Percutaneous Adrenal Radiofrequency Ablation: A Short Review for Endocrinologists
Article information
Abstract
Image-guided radiofrequency ablation (RFA) has been accepted as a minimally invasive treatment for adrenal tumors in patients who are unable to undergo adrenalectomy. Accordingly, this treatment has become more readily available for treating functioning or non-functioning adrenal masses. Thus, endocrinologists need a better understanding of percutaneous RFA of adrenal tumors. The purpose of this review is to briefly describe the basic mechanism of RFA, indications and contraindications, patient preparation prior to RFA, type of complications, how to avoid complications, RFA procedures, and treatment outcomes.
INTRODUCTION
Percutaneous thermal ablation is a minimally invasive treatment for liver [1,2], kidney [3,4], lung [5,6], bone [7,8], and thyroid [9,10] tumors in patients who are unable to undergo surgery. This treatment also has been used to treat adrenal tumors in patients who are poor surgical candidates [11–23]. Thermal ablation techniques for adrenal ablation include radiofrequency ablation (RFA), cryoablation [24,25], and microwave ablation [26,27]. Among these techniques, RFA is most commonly used for adrenal tumors because it creates small ablation zone enough to cover small adrenal tumors.
The purpose of this review is to briefly describe the basic mechanism of RFA, indications and contraindications, patient preparations prior to RFA, type of complications, how to avoid complications, RFA procedures, and treatment outcomes.
BASIC MECHANISM
RFA elevates the temperature of tumor cells, leading to coagulative necrosis [28–31]. Alternating current is created by a radiofrequency generator and is delivered to the tumor through the electrode, which has an uninsulated tip that transmits electrical current to adjacent tissue [28–31]. Ions around the electrode tip vibrate when rapid alternating current is applied and heat energy is produced through ionic agitation. The frictional energy results in increasing tissue temperature and cell proteins begin to denature. The size of the RFA area depends on the electrical conductivity of tumor tissue around the electrode.
Internally-cooled RFA devices usually consist of an electrode, generator, pump, and pad [28–31]. Alternating current is created by the generator and is delivered to the electrode. The pump circulates normal saline to cool the electrode and improve electrical conductivity in the tumor tissue. The grounding pad is applied to the patient’s thigh or body.
INDICATIONS AND CONTRAINDICATIONS
Total or partial adrenalectomy is the treatment of choice for adrenal tumors [32–34]; RFA should be accepted as an alternative in patients who cannot undergo surgery due to high risk of post-operative morbidity or mortality [22,23,34–40]. Moreover, recurrent pheochromocytomas in patients with hereditary diseases such as von Hippel Lindau disease or multiple endocrine neoplasms may be treated with RFA because repeat adrenalectomy cannot avoid adrenal insufficiency.
RFA candidates should have a platelet count greater than 50,000 and an international normalized ratio less than 1.5 to 1.8 [22]. Bleeding tendency or coagulopathy should be corrected prior to RFA. Non-steroid anti-inflammatory or anti-coagulant medications should be stopped at least 1 week prior to RFA. Fever is a contraindication and thus should be controlled prior to RFA.
PATIENT PREPARATION
Hypertensive crisis is frequently encountered during RFA procedures due to excessive excretion of catecholamines from the ablated adrenal medulla [41–45]. Theoretically, this complication is more severe in patients with pheochromocytomas producing epinephrine and norepinephrine [46–49]. Two or three weeks prior to RFA, these patients should undergo preoperative blockade to prevent peri-procedural (or intra-procedural) cardiovascular complications and blood pressure (BP) control. Alpha blockers are recommended as the first choice. If tachycardia develops, then β-blockers should be administered.
General anesthesia is essential to precisely target the lesion and to safely control pain or BP. Hormone-excreting adrenal masses are frequently small in size and precise targeting is important [22]. Conscious sedation is not as advantageous for precise lesion targeting or safe control of pain or BP.
An arterial line should be secured to keep monitoring BP because of the possibility of rapid hemodynamic changes [41–45]. Anesthesiologists should have alpha blockers prepared in a syringe to intravenously inject as soon as rapid increases in BP are detected. Also, interventional radiologists should be ready to turn off the radiofrequency generator because hypertensive crises may not be well controlled with intravenous medication alone.
Percutaneous biopsy is not necessary for histologic diagnosis of functioning adrenal masses such as aldosterone- or cortisol-producing adenomas and pheochromocytomas; biochemical tests and imaging features are sufficient [22]. Image-guided biopsy is necessary only for patients with primary or secondary malignant adrenal tumors.
COMPLICATIONS AND PREVENTION
Major complications requiring hospital stay or intervention are not common, ranging from 0% to 8.3% (median, 1.5%) [11–18]. Hypertensive crisis is the most common complication during the procedure and rapid increases in BP may complicate cerebrovascular and cardiovascular diseases (Fig. 1) [41–45]. Appropriate medication is necessary to prevent hypertensive crisis prior to or during RFA procedures [50–53]. Interventional radiologists should be ready to turn off the generator during RFA procedures because BP is not well controlled due to excessive excretion of catecholamines.
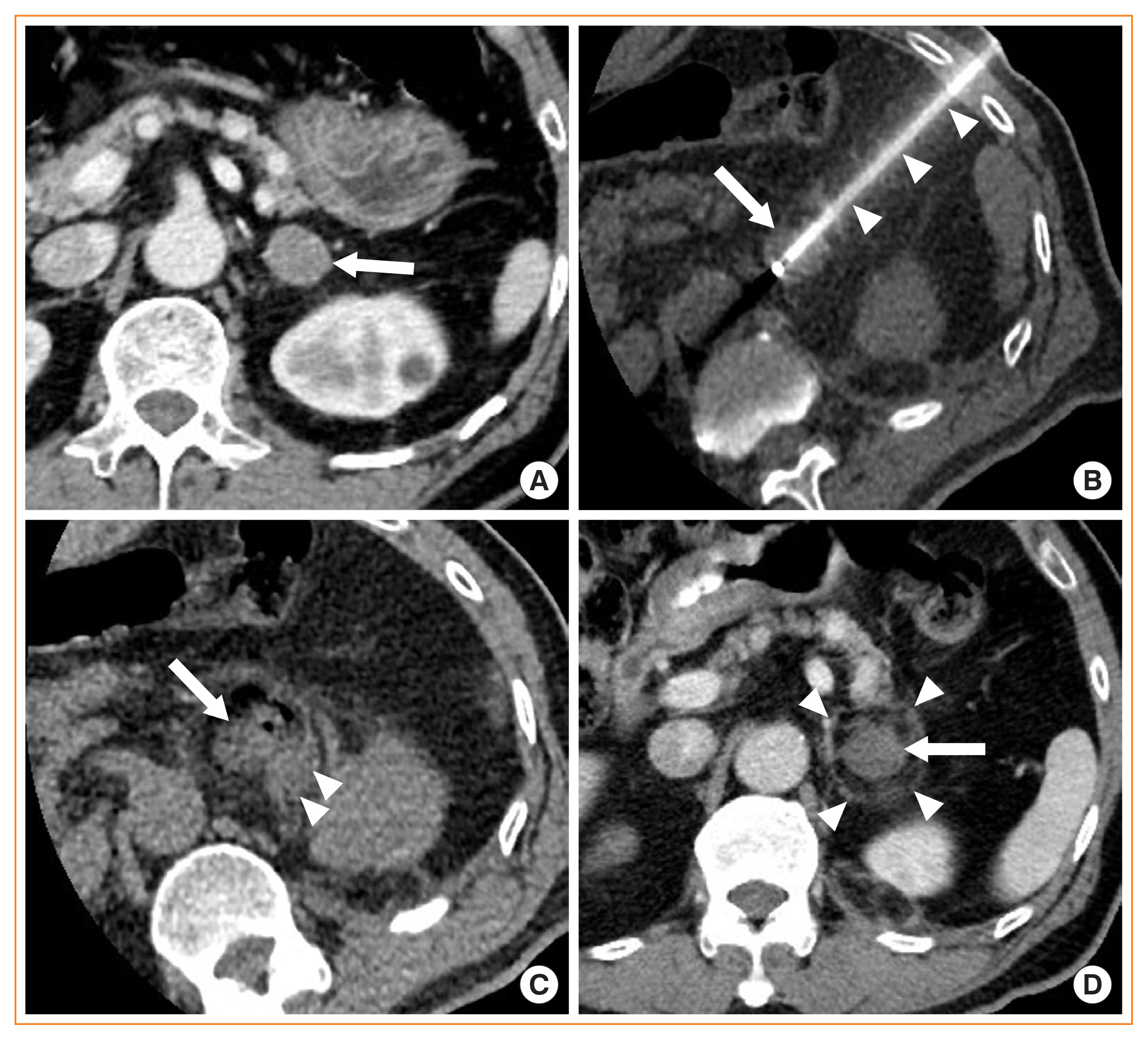
A 60-year-old man with aldosterone-producing adenoma. (A) Contrast-enhanced axial computed tomography (CT) image shows a 2.5 cm left adrenal adenoma (arrow) with good enhancement. Adrenalectomy was failed because of severe adhesion which had developed since left hemicolectomy was performed to treat descending colon cancer. (B) Unenhanced axial CT image obtained during ablation procedures shows a radiofrequency electrode (arrowheads), which is advanced via left intercostal space and placed in the center of the left adrenal adenoma (arrow). (C) Unenhanced axial CT image obtained immediately after ablation was finished shows a small amount of acute hematoma (arrowheads) around the left adrenal adenoma (arrow). Hypertensive crisis occurred three times, but was well controlled with intermittent intravenous injection of alpha blockers. But also, turning off the power of generator was added to the medication in the first cycle of ablation. (D) Contrast-enhanced CT image obtained 1 month after ablation was finished shows no enhancement within the left adrenal adenoma (arrow). Arrowheads indicate the margin of ablation zone. His aldosterone-to-renin ratio was 471.58 before ablation and 5.27 after ablation. However, his blood pressure was reduced, but still ranged slightly higher.
Pneumothorax is a common complication when the lung parenchyma is intervened between the skin and adrenal tumor [20,37–40,54]. A transhepatic approach provides another access route to reach right adrenal masses without lung injury when a patient is supine [22,37–40]. If the right or left side of the patient’s body is down, the ipsilateral lung tends to be collapsed. This position change is very useful to avoid pneumothorax when a patient is prone.
Bowel injury occurs when the tumor-to-bowel distance is less than approximately 5 mm [22,37–40]. Patient’ position change or levering a radiofrequency electrode may displace the tumor away from the bowel. When this does not work, hydrodissection can displace the bowel by means of instilling fluid between the tumor and bowel.
Bleeding is a common complication (Fig. 1) and meticulous manipulation of the radiofrequency electrode is one way to reduce this complication [22,37–40]. However, avoiding major organs is the best way to prevent serious bleeding [22,37–40].
Adrenal insufficiency has been reported as a rare complication following RFA [22,37–40]. Partial adrenalectomy is also useful for avoiding this complication, but is inferior to RFA in terms of preserving adrenal function. Further investigation is needed to compare the adrenal function outcomes of RFA and partial adrenalectomy.
RFA PROCEDURES
Computed tomography (CT) is the best imaging modality to guide adrenal RFA procedures [22,37–40]. Therefore, a ventilator and oxygen for general anesthesia should be available in the CT room. Ultrasound does not provide a good sonic window to access adrenal masses because the lesions are too deep and too small to target. Magnetic resonance imaging (MRI) does not achieve clear imaging of RFA procedures because the electrical current disturbs magnetic field, resulting in MRI distortion or artifacts. Moreover, MRI-compatible RFA devices are not commercially available.
RFA procedures comprise three steps: planning, targeting, and monitoring (Fig. 1) [55]. Planning involves locating the tumor and determining the skin entry site. Targeting is the second step, to place single or multiple electrodes within the tumor. Monitoring is the last step to determine whether the tumor and tumor margin are completely ablated and whether there are significant RFA complications.
RFA OUTCOMES
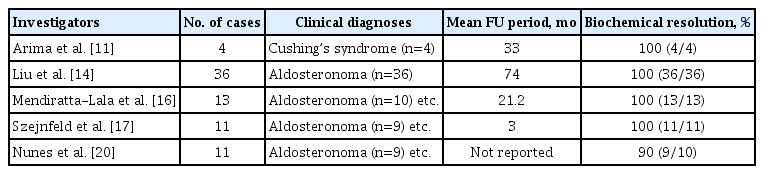
Percutaneous RFA provides excellent clinical and hormonal outcomes for patients with functional benign adrenal masses such as aldosteronoma, cortisol-secreting adenoma, and pheochromocytoma (Table 1) [11,14–17,19,20]. The most common functioning benign tumor treated is aldosteronoma. The number of cortisol-secreting adenomas or pheochromocytomas treated with RFA is much smaller than that of aldosteronomas [14–17,19,20]. Short- and long-term bio-chemical resolution have ranged from 90% to 100% (median, 100%) (Table 1) [11,14–17,19,20]. Normalization of aldosterone, renin, and aldosterone to renin ratio have been achieved in almost all cases of aldosteronoma [14–17,20]. However, persistent hypertension even after hormones are normalized is not uncommon (Fig. 1) [14,15,17,20].
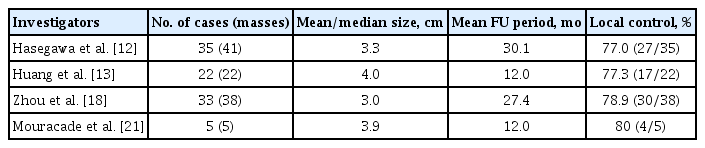
RFA outcomes for treating adrenal metastasis are not as good as those for benign adrenal tumors (Table 2) [12,13,18,21]. The local control rate ranges from 77% to 80% (median, 78%). These treatment outcomes may depend on metastatic tumor size, which is larger, ranging from 3.3 to 4.0 cm (Table 2) [12,13,18, 21,22]. Because cryoablation or microwave ablation can create a larger ablation area, these modalities should be considered for treating adrenal masses 3 cm or larger. Overall survival rate is poor because adrenal metastasis indicates stage IV disease, even in cases with a single metastatic tumor [12,13,18,21,22]. RFA outcomes for treating adrenocortical carcinoma are rarely reported because adrenalectomy is the treatment of choice for the primary adrenal cancer.
CONCLUSIONS
Percutaneous RFA is a safe and effective minimally invasive treatment for benign functioning adrenal tumors. This treatment technique also provides relatively good local control in patients with adrenal metastasis. Thus, RFA is a good alternative treatment in patients who are poor candidates for adrenalectomy.
Notes
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.