and obesity-related diseases is required to effectively confront the childhood obesity epidemic.
ACKNOWLEDGMENTS
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), which was funded by the Ministry of Health and Welfare, Republic of Korea (Grant number HI13C0715).
Notes
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.