Articles
- Page Path
- HOME > Endocrinol Metab > Volume 36(2); 2021 > Article
-
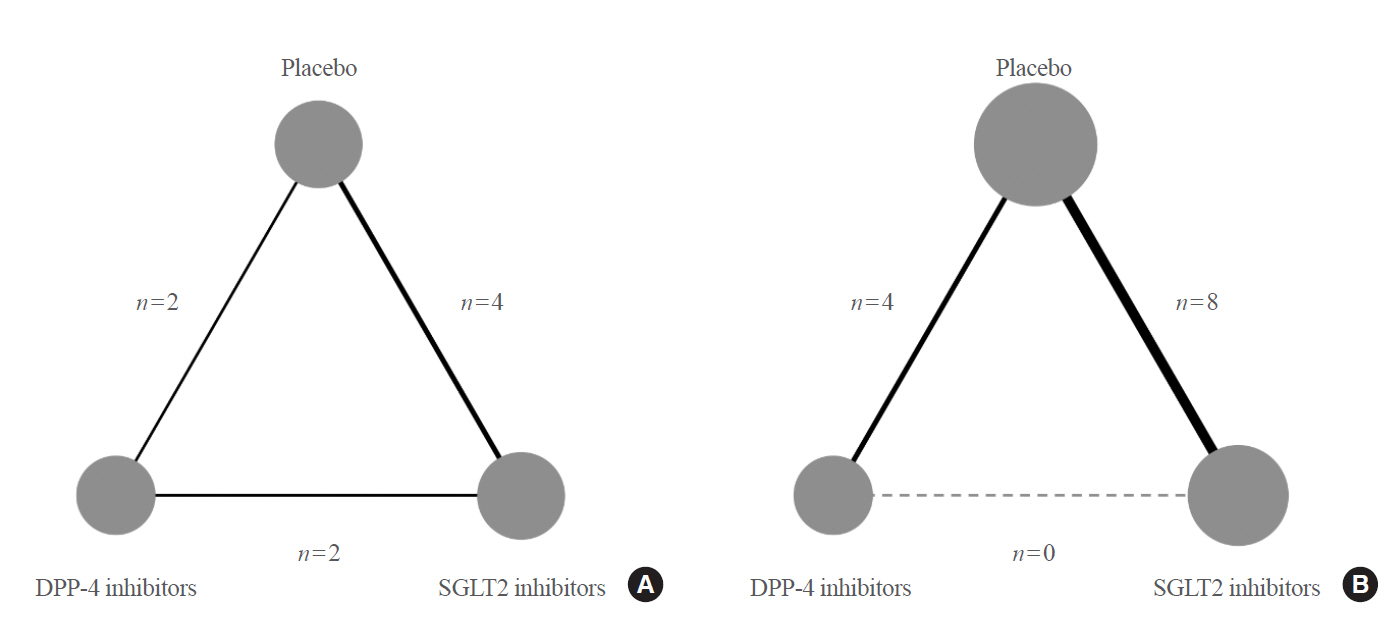
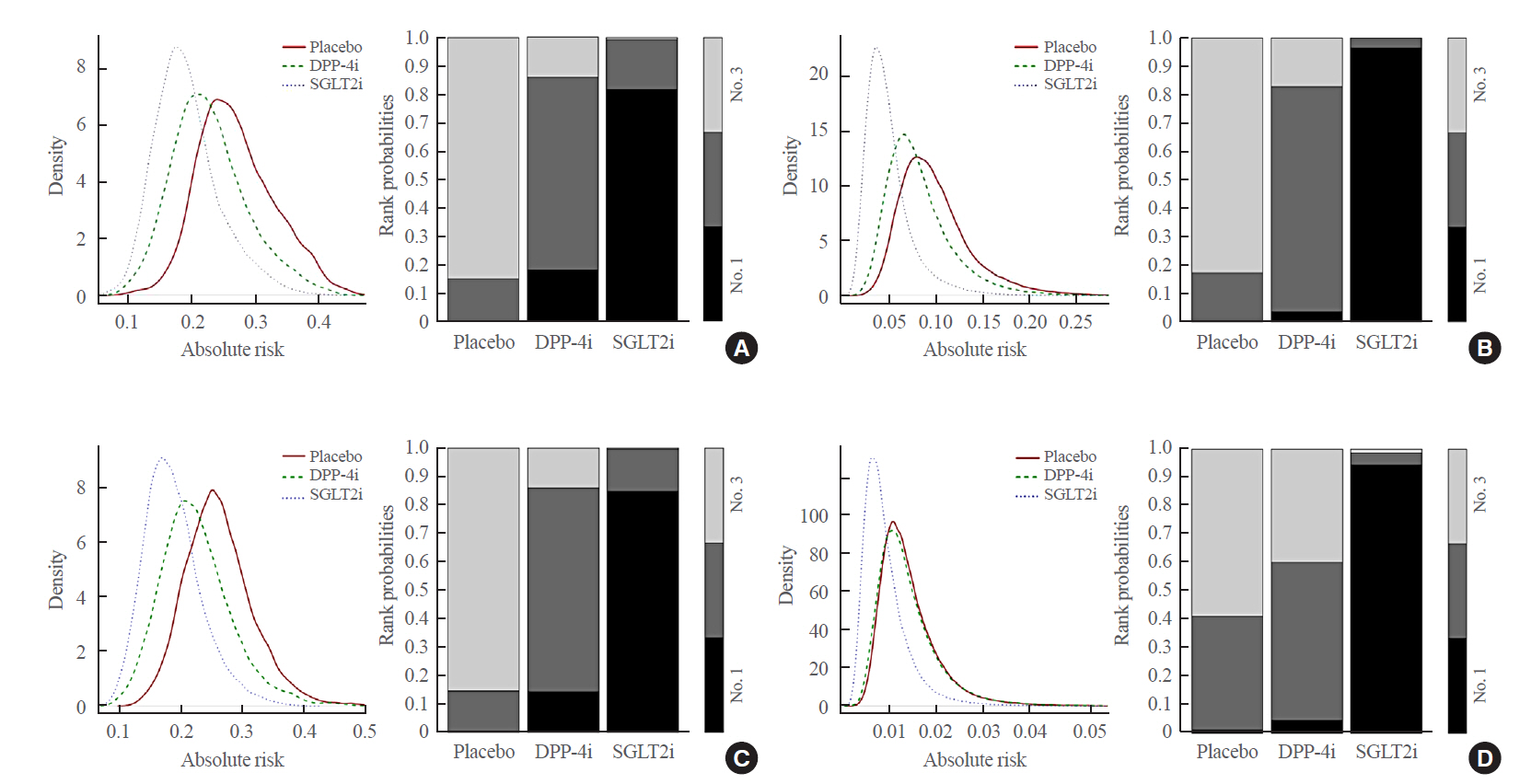
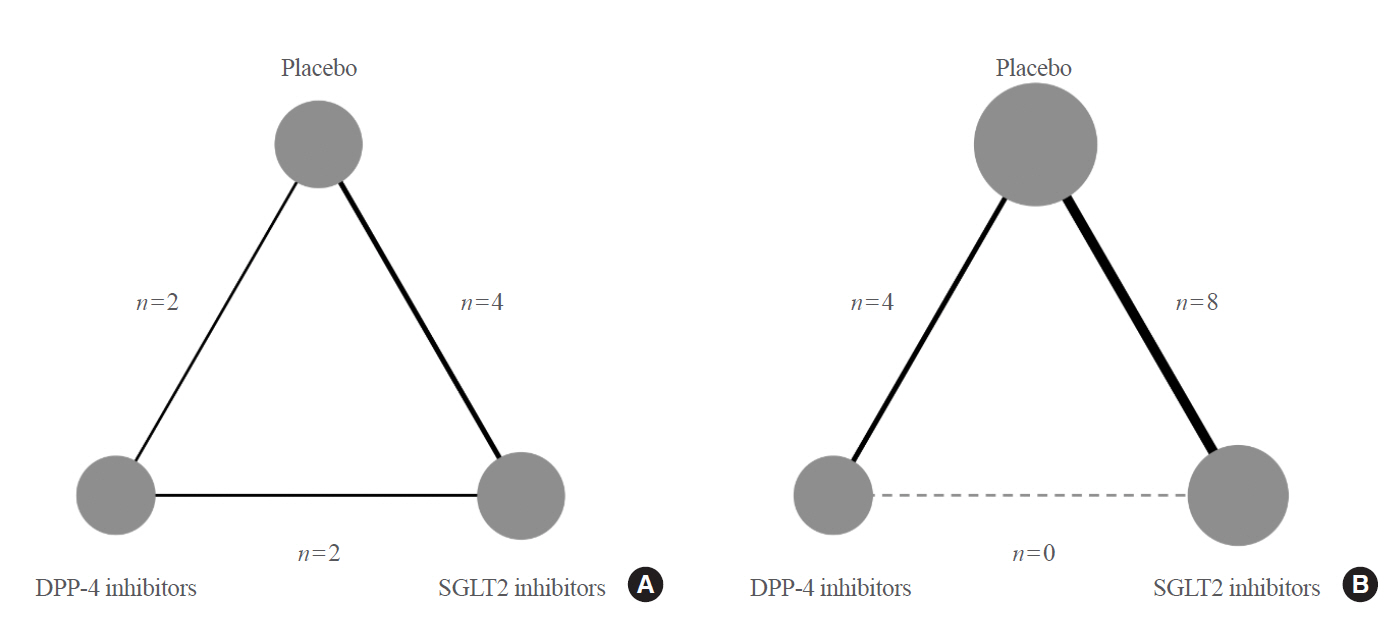
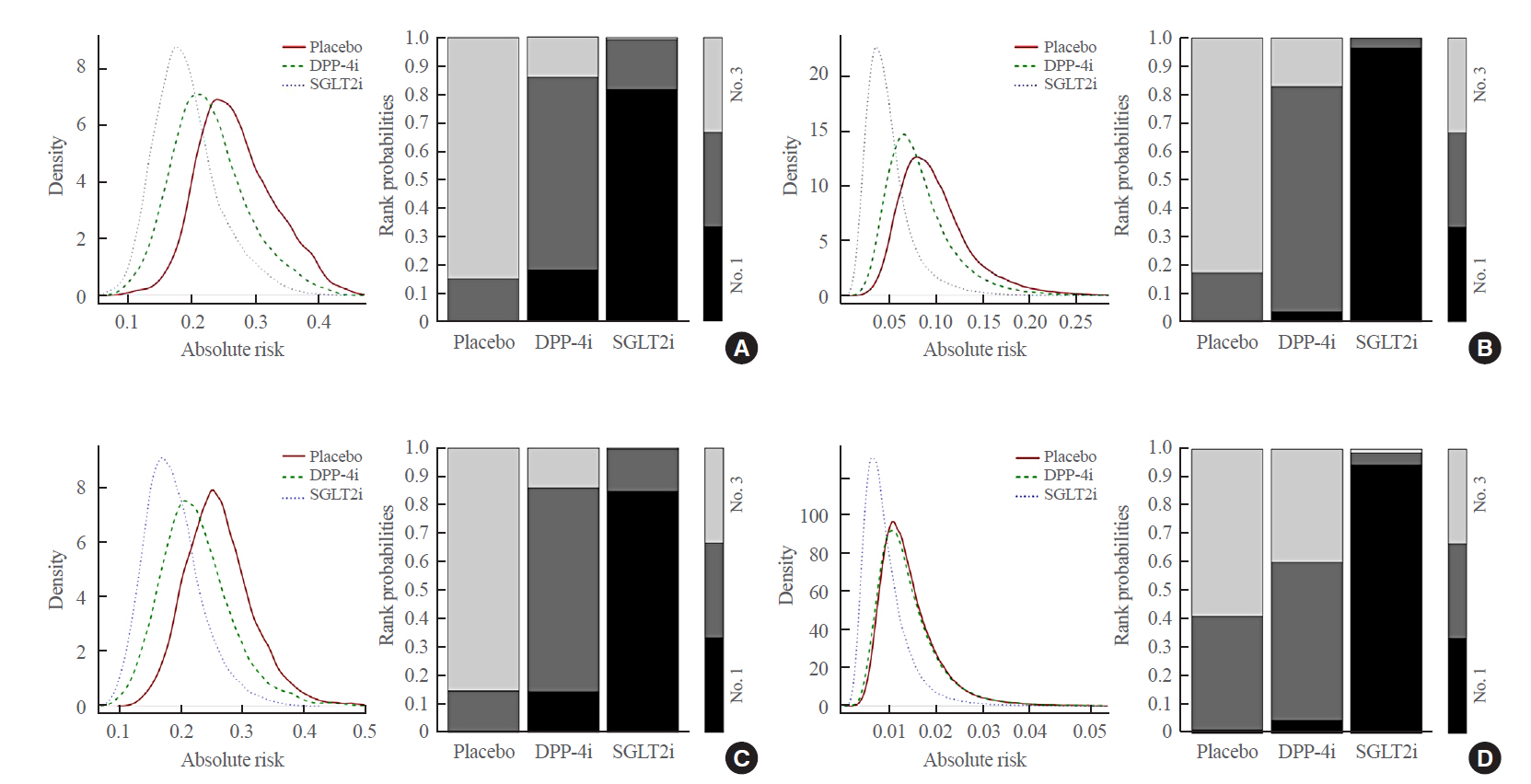
Original ArticleClinical Study Comparative Renal Effects of Dipeptidyl Peptidase-4 Inhibitors and Sodium-Glucose Cotransporter 2 Inhibitors on Individual Outcomes in Patients with Type 2 Diabetes: A Systematic Review and Network Meta-Analysis
-
Jae Hyun Bae1*
 , Eun-Gee Park2*
, Eun-Gee Park2* , Sunhee Kim2, Sin Gon Kim1, Seokyung Hahn3,4,5
, Sunhee Kim2, Sin Gon Kim1, Seokyung Hahn3,4,5 , Nam Hoon Kim1
, Nam Hoon Kim1
-
Endocrinology and Metabolism 2021;36(2):388-400.
DOI: https://doi.org/10.3803/EnM.2020.912
Published online: March 31, 2021
1Department of Internal Medicine, Korea University Anam Hospital, Korea University College of Medicine, Seoul, Korea
2Interdisciplinary Program in Medical Informatics, Seoul National University College of Medicine, Seoul, Korea
3Division of Medical Statistics, Medical Research Collaborating Center, Seoul National University Hospital, Seoul, Korea
4Department of Human Systems Medicine, Seoul National University College of Medicine, Seoul, Korea
5Institute of Health Policy and Management, Medical Research Center, Seoul National University, Seoul, Korea
- Corresponding authors: Nam Hoon Kim. Department of Internal Medicine, Korea University College of Medicine, 73 Goryeodae-ro, Seongbuk-gu, Seoul 02841, Korea, Tel: +82-2-920-6268, Fax: +82-2-953-9355, E-mail: pourlife@korea.ac.kr.
- Seokyung Hahn. Department of Human Systems Medicine, Seoul National University College of Medicine, 103 Daehak-ro, Jongno-gu, Seoul 03080, Korea, Tel: +82-2-740-8911, Fax: +82-2-743-8361, E-mail: hahns@snu.ac.kr
- * These authors contributed equally to this work.
Copyright © 2021 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Figure & Data
References
Citations

- Therapie des Typ-2-Diabetes
Rüdiger Landgraf, Jens Aberle, Andreas L. Birkenfeld, Baptist Gallwitz, Monika Kellerer, Harald H. Klein, Dirk Müller-Wieland, Michael A. Nauck, Tobias Wiesner, Erhard Siegel
Die Diabetologie.2024; 20(2): 212. CrossRef - Ipragliflozin and sitagliptin differentially affect lipid and apolipoprotein profiles in type 2 diabetes: the SUCRE study
Mototsugu Nagao, Jun Sasaki, Kyoko Tanimura-Inagaki, Ichiro Sakuma, Hitoshi Sugihara, Shinichi Oikawa
Cardiovascular Diabetology.2024;[Epub] CrossRef - Comparative Effect of Glucose-Lowering Drugs for Type 2 Diabetes Mellitus on Stroke Prevention: A Systematic Review and Network Meta-Analysis
Ji Soo Kim, Gyeongsil Lee, Kyung-Il Park, Seung-Won Oh
Diabetes & Metabolism Journal.2024; 48(2): 312. CrossRef - Therapy of Type 2 Diabetes
Rüdiger Landgraf, Jens Aberle, Andreas L. Birkenfeld, Baptist Gallwitz, Monika Kellerer, Harald H. Klein, Dirk Müller-Wieland, Michael A. Nauck, Tobias Wiesner, Erhard Siegel
Experimental and Clinical Endocrinology & Diabetes.2024;[Epub] CrossRef - Therapie des Typ-2-Diabetes
Rüdiger Landgraf, Jens Aberle, Andreas L. Birkenfeld, Baptist Gallwitz, Monika Kellerer, Harald H. Klein, Dirk Müller-Wieland, Michael A. Nauck, Tobias Wiesner, Erhard Siegel
Die Diabetologie.2023; 19(5): 658. CrossRef - Renoprotective Effect of Thai Patients with Type 2 Diabetes Mellitus Treated with SGLT-2 Inhibitors versus DPP-4 Inhibitors: A Real-World Observational Study
Apichaya Chanawong, Suriyon Uitrakul, Supatcha Incomenoy, Natnicha Poonchuay, Rizky Abdulah
Advances in Pharmacological and Pharmaceutical Sciences.2023; 2023: 1. CrossRef - Different nursing interventions on sleep quality among critically ill patients: A systematic review and network meta-analysis
Daijin Huang, Yumei Li, Jing Ye, Chang Liu, Dongyan Shen, Yunhui Lv
Medicine.2023; 102(52): e36298. CrossRef - New trends in the approach to the treatment of type 2 diabetes - observations and benefits in the outpatient practice of a diabetologist
Pavel Weber, Hana Meluzínová, Dana Weberová
Klinická farmakologie a farmacie.2022; 35(4): 118. CrossRef - Comparative efficacy of novel antidiabetic drugs on cardiovascular and renal outcomes in patients with diabetic kidney disease: A systematic review and network meta‐analysis
Hongwei Cao, Tao Liu, Li Wang, Qiuhe Ji
Diabetes, Obesity and Metabolism.2022; 24(8): 1448. CrossRef - Therapie des Typ-2-Diabetes
Rüdiger Landgraf, Jens Aberle, Andreas L. Birkenfeld, Baptist Gallwitz, Monika Kellerer, Harald H. Klein, Dirk Müller-Wieland, Michael A. Nauck, Tobias Wiesner, Erhard Siegel
Die Diabetologie.2022; 18(5): 623. CrossRef - Significant reduction in chronic kidney disease progression with sodium‐glucose cotransporter‐2 inhibitors compared to dipeptidyl peptidase‐4 inhibitors in adults with type 2 diabetes in a UK clinical setting: An observational outcomes study based on inte
Iskandar Idris, Ruiqi Zhang, Jil B. Mamza, Mike Ford, Tamsin Morris, Amitava Banerjee, Kamlesh Khunti
Diabetes, Obesity and Metabolism.2022; 24(11): 2138. CrossRef - Therapy of Type 2 Diabetes
Rüdiger Landgraf, Jens Aberle, Andreas L. Birkenfeld, Baptist Gallwitz, Monika Kellerer, Harald Klein, Dirk Müller-Wieland, Michael A. Nauck, Tobias Wiesner, Erhard Siegel
Experimental and Clinical Endocrinology & Diabetes.2022; 130(S 01): S80. CrossRef - Molecular Mechanistic Pathways Targeted by Natural Compounds in the Prevention and Treatment of Diabetic Kidney Disease
Kaixuan Zhou, Xue Zi, Jiayu Song, Qiulu Zhao, Jia Liu, Huiwei Bao, Lijing Li
Molecules.2022; 27(19): 6221. CrossRef - Lower risk of gout in sodium glucose cotransporter 2 (SGLT2) inhibitors versus dipeptidyl peptidase-4 (DPP4) inhibitors in type-2 diabetes
Jiandong Zhou, Xuejin Liu, Oscar Hou-In Chou, Lifang Li, Sharen Lee, Wing Tak Wong, Qingpeng Zhang, Carlin Chang, Tong Liu, Gary Tse, Fengshi Jing, Bernard Man Yung Cheung
Rheumatology.2022;[Epub] CrossRef - New Era for Renal-Protective Therapy in Type 2 Diabetes: Better Renal Outcomes in Patients with Type 2 Diabetes Taking Sodium-Glucose Cotransporter 2 Inhibitors versus Dipeptidyl Peptidase-4 Inhibitors
Chan-Hee Jung
Endocrinology and Metabolism.2021; 36(2): 339. CrossRef - Efficacy / safety balance of DPP-4 inhibitors versus SGLT2 inhibitors in elderly patients with type 2 diabetes
André J. Scheen
Diabetes & Metabolism.2021; 47(6): 101275. CrossRef

 KES
KES
 PubReader
PubReader ePub Link
ePub Link Cite
Cite