Search
- Page Path
- HOME > Search
Original Articles
- Clinical Study
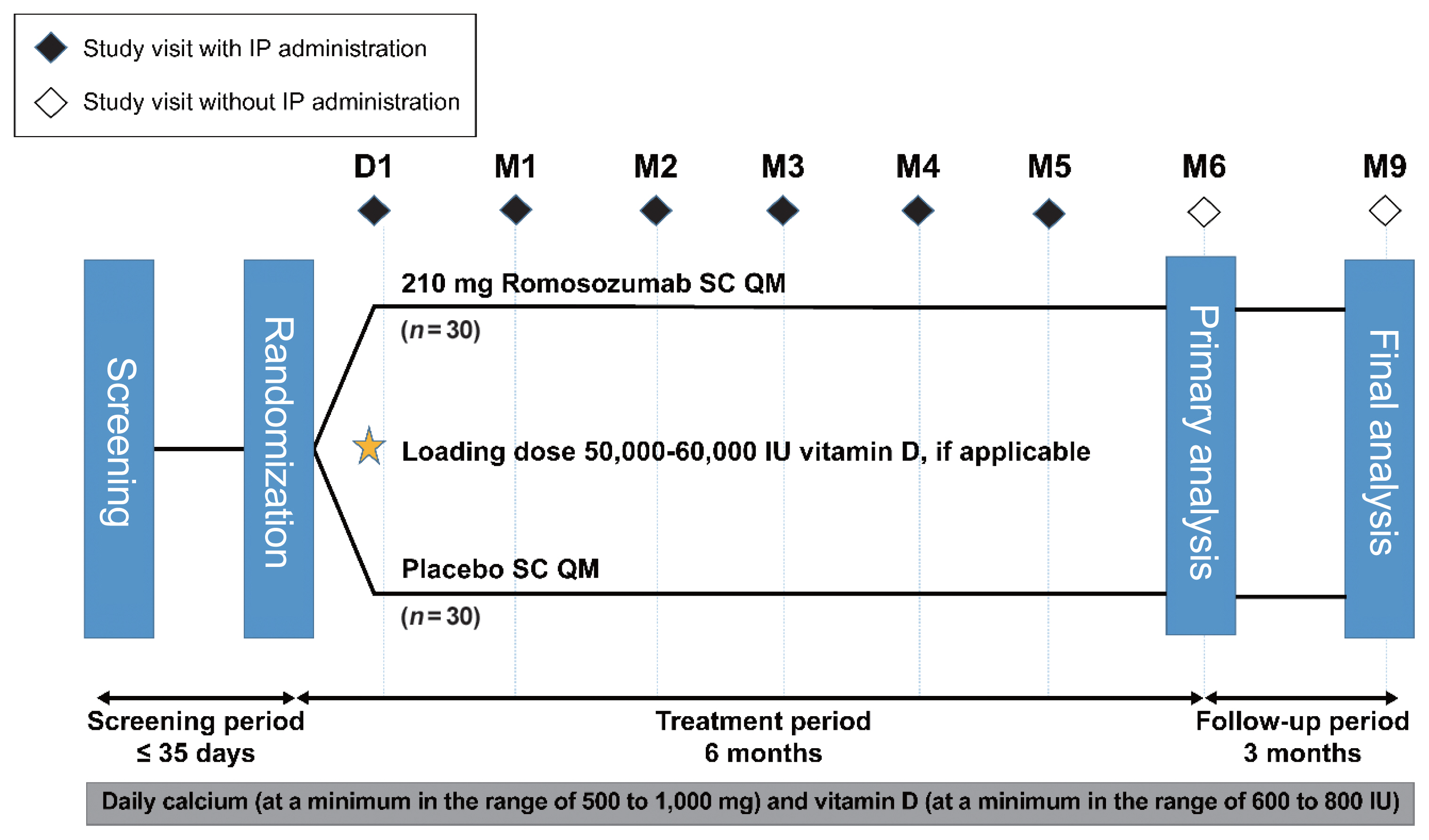
- Romosozumab in Postmenopausal Korean Women with Osteoporosis: A Randomized, Double-Blind, Placebo-Controlled Efficacy and Safety Study
- Ki-Hyun Baek, Yoon-Sok Chung, Jung-Min Koh, In Joo Kim, Kyoung Min Kim, Yong-Ki Min, Ki Deok Park, Rajani Dinavahi, Judy Maddox, Wenjing Yang, Sooa Kim, Sang Jin Lee, Hyungjin Cho, Sung-Kil Lim
- Endocrinol Metab. 2021;36(1):60-69. Published online February 24, 2021
- DOI: https://doi.org/10.3803/EnM.2020.848
- 6,878 View
- 390 Download
- 7 Web of Science
- 10 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
This phase 3 study evaluated the efficacy and safety of 6-month treatment with romosozumab in Korean postmenopausal women with osteoporosis.
Methods
Sixty-seven postmenopausal women with osteoporosis (bone mineral density [BMD] T-scores ≤–2.5 at the lumbar spine, total hip, or femoral neck) were randomized (1:1) to receive monthly subcutaneous injections of romosozumab (210 mg; n=34) or placebo (n=33) for 6 months.
Results
At month 6, the difference in the least square (LS) mean percent change from baseline in lumbar spine BMD (primary efficacy endpoint) between the romosozumab (9.5%) and placebo (–0.1%) groups was significant (9.6%; 95% confidence interval, 7.6 to 11.5; P<0.001). The difference in the LS mean percent change from baseline was also significant for total hip and femoral neck BMD (secondary efficacy endpoints). After treatment with romosozumab, the percent change from baseline in procollagen type 1 N-terminal propeptide transiently increased at months 1 and 3, while that in C-terminal telopeptide of type 1 collagen showed a sustained decrease. No events of cancer, hypocalcemia, injection site reaction, positively adjudicated atypical femoral fracture or osteonecrosis of the jaw, or positively adjudicated serious cardiovascular adverse events were observed. At month 9, 17.6% and 2.9% of patients in the romosozumab group developed binding and neutralizing antibodies, respectively.
Conclusion
Treatment with romosozumab for 6 months was well tolerated and significantly increased lumbar spine, total hip, and femoral neck BMD compared with placebo in Korean postmenopausal women with osteoporosis (ClinicalTrials.gov identifier NCT02791516). -
Citations
Citations to this article as recorded by- A pharmacovigilance analysis of FDA adverse event reporting system events for romosozumab
Zepeng Chen, Ming Li, Shuzhen Li, Yuxi Li, Junyan Wu, Kaifeng Qiu, Xiaoxia Yu, Lin Huang, Guanghui Chen
Expert Opinion on Drug Safety.2023; 22(4): 339. CrossRef - Evaluation of the efficacy and safety of romosozumab (evenity) for the treatment of osteoporotic vertebral compression fracture in postmenopausal women: A systematic review and meta‐analysis of randomized controlled trials (CDM‐J)
Wenbo Huang, Masashi Nagao, Naohiro Yonemoto, Sen Guo, Takeshi Tanigawa, Yuji Nishizaki
Pharmacoepidemiology and Drug Safety.2023; 32(6): 671. CrossRef - Efficacy and Cardiovascular Safety of Romosozumab: A Meta-analysis and Systematic Review
Seo-Yong Choi, Jeong-Min Kim, Sang-Hyeon Oh, Seunghyun Cheon, Jee-Eun Chung
Korean Journal of Clinical Pharmacy.2023; 33(2): 128. CrossRef - Clinical Studies On Romosozumab: An Alternative For Individuals With A High Risk Of Osteoporotic Fractures: A Current Concepts Review (Part I)
E. Carlos Rodriguez-Merchan, Alonso Moreno-Garcia, Hortensia De la Corte-Rodriguez
SurgiColl.2023;[Epub] CrossRef - Romosozumab in osteoporosis: yesterday, today and tomorrow
Dong Wu, Lei Li, Zhun Wen, Guangbin Wang
Journal of Translational Medicine.2023;[Epub] CrossRef - Efficacy and safety of anti-sclerostin antibodies in the treatment of osteoporosis: A meta-analysis and systematic review
Frideriki Poutoglidou, Efthimios Samoladas, Nikolaos Raikos, Dimitrios Kouvelas
Journal of Clinical Densitometry.2022; 25(3): 401. CrossRef - Benefits of lumican on human bone health: clinical evidence using bone marrow aspirates
Yun Sun Lee, So Jeong Park, Jin Young Lee, Eunah Choi, Beom-Jun Kim
The Korean Journal of Internal Medicine.2022; 37(4): 821. CrossRef - What is the risk of cardiovascular events in osteoporotic patients treated with romosozumab?
I. R. Reid
Expert Opinion on Drug Safety.2022; 21(12): 1441. CrossRef - Proxied Therapeutic Inhibition on Wnt Signaling Antagonists and Risk of Cardiovascular Diseases: Multi-Omics Analyses
Yu Qian, Cheng-Da Yuan, Saber Khederzadeh, Ming-Yu Han, Hai-Xia Liu, Mo-Chang Qiu, Jian-Hua Gao, Wei-Lin Wang, Yun-Piao Hou, Guo-Bo Chen, Ke-Qi Liu, Lin Xu, David Karasik, Shu-Yang Xie, Hou-Feng Zheng
SSRN Electronic Journal .2022;[Epub] CrossRef - Multi-Omics Analyses Identify Pleiotropy and Causality Between Circulating Sclerostin and Atrial Fibrillation
Yu Qian, Peng-Lin Guan, Saber Khederzadeh, Ke-Qi Liu, Cheng-Da Yuan, Ming-Yu Han, Hai-Xia Liu, Mo-Chang Qiu, Jian-Hua Gao, Wei-Lin Wang, Yun-Piao Hou, Guo-Bo Chen, Lin Xu, David Karasik, Shu-Yang Xie, sheng zhifeng, Hou-Feng Zheng
SSRN Electronic Journal .2022;[Epub] CrossRef
- A pharmacovigilance analysis of FDA adverse event reporting system events for romosozumab

- The Changes of Cytokines and Bone Turnover Markers after Bone Marrow Transplantation.
- Won Young Lee, Moo Il Kang, Ki Won Oh, Hye Soo Kim, Seong Dae Mun, Je Ho Han, Hyun Shik Son, Sung Koo Kang, Wan Sik Shin, Woo Sung Min, Choon Choo Kim
- J Korean Endocr Soc. 2000;15(1):85-96. Published online January 1, 2001
- 987 View
- 18 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Loss of bone mass is usually detected after BMT. The causes of bone loss are related with gonadal dysfunction and immunosuppressants. Cytokines, especially IL-6, play an important role in the pathogenesis of postmenopausal osteoporosis. However, the pathogenetic role of cytokines in post-BMT bone loss is unknown and data on the changes of cytokines in accordance with bone turnover markers are scarce. The aim of this study is to assess the relationship of bone turnover markers and cytokines of peripheral blood and bone marrow before and after allogeneic BMT. METHODS: This prospective study included two analyses. The first was a study of 46 BMT recipients, examining the relationship between bone turnover markers and cytokines of serum which were measured before and 1, 2, 3, 4 week and 3 months after BMT. The second was a study of 14 BMT patients, measuring bone marrow plasma cytokines such as IL-6 and TNF-alpha at post-BMT 3 week and bone turnover marker at the same time to assess the relationship between two parameters. RESULTS: Serum ICTP, bone resorption marker, increased progressively until 4 weeks (peak) after BMT and then decreased thereafter. Serum osteocalcin, bone formation marker, decreased progressively until 3 weeks after BMT and then increased thereafter. There was positive correlation between serum ICTP and bone marrow IL-6 levels at the post-BMT 3 week with a statistical significance, but the correlation between bone turnover markers and bone marrow TNF-alpha or peripheral blood cytokines was not found. CONCLUSION: Our data suggest that the progressive increase of bone resorption after BMT is related with the increase of bone marrow IL-6, which is a potent stimulator of bone resorption in vivo.

- Bone Turnover and Bone Mineral Density in Acromegaly.
- Sun Wook Kim, Hee Jin Kim, Seon Hwa Lee, Won Bae Kim, Do Joon Park, Chan Soo Shin, Kyong Soo Park, Seong Yeon Kim, Bo Youn Cho, Hong Kyu Lee
- J Korean Endocr Soc. 1999;14(4):688-697. Published online January 1, 2001
- 1,017 View
- 20 Download
-
 Abstract
Abstract
 PDF
PDF - BACKGROUND
Abnormalities of calcium homeostasis and bone remodelling were known in acromegaly, but controversy concerning the influence of chronically elevated serum growth hormone (GH)/insulin like growth factor-I (IGF-I) concentrations on bone metabolism has been existed. In this study, we assess the effect of chronically elevated serum GH/IGF-I levels on bone metabolism and bone mineral density (BMD) in patients with acromegaly and compare the markers of bone metabolism and BMD of active acromegaly according to gonadal function. METHODS: We measured biochemical markers of bone turnover and BMD in 50 acromegalic patients (41 active disease, 9 inactive disease) and 41 control subjects. RESULTS: Active acromegalic patients had significantly higher serum oteocalcin levels (13.8+/-7.7 versus 6.8+/-4.7, 6.0+/--3.4 ng/mL, p<0.05) and urinary type I cross-linked N-telopeptide (101.7+/-64.2 versus 49.3+/-33.3, 56.1+/-39.4 nM BCM/mM Cr, p<0.05) compared with inactive acromegaly and control subjects. Serum IGF-I levels correlated positively with serum osteocalcin levels(r=0.69, p<0.05) and urinary type I cross-linked N-telopeptide (r=0.44, p<0.05). In the female patients with active acromegaly, BMD (T-score) was elevated in the femoral neck(1.00+/-0.14 versus 0.89+/-0.12,p<0.05) and trochanter (0.88+/-0.15 versus 0.77+/-0.11, p<0.05), whereas BMD of lumbar spine(1.13+/- 0.17 versus 1.17+/-0.17, p>0.05) and femoral Ward's triangle (0.78+/-0.16 versus 0.77+0.13, p>0.05) were not different from those of control subjects. In the patients with active acromegaly, serum osteocalcin levels (16.4+/-8.8 versus 14.9+/-10.1 ng/mL, p>0.05) as well as urinary type I cross-linked N-telopeptide (104.8+/-68.1 versus 122.0+/-80.3 nM BCM/mM Cr, p>0.05) were not different according to gonadal function. Also, femoral and spinal BMD were not different according to the gonadal function. CONCLUSION: GH/IGF-I excess increase bone turnover and might achieve a positive bone balance at each remodelling cycle. Markers of bone turnover and BMD of skeletal bone were not influenced by gonadal function in the patients with active acromegaly.


 KES
KES

 First
First Prev
Prev



