Efficacy and Safety of Long-Term Methimazole versus Radioactive Iodine in the Treatment of Toxic Multinodular Goiter
Article information
Abstract
Background
This study compared the degree of sustained control of hyperthyroidism in patients with toxic multinodular goiter (TMNG) treated with long-term methimazole (LT-MMI) or radioactive iodine (RAI).
Methods
In this clinical trial, 130 untreated patients with TMNG were randomized to either LT-MMI or RAI treatment. Both groups were followed for 108 to 148 months, with median follow-up durations of 120 and 132 months in the LT-MMI and RAI groups, respectively. Both groups of patients were followed every 1 to 3 months in the first year and every 6 months thereafter.
Results
After excluding patients in whom the treatment modality was changed and those who were lost to follow-up, 53 patients in the LT-MMI group and 54 in the RAI group completed the study. At the end of the study period, 50 (96%) and 25 (46%) patients were euthyroid, and two (4%) and 25 (46%) were hypothyroid in LT-MMI and RAI groups, respectively. In the RAI group, four (8%) patients had subclinical hyperthyroidism. The mean time to euthyroidism was 4.3±1.3 months in LT-MMI patients and 16.3± 15.0 months in RAI recipients (P<0.001). Patients treated with LT-MMI spent 95.8%±5.9% of the 12-year study period in a euthyroid state, whereas this proportion was 72.4%±14.8% in the RAI-treated patients (P<0.001). No major treatment-related adverse events were observed in either group.
Conclusion
In patients with TMNG, LT-MMI therapy is superior to RAI treatment, as shown by the earlier achievement of euthyroidism and the longer duration of sustained normal serum thyrotropin.
INTRODUCTION
Toxic multinodular goiter (TMNG) is a common cause of thyrotoxicosis secondary to Graves’ disease [1]. In this disease, heterogeneous thyroid enlargement, which signifies the presence of autonomously hyperfunctioning thyroid nodules, along with a lack of thyroid autoimmunity, results in a steady hyperthyroid state that does not undergo remission [2]. Therefore, most guidelines have considered ablation therapy an appropriate treatment strategy for TMNG [1,3]. A substantial majority of patients are treated with radioactive iodine (RAI) [4,5], and thyroidectomy is usually considered for nodules that are resistant to other treatment modalities, cases of coexisting thyroid cancer, and large goiters [6].
Antithyroid drugs (ATDs) have been used to achieve euthyroidism before surgery and RAI ablation in older patients [1]. Due to the reported recurrence of hyperthyroidism in 95% of patients upon discontinuation of ATD treatment in TMNG in some studies [7], short-term ATD treatment is not recommended. In fact, due to these concerns, most guidelines have limited the use of ATD therapy to elderly patients with comorbidities, patients considered high-risk for surgery or poor candidates for ablation, and those with decreased life expectancy [3].
Over the last three decades, many studies have demonstrated efficiency and safety of long-term ATD therapy in Graves’ disease [8-12]. We have also shown that long-term low-dose methimazole (MMI) treatment for 60 to 100 months is a safe and effective method for the management of TMNG, and is not inferior to RAI therapy [13].
Cardiovascular safety is an issue of critical importance in the management of hyperthyroidism. Studies focusing on the consequences of a hyperthyroid state on the cardiovascular system have shown increased risks of mortality and morbidity secondary to cardiovascular events in partially treated or untreated hyperthyroid patients [14,15]. Therefore, rapid and complete correction of hyperthyroidism is desired and advised [16]. The objective of the present study was to examine the time to euthyroidism, the time spent in various thyroid states, and cardiovascular risk factors in the setting of long-term methimazole (LT-MMI) therapy in comparison to RAI treatment in patients with TMNG.
METHODS
Trial design
This study explored the extended follow-up findings of participants in a previously reported randomized, parallel-group clinical trial initiated in April 2005 in Tehran, Iran, an area of iodine sufficiency [17]. The clinical trial was registered and approved (IRCT201009224794N1) with an ethics code of 275EC. The research protocol was approved by the ethics committee of the Research Institute for Endocrine Sciences, Shahid Beheshti University of Medical Sciences (IR.SBMU.ENDOCRINE.REC.1400.001) and informed consent was obtained.
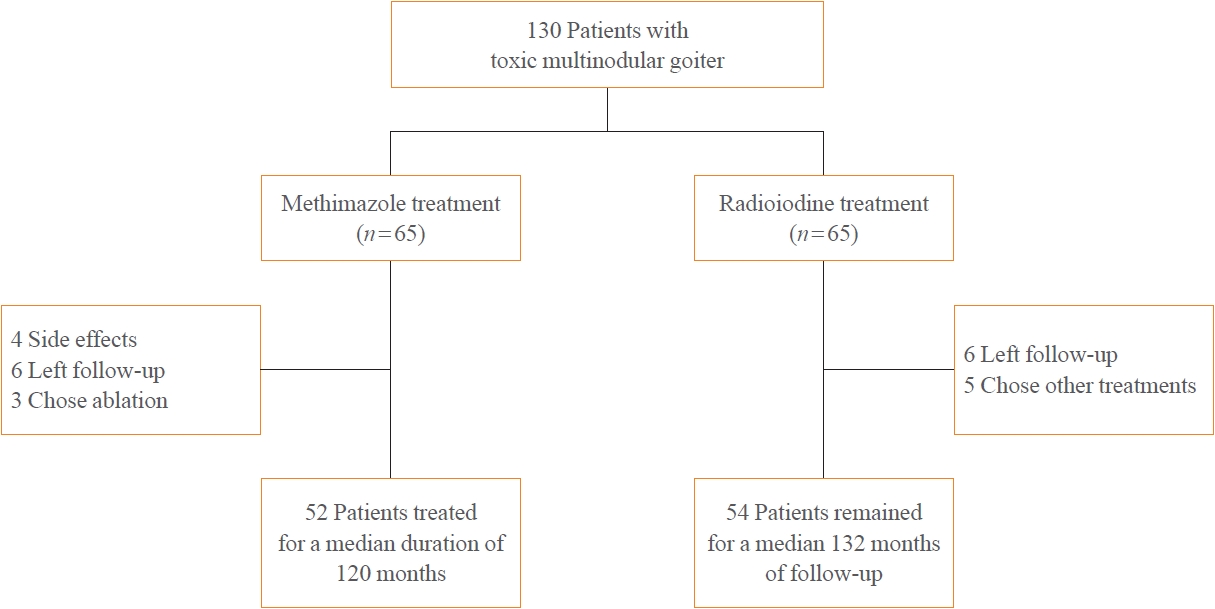
The details of the research design and results of 8-year follow-up have been previously published [13]. Briefly, patients aged ≤60 years with untreated TMNG were randomized at a ratio of 1:1 to undergo RAI or LT-MMI treatment. Upon conclusion of recruitment, 130 patients met the inclusion criteria. Fig. 1 shows the study flow diagram. The diagnosis was made based on documented hyperthyroidism (serum thyroid-stimulating hormone [TSH] <0.4 mIU/L, free thyroxin [fT4] >23 pmol/L, and/or serum triiodothyronine [T3] >200 ng/dL) and palpation, thyroid sonography/thyroid scintigraphy, and/or negative serum TSH receptor antibodies. Patients with cardiovascular disease, cirrhosis, chronic kidney disease, pregnancy, breastfeeding, use of iodine or amiodarone, and altered mental function were excluded.
Procedures
At the first visit after randomization, baseline clinical information was obtained, a physical examination was performed, and the thyroid weight was estimated. Patients were followed every 2 to 3 months in the 1st year and every 6 months thereafter, for a total of 108 to 148 months. In patients with overt hypothyroidism or hyperthyroidism, the intervals between follow-up visits were shortened to 1 to 3 months until euthyroidism was achieved. Serum TSH, fT4, and T3 concentrations were determined at each visit.
Of the 65 patients randomized to the RAI group, five were lost to follow-up and six chose other treatment modalities. Therefore, 54 patients received 50 µCi of I131 per gram of estimated thyroid gland weight divided by 24-hour RAI uptake (as a decimal). Sixty-five other patients received 10 to 20 mg of MMI daily for the 1st month, after which the titration method was applied to maintain serum fT4 and TSH within the normal range. In the MMI group, four patients experienced adverse events related to the medication, three chose to undergo thyroidectomy, and five were lost to follow-up. The remaining 53 patients were treated with LT-MMI. The mean duration of followup was 120 and 132 months for the LT-MMI and RAI groups, respectively.
When subclinical hypothyroidism or hyperthyroidism occurred in the MMI patients, the dose of MMI was adjusted with the target of keeping thyroid function tests within the normal ranges. In the RAI group, the persistence of overt hyperthyroidism for longer than 6 months was treated with additional dose(s) of RAI or LT-MMI treatment (according to the abovementioned protocol). On the contrary, subclinical hypothyroidism or hyperthyroidism was not treated (except for patients aged ≥65 years with subclinical hyperthyroidism). Overt hypothyroidism in both groups was treated with levothyroxine.
Definitions
Hyperthyroidism was defined as TSH <0.4 mIU/L (normal reference range, 0.4 to 5.06) with fT4 >23 pmol/L (normal reference range, 9 to 23) and/or T3 >200 ng/dL (normal reference range, 75 to 200). Subclinical hyperthyroidism was defined as TSH <0.4 mIU/L with serum fT4 and T3 within the reference range. Overt hypothyroidism was considered as TSH above the upper limit of normal (i.e., 5.06 mIU/L for the Tehranian population) with fT4 <9 pmol/L. Subclinical hypothyroidism was defined as TSH >5.06 mIU/L with normal serum fT4 and T3 concentrations [18].
Prediabetes and diabetes were defined as recommended by American Diabetes Association [19]. Hypertension was diagnosed with a confirmed systolic blood pressure >140 mm Hg and/or diastolic blood pressure >90 mm Hg or the use of antihypertensive medications.
Study outcomes
The primary outcome was sustained euthyroidism until the end of the follow-up period. Key secondary outcomes were both overt and subclinical hypothyroidism and hyperthyroidism. For the present study, the time to euthyroidism and percentage of time spent in euthyroid, hypothyroid, and hyperthyroid states were calculated, and information regarding cardiovascular events and death and other comorbidities was obtained from 12 years of follow-up data collected throughout the course of the study. Adverse events such as agranulocytosis, skin reactions, arthralgia, hepatic side effects, and the occurrence of malignancy were also investigated.
Statistical analysis
The sample size for the trial was calculated to detect non-inferiority if the risk was at least 10%, the lower bound of the confidence interval for risk ratio of MMI versus RAI was <1.1, with a type 1 error of 0.05 and a power of at least 0.8, including an attrition rate of 20%. The baseline characteristics of the participants are shown as mean±standard deviation or frequency (%). Significant differences between the two study groups were assessed by the chi-square and Fisher exact tests for categorical variables and the Mann-Whitney test, analysis of variance, and the Student t test for continuous variables. All analyses were conducted on an intention-to-treat basis.
The time to euthyroidism (i.e., swift amelioration of hyperthyroidism) is considered important because prompt restoration of euthyroidism is critical in the prevention of adverse cardiovascular events [17,18]. Therefore, the velocity of decline and normalization of serum fT4 and T3 and the increase in suppressed TSH to normal levels were compared in both study groups. In addition, the time spent in a euthyroid state after achieving it is another critical outcome for the prevention of adverse events. Therefore, the percentage of time that serum TSH, T4, and T3 stayed in normal range during years of follow-up was calculated and compared between the two groups. The time to euthyroidism was calculated from the start of the intervention until the achievement of normal serum fT4, T3, and TSH. The event date for thyroid status cases was described as the midpoint between the date of the follow-up visit at which a particular thyroid status was detected, and the most recent follow-up visit preceding the diagnosis. All analyses were performed using SPSS for Windows version 19 (IBM Corp., Armonk, NY, USA), with a two-tailed P<0.05 being considered significant.
RESULTS
Study patients
At baseline, there were 53 patients in the MMI group and 54 patients in the RAI group, with average ages of 49.5±6.0 and 50.0±5.7 years, respectively. The baseline demographics, clinical characteristics, and laboratory variables of the two groups, including sex, smoking, goiter size, and serum concentrations of fT4, T3, and TSH, did not show statistically significant between-group differences (Supplemental Table S1) [13].
Effect of treatment
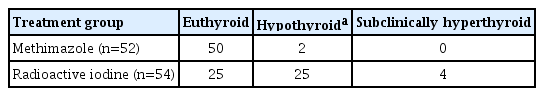
In the MMI group, the mean daily dose of MMI declined from 14±5 to 4.0±1.3 mg by the end of 12 years of MMI therapy. At the end of the study, one patient was lost to follow-up; all 50 patients who completed sustained MMI treatment were euthyroid, and two patients on levothyroxine therapy were also euthyroid (Table 1).
In the RAI group, 12 years after receiving 16.7±2.7 mCi (618±100 MBq) of RAI, 25, 25, and four patients were euthyroid, hypothyroid, and subclinically hyperthyroid, respectively (Table 1). Twelve patients experienced recurrence of hyperthyroidism within the 12 years of follow-up: three preferred continuous LT-MMI therapy, three became hypothyroid after receiving a second dose of RAI, two were subclinically hyperthyroid, and four achieved euthyroidism after additional doses of RAI. One patient received a third dose and another patient was treated with third and fourth doses of RAI. Accordingly, in the RAI group, three subgroups could be recognized: euthyroid (n=25), hypothyroid after the first dose of RAI (n=17), and the subgroup who experienced relapse and required additional rounds of RAI therapy for hyperthyroidism (n=12).
Time to euthyroidism
The mean time to reach a normal serum TSH concentration was 4.3±1.3 months (range, 2 to 15) in the MMI group and 11.4±6.1 months (range, 3 to 24) in the RAI group (P<0.001) (Fig. 2).
Time spent in euthyroidism
Patients treated with MMI remained in a euthyroid state for 95.8%±5.7% of the 12 years of follow-up (range, 78% to 100%); however, in the RAI group, the mean time spent in euthyroidism was 72.4%±14.8% (range, 50% to 100%) (RAI group; P<0.001). The lowest value belonged to those patients who experienced recurrence of hyperthyroidism after the first dose of RAI treatment, who only spent 59%±10% (range, 50% to 75%) of the 12-year follow-up time in euthyroidism (Table 2).
Risk of hyperthyroidism
The time spent in subclinical hyperthyroidism was 3.3%±6.0% (range, 0% to 22%) among patients treated with MMI and 17.4%±12.8% (range, 0% to 50%) in the RAI group (Table 2). This frequency was lowest in those who became hypothyroid and highest in patients who experienced recurrence after RAI treatment. Relapse of overt hyperthyroidism was not observed in patients in the MMI group or patients who attained euthyroidism or hypothyroidism after the first round of RAI therapy. However, patients who experienced relapse of hyperthyroidism after RAI therapy spent 7.5%±6.2% and 40%±23% of the 12-year period in overt and subclinical hyperthyroidism, respectively.
Risk of hypothyroidism
During treatment with MMI, no cases of overt hypothyroidism were seen, and TSH levels above 5.01 mIU/L occurred during 0.9%±0.5% of the treatment time. In the RAI group, the occurrence of subclinical and overt hypothyroidism accounted for 6.0%±9.3% and 2.5%±3.3% of the total duration of 12 years, respectively. Table 2 demonstrates the time spent in various thyroid states in both study groups during 12 years of intervention.
Impact on body weight
The mean weight gain was 1.1±3.1 and 2.7±4.0 kg in the MMI and RAI groups, respectively (P<0.021). In the RAI group, the mean weight gain was 0.4±1.8, 3.3±4.2, and 3.4±4.6 kg in euthyroid, relapsed, and hypothyroid patients after RAI therapy, respectively (P<0.011).
Comorbidities
The prevalence of diabetes, prediabetes, and hypertension was 6%, 15%, and 22% in the MMI group and 8%, 14%, and 25% in the RAI group, respectively. There were no cases of major cardiovascular events, including myocardial infarction, stroke, atrial fibrillation, or death, in either group. Two and four cases of breast cancer occurred in the MMI and RAI groups, respectively.
Thyroid status in the elderly
At the start of the intervention, all patients were less than 61 years of age. At the end of the study, however, 12 and 13 patients were older than 65 years of age in the MMI and RAI groups, respectively. Table 3 shows the characteristics of these patients and compares outcomes between the two study groups. The time to euthyroidism was significantly shorter in MMI-treated patients than in the RAI group (3.3±1.0 months vs. 13.9±8.8 months, P<0.001), and five out of the 12 (42%) spent 100% of the follow-up time in euthyroidism. The other seven patients in this group spent 90% to 95% of the 12-year period in euthyroidism. In the RAI group, only one patient was euthyroid 100% of the time; eight of the 13 patients (61%) spent ≤80% and four (31%) spent ≤58% of the follow-up time in euthyroidism.
Adverse events
In the MMI group, three cases of skin reactions and one case of elevated liver enzymes occurred in the first 3 months of therapy. Mild and transient discomfort occurred in six patients in the RAI group. No other adverse events were reported after fourth month of the study in either group.
DISCUSSION
The present study is the first randomized clinical trial comparing time to euthyroidism and time spent in euthyroidism for 12 years after LT-MMI and RAI treatment in patients with TMNG. The findings show that there were significant differences in both time to euthyroidism and time spent in euthyroidism between the two modes of treatment, and that LT-MMI treatment strategy led to more favorable results.
In contrast to Graves’ disease, the natural course of TMNG involves constant production of excess thyroid hormones without any remission and relapse episodes [2]. Therefore, physicians have accepted ablative therapy, mostly RAI administration, as the choice of therapy for TMNG [3-6]. ATD treatment for more than 6 months is not recommended, and thionamide drugs are mostly used for preparation before ablation [7].
We have previously reported the non-inferiority of LT-MMI treatment in restoring normal thyroid function compared to RAI therapy and suggested that this treatment modality is a valid alternative to ablation in the treatment of patients with TMNG [13]. The present study expands our experience with 12 years of follow-up and demonstrates a more rapid conversion of hyperthyroidism to euthyroidism and significantly more time spent with normal TSH levels during 12 years in LT-MMI-treated patients, as compared to RAI therapy. This is in concordance with findings for Graves’ disease, showing fewer variations in serum TSH levels in long-term ATD patients than in those taking levothyroxine for RAI-induced hypothyroidism [9,11]. In addition, the exacerbation of overt hyperthyroidism during LT-MMI therapy was very rare [13] and no major adverse events occurred after 1st year of treatment [8,13,20].
Studies have shown increased risks of cardiovascular mortality and morbidity in patients with hyperthyroidism and increased all-cause mortality in both patients with Graves’ disease and toxic nodular goiter [21]. The duration of low TSH levels has been associated with a higher risk of adverse cardiovascular outcomes in both treated and untreated patients with hyperthyroidism [14,22]. Increased all-cause mortality has been observed during ATD treatment and after RAI therapy not resulting in hypothyroidism [23]. It has been stated that early and effective control of hyperthyroidism, regardless of the therapeutic modality, was associated with improved survival [15]. Therefore, it may be assumed that the more rapid and complete correction of hyperthyroidism, including normalization of TSH, is advisable [16]. These findings, along with problems with the lifelong levothyroxine treatment of hypothyroidism (described below), challenge the recommended aim of treatment of hyperthyroidism as reaching either euthyroid or hypothyroid status [3].
The comparison of time to euthyroidism and time spent in a euthyroid state between the LT-MMI- and RAI-treated groups in the present study demonstrated the superiority of LT-MMI therapy to RAI ablation because of the potentially lower risk of cardiovascular events. A review of the literature regarding this concept has been previously published [24]. In addition, nearly half of RAI-treated patients developed hypothyroidism requiring life-long levothyroxine therapy. Besides fluctuations in serum TSH concentrations in many patients [24,25], studies have reported impaired psychological well-being, decreased resting energy expenditure, increased body weight, dyslipidemia, and lower T3-to-T4 ratios in levothyroxine-treated patients compared to normal control and euthyroid Graves’ individuals [26-29]. These relevant issues and the findings of the present study may be appropriate in discussions of the trade-off between continuous lifelong MMI versus lifelong levothyroxine before treating patients with TMNG.
A major strength of this study is that we developed the research aim, study design, and execution to specifically address a knowledge gap regarding the comparative effectiveness of LT-MMI and RAI therapies on indices of successful management of hyperthyroidism in patients with TMNG. This issue has not been addressed before; therefore, the findings could assist in decision-making for this important clinical condition.
Several limitations should be considered. First, the findings may not apply to patients with TMNG residing in areas of iodine deficiency. Second, the size of goiter in this study was two to three times the normal thyroid size, and findings may not be similar in patients with very large goiters. Third, because of limitations in the number of visits and TSH testing, the time to euthyroidism and time spent in euthyroidism may not have been captured with perfect accuracy. Fourth, patients in the MMI group with subclinical hypothyroid or hyperthyroid status were treated to attain euthyroidism, while those in the RAI group were only observed unless they were ≥65 years of age. Fifth, due to the limited number of patients, outcomes related to cardiovascular safety and mortality could not be assessed. A welldesigned study with longer duration and appropriate cardiovascular outcomes should be performed. Lastly, this study was not double-blinded, and selection and assignment biases may have existed.
In conclusion, although both LT-MMI and RAI therapies were shown to be effective in inducing euthyroidism in patients with TMNG during 12 years of follow-up, LT-MMI may be superior to RAI due to the rapid conversion from hyperthyroidism to euthyroidism, substantially lower fluctuations in serum TSH levels, and its potentially lower risk of cardiovascular events and mortality. Further larger-scale clinical trials focusing on the outcomes of various treatment approaches in patients with TMNG could shed more light on the morbidity and mortality related to various therapeutic modalities.
Supplementary Material
Supplemental Table S1.
Baseline Characteristics of the Two Study Groupsa
Notes
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
AUTHOR CONTRIBUTIONS
Conception or design: F.A. Acquisition, analysis, or interpretation of data: F.A., H.A., A.A. Drafting the work or revising: F.A., N.S., M.A.T., H.A., L.M., A.A. Final approval of the manuscript: F.A., N.S., M.A.T., H.A., L.M., A.A.