Search
- Page Path
- HOME > Search
- Hypothalamus and pituitary gland
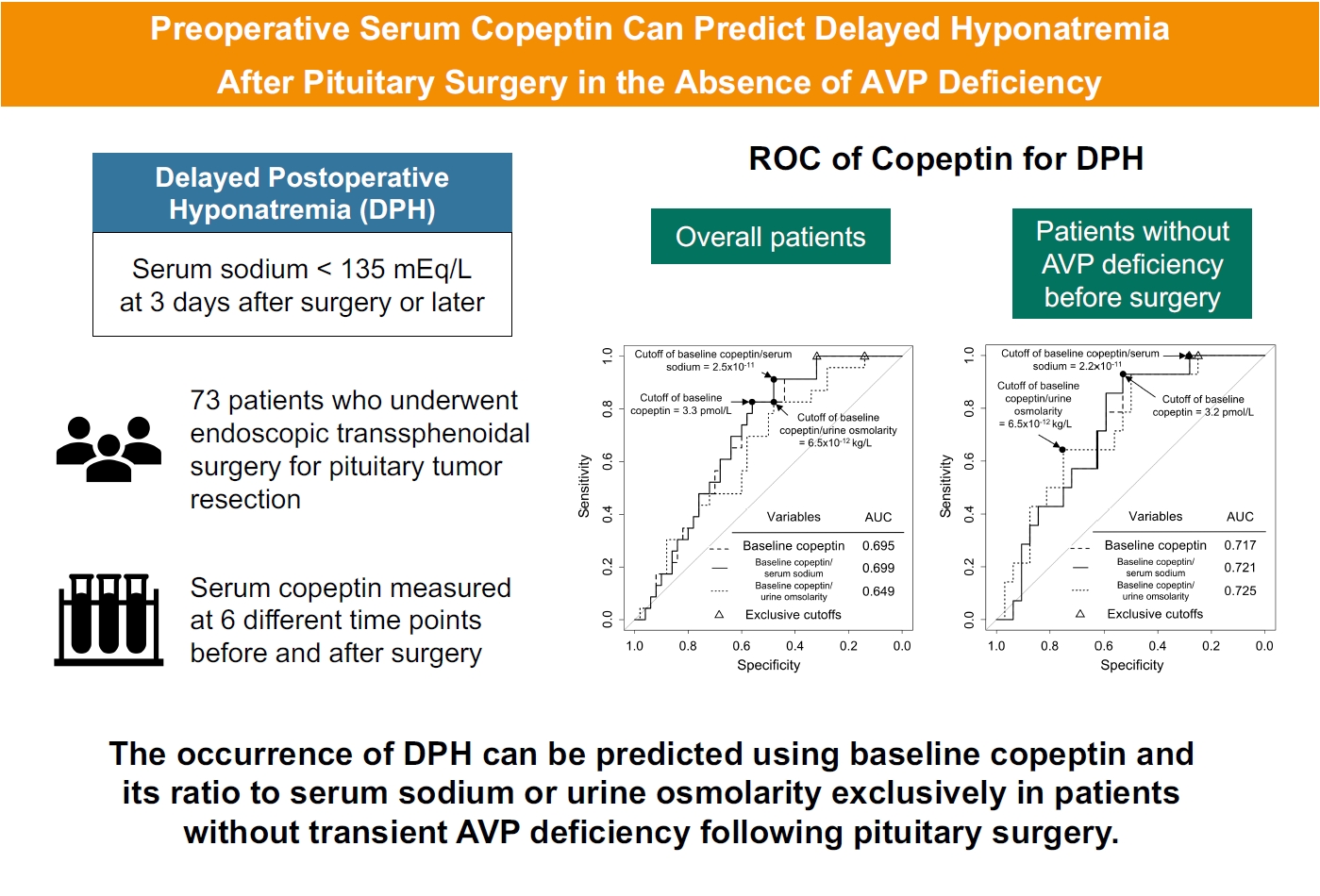
- Preoperative Serum Copeptin Can Predict Delayed Hyponatremia after Pituitary Surgery in the Absence of Arginine Vasopressin Deficiency
- Ho Kang, Seung Shin Park, Yoo Hyung Kim, Hwan Sub Lim, Mi-Kyeong Lee, Kyoung-Ryul Lee, Jung Hee Kim, Yong Hwy Kim
- Endocrinol Metab. 2024;39(1):164-175. Published online January 3, 2024
- DOI: https://doi.org/10.3803/EnM.2023.1792
- 1,035 View
- 49 Download
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Delayed postoperative hyponatremia (DPH) is the most common cause of readmission after pituitary surgery. In this study, we aimed to evaluate the cutoff values of serum copeptin and determine the optimal timing for copeptin measurement for the prediction of the occurrence of DPH in patients who undergo endoscopic transsphenoidal approach (eTSA) surgery and tumor resection.
Methods
This was a prospective observational study of 73 patients who underwent eTSA surgery for pituitary or stalk lesions. Copeptin levels were measured before surgery, 1 hour after extubation, and on postoperative days 1, 2, 7, and 90.
Results
Among 73 patients, 23 patients (31.5%) developed DPH. The baseline ratio of copeptin to serum sodium level showed the highest predictive performance (area under the curve [AUROC], 0.699), and its optimal cutoff to maximize Youden’s index was 2.5×10–11, with a sensitivity of 91.3% and negative predictive value of 92.0%. No significant predictors were identified for patients with transient arginine vasopressin (AVP) deficiency. However, for patients without transient AVP deficiency, the copeptin-to-urine osmolarity ratio at baseline demonstrated the highest predictive performance (AUROC, 0.725). An optimal cutoff of 6.5×10–12 maximized Youden’s index, with a sensitivity of 92.9% and a negative predictive value of 94.1%.
Conclusion
The occurrence of DPH can be predicted using baseline copeptin and its ratio with serum sodium or urine osmolarity only in patients without transient AVP deficiency after pituitary surgery.

- Hypothalamus and Pituitary Gland
- Advances in Pituitary Surgery
- Yoon Hwan Byun, Ho Kang, Yong Hwy Kim
- Endocrinol Metab. 2022;37(4):608-616. Published online August 19, 2022
- DOI: https://doi.org/10.3803/EnM.2022.1546
- 3,220 View
- 154 Download
- 2 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Pituitary surgery has advanced considerably in recent years with the exploration and development of various endoscopic approaches and techniques. Different endoscopic skull base approaches are being applied to access sellar tumors in different locations. Moreover, extracapsular dissection and cavernous sinus exploration have enabled gross total resection of sellar tumors where it could not have been achieved in the past. Techniques for skull base reconstruction have also progressed, allowing surgeons to remove larger and more complicated tumors than before. This review article discusses different endoscopic skull base approaches, surgical techniques for removing pituitary adenomas, and reconstruction methods for repairing postoperative low-flow and high-flow cerebrospinal fluid leakage.
-
Citations
Citations to this article as recorded by- Serum and hair steroid profiles in patients with nonfunctioning pituitary adenoma undergoing surgery: A prospective observational study
Seung Shin Park, Yong Hwy Kim, Ho Kang, Chang Ho Ahn, Dong Jun Byun, Man Ho Choi, Jung Hee Kim
The Journal of Steroid Biochemistry and Molecular Biology.2023; 230: 106276. CrossRef - Pituitary disease and anaesthesia
Kim Rhodes, Robert John, Astri Luoma
Anaesthesia & Intensive Care Medicine.2023; 24(6): 348. CrossRef - Орфанні ендокринні захворювання: сучасні тенденції хірургічного лікування в Україні
M.D. Tronko, B.B. Guda
Endokrynologia.2022; 27(4): 287. CrossRef
- Serum and hair steroid profiles in patients with nonfunctioning pituitary adenoma undergoing surgery: A prospective observational study

- Miscellaneous
- COVID-19 Vaccination for Endocrine Patients: A Position Statement from the Korean Endocrine Society
- Cheol Ryong Ku, Kyong Yeun Jung, Chang Ho Ahn, Jun Sung Moon, Ju Hee Lee, Eun Heui Kim, Hyemi Kwon, Hee Kyung Kim, Sunghwan Suh, Sangmo Hong, Jeonghoon Ha, Eun Roh, Jin Hwa Kim, Mi-kyung Kim, the Committee of Clinical Practice Guideline of the Korean Endocrine Society
- Endocrinol Metab. 2021;36(4):757-765. Published online August 17, 2021
- DOI: https://doi.org/10.3803/EnM.2021.404
- 10,360 View
- 419 Download
- 19 Web of Science
- 21 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Since the first outbreak of coronavirus disease 2019 (COVID-19), ongoing efforts have been made to discover an efficacious vaccine against COVID-19 to combat the pandemic. In most countries, both mRNA and DNA vaccines have been administered, and their side effects have also been reported. The clinical course of COVID-19 and the effects of vaccination against COVID-19 are both influenced by patients’ health status and involve a systemic physiological response. In view of the systemic function of endocrine hormones, endocrine disorders themselves and the therapeutics used to treat them can influence the outcomes of vaccination for COVID-19. However, there are very limited data to support the development of clinical guidelines for patients with specific medical backgrounds based on large clinical trials. In the current severe circumstances of the COVID-19 pandemic, position statements made by clinical specialists are essential to provide appropriate recommendations based on both medical evidence and clinical experiences. As endocrinologists, we would like to present the medical background of COVID-19 vaccination, as well as precautions to prevent the side effects of COVID-19 vaccination in patients with specific endocrine disorders, including adrenal insufficiency, diabetes mellitus, osteoporosis, autoimmune thyroid disease, hypogonadism, and pituitary disorders.
-
Citations
Citations to this article as recorded by- COVID-19 mRNA vaccine may trigger subacute thyroiditis
Mehmet Sözen, Ömercan Topaloğlu, Berrin Çetinarslan, Alev Selek, Zeynep Cantürk, Emre Gezer, Damla Köksalan, Taner Bayraktaroğlu
Human Vaccines & Immunotherapeutics.2024; 17(12): 5120. CrossRef - The role of co-morbidities in the development of an AEFI after COVID-19 vaccination in a large prospective cohort with patient-reported outcomes in the Netherlands
C. Ouaddouh, J.W. Duijster, T. Lieber, F.P.A.M. van Hunsel
Expert Opinion on Drug Safety.2024; 23(3): 323. CrossRef - Thyroid dysfunction in COVID-19
David Tak Wai Lui, Chi Ho Lee, Yu Cho Woo, Ivan Fan Ngai Hung, Karen Siu Ling Lam
Nature Reviews Endocrinology.2024;[Epub] CrossRef - Adult-Onset Type 1 Diabetes Development Following COVID-19 mRNA Vaccination
Hyeyeon Moon, Sunghwan Suh, Mi Kyoung Park
Journal of Korean Medical Science.2023;[Epub] CrossRef - Prior immunization status of COVID-19 patients and disease severity: A multicenter retrospective cohort study assessing the different types of immunity
Javaria Aslam, Faisal Shahzad Khan, Muhammad Talha Haris, Hewad Hewadmal, Maryam Khalid, Mohammad Y. Alshahrani, Qurrat-ul-ain Aslam, Irrum Aneela, Urooj Zafar
Vaccine.2023; 41(2): 598. CrossRef - Mortality and Severity of Coronavirus Disease 2019 in Patients with Long-Term Glucocorticoid Therapy: A Korean Nationwide Cohort Study
Eu Jeong Ku, Keeho Song, Kyoung Min Kim, Gi Hyeon Seo, Soon Jib Yoo
Endocrinology and Metabolism.2023; 38(2): 253. CrossRef - Pituitary Diseases and COVID-19 Outcomes in South Korea: A Nationwide Cohort Study
Jeonghoon Ha, Kyoung Min Kim, Dong-Jun Lim, Keeho Song, Gi Hyeon Seo
Journal of Clinical Medicine.2023; 12(14): 4799. CrossRef - Inactivated SARS-CoV-2 vaccination does not disturb the clinical course of Graves’ disease: An observational cohort study
Shichen Xu, Huixin Yu, Xian Cheng, Jing Wu, Jiandong Bao, Li Zhang
Vaccine.2023; 41(38): 5648. CrossRef - Adrenal Crisis Associated With COVID-19 Vaccination in Patients With Adrenal Insufficiency
Yukako Kurematsu, Takako Mohri, Sadanori Okada, Yutaka Takahashi
JCEM Case Reports.2023;[Epub] CrossRef - Adverse Events Associated with COVID-19 Vaccination in Adolescents with Endocrinological Disorders: A Cross-Sectional Study
İbrahim Mert Erbaş, İrem Ceren Erbaş, Gözde Akın Kağızmanlı, Kübra Yüksek Acinikli, Özge Besci, Korcan Demir, Ece Böber, Nurşen Belet, Ayhan Abacı
Journal of Clinical Research in Pediatric Endocrinology.2023; 15(3): 248. CrossRef - Neue Aspekte der Glukokortikoidsubstitution bei Nebennierenrindeninsuffizienz
Tina Kienitz, Gesine Meyer
Der Internist.2022; 63(1): 12. CrossRef - Endocrine Follow-up During Post-Acute COVID-19: Practical Recommendations Based on Available Clinical Evidence
Rimesh Pal, Ameya Joshi, Sanjay K. Bhadada, Mainak Banerjee, Suresh Vaikkakara, Satinath Mukhopadhyay
Endocrine Practice.2022; 28(4): 425. CrossRef - Safety of Inactivated and mRNA COVID-19 Vaccination Among Patients Treated for Hypothyroidism: A Population-Based Cohort Study
Xi Xiong, Carlos King Ho Wong, Ivan Chi Ho Au, Francisco Tsz Tsun Lai, Xue Li, Eric Yuk Fai Wan, Celine Sze Ling Chui, Esther Wai Yin Chan, Franco Wing Tak Cheng, Kristy Tsz Kwan Lau, Chi Ho Lee, Yu Cho Woo, David Tak Wai Lui, Ian Chi Kei Wong
Thyroid.2022; 32(5): 505. CrossRef - The New Entity of Subacute Thyroiditis amid the COVID-19 Pandemic: From Infection to Vaccine
Mihaela Popescu, Adina Ghemigian, Corina Maria Vasile, Andrei Costache, Mara Carsote, Alice Elena Ghenea
Diagnostics.2022; 12(4): 960. CrossRef - Adrenal Crisis Secondary to COVID-19 Vaccination in a Patient With Hypopituitarism
Nikolina Markovic, Anila Faizan, Chirag Boradia, Sridhar Nambi
AACE Clinical Case Reports.2022; 8(4): 171. CrossRef - The Effect of Inactivated SARS-CoV-2 Vaccines on TRAB in Graves’ Disease
LingHong Huang, ZhengRong Jiang, JingXiong Zhou, YuPing Chen, HuiBin Huang
Frontiers in Endocrinology.2022;[Epub] CrossRef - Osteoporosis in Patients With Respiratory Diseases
Yue Ma, Shui Qiu, Renyi Zhou
Frontiers in Physiology.2022;[Epub] CrossRef - Pilot Findings on SARS-CoV-2 Vaccine-Induced Pituitary Diseases: A Mini Review from Diagnosis to Pathophysiology
Ach Taieb, El Euch Mounira
Vaccines.2022; 10(12): 2004. CrossRef - Forty Years Together, New Leap Forward! The 40th Anniversary of the Korean Endocrine Society
Jong Chul Won, Ki-Hyun Baek
Endocrinology and Metabolism.2022; 37(6): 851. CrossRef - No need of glucocorticoid dose adjustment in patients with adrenal insufficiency before COVID-19 vaccine
Tania Pilli, Cristina Dalmiglio, Gilda Dalmazio, Alfonso Sagnella, Raffaella Forleo, Lucia Brilli, Fabio Maino, Cristina Ciuoli, Maria Grazia Castagna
European Journal of Endocrinology.2022; 187(1): K7. CrossRef - Diabetes and COVID-19 Vaccination
Hae Dong Choi, Jun Sung Moon
The Journal of Korean Diabetes.2021; 22(4): 221. CrossRef
- COVID-19 mRNA vaccine may trigger subacute thyroiditis

- Hypothalamus and Pituitary Gland
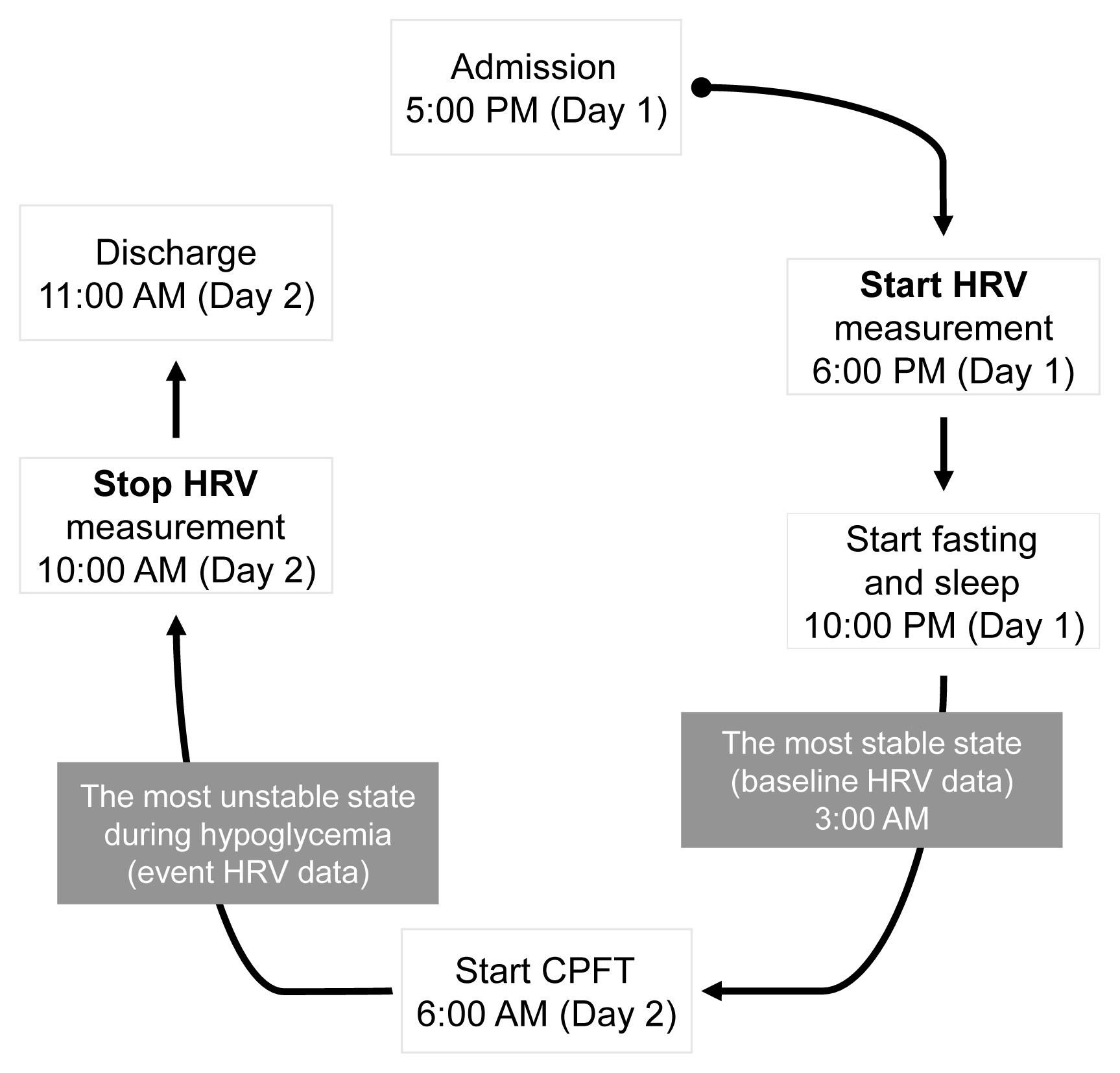
- Heart Rate Variability in Postoperative Patients with Nonfunctioning Pituitary Adenoma
- Jeonghoon Ha, Hansang Baek, Chaiho Jeong, Minsoo Yeo, Seung-Hwan Lee, Jae Hyoung Cho, Ki-Hyun Baek, Moo Il Kang, Dong-Jun Lim
- Endocrinol Metab. 2021;36(3):678-687. Published online June 10, 2021
- DOI: https://doi.org/10.3803/EnM.2021.978
- 4,549 View
- 109 Download
- 3 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Decreased heart rate variability (HRV) has been reported to be associated with cardiac autonomic dysfunction. Hypopituitarism in nonfunctioning pituitary adenoma (NFPA) is often linked to increased cardiovascular mortality. We therefore hypothesized that postoperative NFPA patients with hormone deficiency have an elevated risk of HRV alterations indicating cardiac autonomic dysfunction.
Methods
A total of 22 patients with NFPA were enrolled in the study. Between 3 and 6 months after surgery, a combined pituitary function test (CPFT) was performed, and HRV was measured. The period of sleep before the CPFT was deemed the most stable period, and the hypoglycemic period that occurred during the CPFT was defined as the most unstable period. Changes in HRV parameters in stable and unstable periods were observed and compared depending on the status of hormone deficiencies.
Results
In patients with adrenocorticotropic hormone (ACTH) deficiency with other pituitary hormone deficiencies, the low frequency to high frequency ratio, which represents overall autonomic function and is increased in the disease state, was higher (P=0.005). Additionally, the standard deviation of the normal-to-normal interval, which decreases in the autonomic dysfunction state, was lower (P=0.030) during the hypoglycemic period. In panhypopituitarism, the low frequency to high frequency ratio during the hypoglycemic period was increased (P=0.007).
Conclusion
HRV analysis during CPFT enables estimation of cardiac autonomic dysfunction in patients with NFPA who develop ACTH deficiency with other pituitary hormone deficiencies or panhypopituitarism after surgery. These patients may require a preemptive assessment of cardiovascular risk. -
Citations
Citations to this article as recorded by- Heart Rate Variability in Subjects with Severe Allergic Background Undergoing COVID-19 Vaccination
Maria Bernadette Cilona, Filippo D’Amico, Chiara Asperti, Giuseppe Alvise Ramirez, Stefano Turi, Giovanni Benanti, Shai Marc Bohane, Serena Nannipieri, Rosa Labanca, Matteo Gervasini, Federica Russetti, Naomi Viapiana, Martina Lezzi, Giovanni Landoni, Lor
Vaccines.2023; 11(3): 567. CrossRef - Pituitary Diseases and COVID-19 Outcomes in South Korea: A Nationwide Cohort Study
Jeonghoon Ha, Kyoung Min Kim, Dong-Jun Lim, Keeho Song, Gi Hyeon Seo
Journal of Clinical Medicine.2023; 12(14): 4799. CrossRef - Effect of a 16-Session Qigong Program in Non-Hodgkin Lymphoma Survivors: A Randomized Clinical Trial
Keyla Vargas-Román, Emilia I. De la Fuente-Solana, Jonathan Cortés-Martín, Juan Carlos Sánchez-García, Christian J. González-Vargas, Lourdes Díaz-Rodríguez
Journal of Clinical Medicine.2022; 11(12): 3421. CrossRef
- Heart Rate Variability in Subjects with Severe Allergic Background Undergoing COVID-19 Vaccination

- Hypothalamus and Pituitary gland
- Best Achievements in Pituitary and Adrenal Diseases in 2020
- Chang Ho Ahn, Jung Hee Kim
- Endocrinol Metab. 2021;36(1):51-56. Published online February 24, 2021
- DOI: https://doi.org/10.3803/EnM.2021.108

- 4,497 View
- 162 Download
- 2 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Significant progress in pituitary and adrenal diseases was made in 2020. This review presents major translational and clinical advances in research on pituitary and adrenal diseases, encompassing their epidemiology, pathogenesis, diagnosis, and clinical management. We address the controversy regarding the nomenclature of pituitary neuroendocrine tumors, omics-based molecular classification of pituitary adenomas, and novel drugs for Cushing’s disease in the field of pituitary diseases. In the field of adrenal diseases, we cover big data-driven epidemiology of adrenal tumors, steroid profiling as a new diagnostic tool, and the utility of scoring systems in the decision-making process of managing primary aldosteronism. This brief article will broaden readers’ understanding of pituitary and adrenal diseases.
-
Citations
Citations to this article as recorded by- Surgical management and outcomes of spinal metastasis of malignant adrenal tumor: A retrospective study of six cases and literature review
Xiangzhi Ni, Jing Wang, Jiashi Cao, Kun Zhang, Shuming Hou, Xing Huang, Yuanjin Song, Xin Gao, Jianru Xiao, Tielong Liu
Frontiers in Oncology.2023;[Epub] CrossRef - 2023 Korean Endocrine Society Consensus Guidelines for the Diagnosis and Management of Primary Aldosteronism
Jeonghoon Ha, Jung Hwan Park, Kyoung Jin Kim, Jung Hee Kim, Kyong Yeun Jung, Jeongmin Lee, Jong Han Choi, Seung Hun Lee, Namki Hong, Jung Soo Lim, Byung Kwan Park, Jung-Han Kim, Kyeong Cheon Jung, Jooyoung Cho, Mi-kyung Kim, Choon Hee Chung
Endocrinology and Metabolism.2023; 38(6): 597. CrossRef
- Surgical management and outcomes of spinal metastasis of malignant adrenal tumor: A retrospective study of six cases and literature review

- Hypothalamus and Pituitary gland
- Epidemiology of Functioning Pituitary Adenomas
- Sang Ouk Chin
- Endocrinol Metab. 2020;35(2):237-242. Published online June 24, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.2.237
- 7,878 View
- 255 Download
- 11 Web of Science
- 12 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Pituitary adenomas (PAs) are defined as benign monoclonal tumors in the pituitary gland that cause symptoms due to either hormonal hypersecretion or a space-occupying effect, and are classified as functioning or non-functioning. Because of their rarity and slow-growing with symptomless nature in most cases, it has been challenging to investigate the epidemiology of PAs. Considering their public health impact and association with increased morbidity and mortality, however, it is essential to understand the prevalence and incidence of PAs in order to improve patient outcomes and to minimize the resultant burden on the health care system. Fortunately, developments in imaging modalities and easier access to large-scale population data have enabled investigators to analyze the epidemiology of PAs more accurately. This review summarizes previously reported epidemiologic data on functioning PAs in Korea and other countries.
-
Citations
Citations to this article as recorded by- Spherical type of amyloidogenic pituitary prolactinoma in a 50 year old male
Madhala Divya, Balasubramanian Archana, Lawrence D Cruze, D. Balasubramanian
Interdisciplinary Neurosurgery.2024; 36: 101957. CrossRef - Transcriptome of GH-producing pituitary neuroendocrine tumours and models are significantly affected by somatostatin analogues
Rihards Saksis, Olesja Rogoza, Helvijs Niedra, Kaspars Megnis, Ilona Mandrika, Inga Balcere, Liva Steina, Janis Stukens, Austra Breiksa, Jurijs Nazarovs, Jelizaveta Sokolovska, Ilze Konrade, Raitis Peculis, Vita Rovite
Cancer Cell International.2023;[Epub] CrossRef - Prognostic Models in Growth-Hormone- and Prolactin-Secreting Pituitary Neuroendocrine Tumors: A Systematic Review
Roxana-Ioana Dumitriu-Stan, Iulia-Florentina Burcea, Teodor Salmen, Catalina Poiana
Diagnostics.2023; 13(12): 2118. CrossRef - Salivary microbiome profiles for different clinical phenotypes of pituitary adenomas by single-molecular long-read sequencing
Xuefei Ji, Pingping Li, Qinglong Guo, Liao Guan, Peng Gao, Bingshan Wu, Hongwei Cheng, Jin Xiao, Lei Ye, Justin R. Kaspar
Microbiology Spectrum.2023;[Epub] CrossRef - Endoscopic endonasal surgical management of giant pituitary adenomas with extension into ventricle system
Mykola O. Guk, Oleksii V. Ukrainets
Ukrainian Neurosurgical Journal.2023; 29(4): 13. CrossRef - Uso de resonancia magnética nuclear intraoperatoria en la resección transesfenoidal de adenomas hipofisiarios: ¿qué resultados se han obtenido?
María Laura Boschetti Saer, Levino Roberto Boschetti, Jose Pastor Linarez Veloz, Michael Ortega-Sierra
Archivos de Neurociencias.2023;[Epub] CrossRef - Epidemiology of common and uncommon adult pituitary tumors in the U.S. according to the 2017 World Health Organization classification
Luz E. Castellanos, Catherine Gutierrez, Timothy Smith, Edward R. Laws, J. Bryan Iorgulescu
Pituitary.2022; 25(1): 201. CrossRef - The kinome, cyclins and cyclin-dependent kinases of pituitary adenomas, a look into the gene expression profile among tumors from different lineages
Keiko Taniguchi-Ponciano, Lesly A. Portocarrero-Ortiz, Gerardo Guinto, Sergio Moreno-Jimenez, Erick Gomez-Apo, Laura Chavez-Macias, Eduardo Peña-Martínez, Gloria Silva-Román, Sandra Vela-Patiño, Jesús Ordoñez-García, Sergio Andonegui-Elguera, Aldo Ferreir
BMC Medical Genomics.2022;[Epub] CrossRef - IL-10 Gene Rs1800871, Rs1800872, and Rs1800896 Polymorphisms and IL-10 Serum Levels Association with Pituitary Adenoma
Migle Palivonaite, Greta Gedvilaite, Brigita Glebauskiene, Loresa Kriauciuniene, Vita Rovite, Rasa Liutkeviciene
Biomedicines.2022; 10(8): 1921. CrossRef - Genome wide analysis of circulating miRNAs in growth hormone secreting pituitary neuroendocrine tumor patients’ plasma
Helvijs Niedra, Raitis Peculis, Helena Daiga Litvina, Kaspars Megnis, Ilona Mandrika, Inga Balcere, Mihails Romanovs, Liva Steina, Janis Stukens, Austra Breiksa, Jurijs Nazarovs, Jelizaveta Sokolovska, Rasa Liutkeviciene, Alvita Vilkevicute, Ilze Konrade,
Frontiers in Oncology.2022;[Epub] CrossRef - Identification of mutant K-RAS in pituitary macroadenoma
Veronica Aran, Manoela Heringer, Paulo Jose da Mata, Leandro Kasuki, Renan Lyra Miranda, Felipe Andreiuolo, Leila Chimelli, Paulo Niemeyer Filho, Monica Roberto Gadelha, Vivaldo Moura Neto
Pituitary.2021; 24(5): 746. CrossRef - Surgery is a safe, effective first-line treatment modality for noninvasive prolactinomas
Ji Yong Park, Wonsuk Choi, A Ram Hong, Jee Hee Yoon, Hee Kyung Kim, Woo-Youl Jang, Shin Jung, Ho-Cheol Kang
Pituitary.2021; 24(6): 955. CrossRef
- Spherical type of amyloidogenic pituitary prolactinoma in a 50 year old male

- Hypothalamus and Pituitary gland
- Precision Therapy in Acromegaly Caused by Pituitary Tumors: How Close Is It to Reality?
- Cheol Ryong Ku, Vladimir Melnikov, Zhaoyun Zhang, Eun Jig Lee
- Endocrinol Metab. 2020;35(2):206-216. Published online June 24, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.2.206
- 6,719 View
- 250 Download
- 8 Web of Science
- 8 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
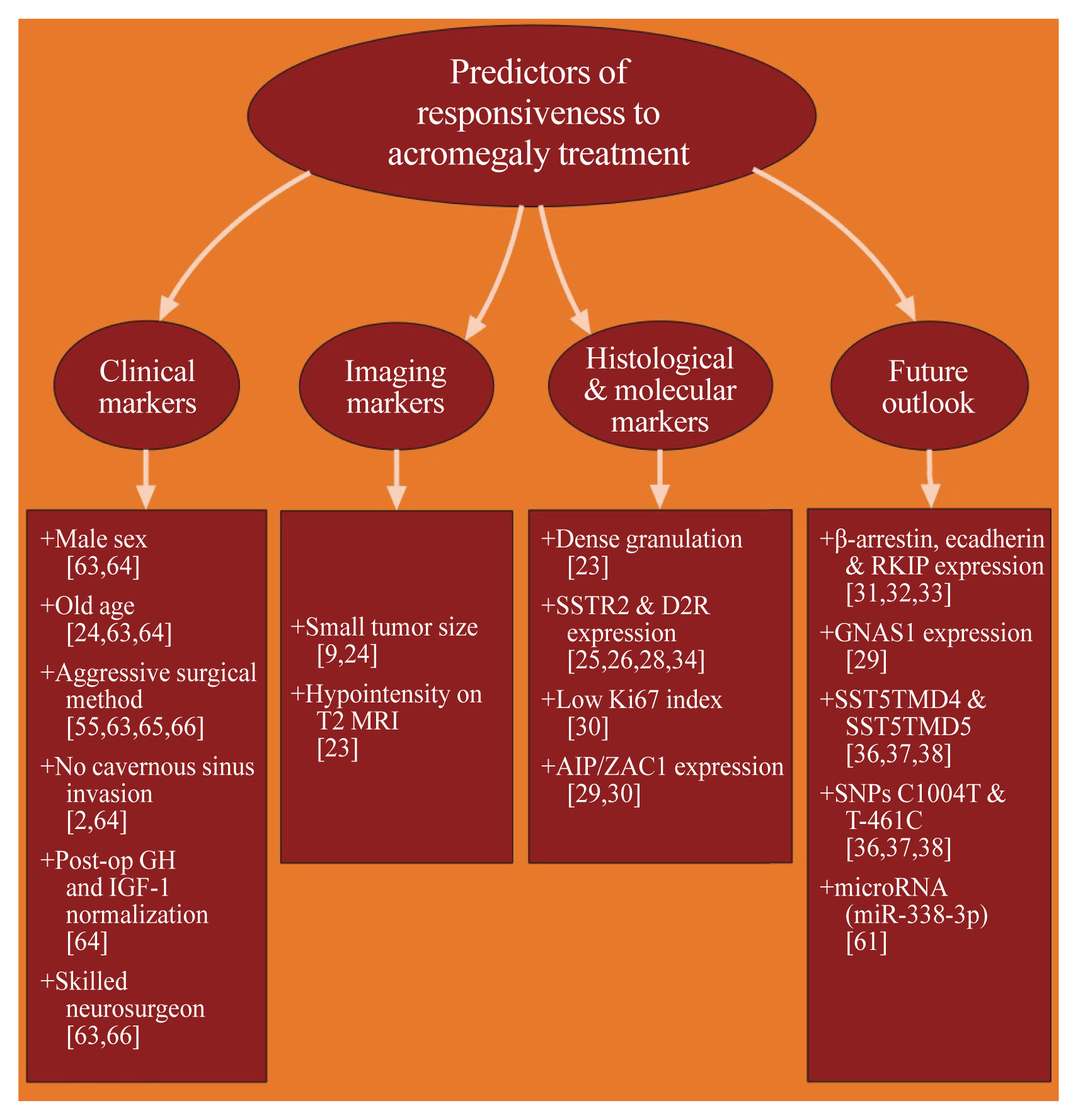
ePub - Acromegaly presents with an enigmatic range of symptoms and comorbidities caused by chronic and progressive growth hormone elevations, commonly due to endocrinologic hypersecretion from a pituitary gland tumor. Comprehensive national acromegaly databases have been appearing over the years, allowing for international comparisons of data, although still presenting varying prevalence and incidence rates. Lack of large-scale analysis in geographical and ethnic differences in clinical presentation and management requires further research. Assessment of current and novel predictors of responsiveness to distinct therapy can lead to multilevel categorization of patients, allowing integration into new clinical guidelines and reduction of increased morbidity and mortality associated with acromegaly. This review compares current data from epidemiological studies and assesses the present-day application of prognostic factors in medical practice, the reality of precision therapy, as well as its future prospects in acromegaly, with a special focus on its relevance to the South Korean population.
-
Citations
Citations to this article as recorded by- Biomarkers of response to treatment in acromegaly
Leandro Kasuki, Elisa Lamback, Ximene Antunes, Mônica R. Gadelha
Expert Review of Endocrinology & Metabolism.2024; 19(1): 71. CrossRef - Multiomics Approach to Acromegaly: Unveiling Translational Insights for Precision Medicine
Kyungwon Kim, Cheol Ryong Ku, Eun Jig Lee
Endocrinology and Metabolism.2023; 38(5): 463. CrossRef - Risk of depression in patients with acromegaly in Korea (2006-2016): a nationwide population-based study
Shinje Moon, Sangmo Hong, Kyungdo Han, Cheol-Young Park
European Journal of Endocrinology.2023; 189(3): 363. CrossRef - The Future of Somatostatin Receptor Ligands in Acromegaly
Monica R Gadelha, Luiz Eduardo Wildemberg, Leandro Kasuki
The Journal of Clinical Endocrinology & Metabolism.2022; 107(2): 297. CrossRef - Innovative therapeutics in acromegaly
Leandro Kasuki, Mônica R. Gadelha
Best Practice & Research Clinical Endocrinology & Metabolism.2022; 36(6): 101679. CrossRef - Risk of Neurodegenerative Diseases in Patients With Acromegaly
Sangmo Hong, Kyungdo Han, Kyung-Soo Kim, Cheol-Young Park
Neurology.2022;[Epub] CrossRef - Machine Learning-based Prediction Model for Treatment of Acromegaly With First-generation Somatostatin Receptor Ligands
Luiz Eduardo Wildemberg, Aline Helen da Silva Camacho, Renan Lyra Miranda, Paula C L Elias, Nina R de Castro Musolino, Debora Nazato, Raquel Jallad, Martha K P Huayllas, Jose Italo S Mota, Tobias Almeida, Evandro Portes, Antonio Ribeiro-Oliveira, Lucio Vi
The Journal of Clinical Endocrinology & Metabolism.2021; 106(7): 2047. CrossRef - Skin anomalies in acromegalic patients (Review of the practical aspects)
Florica Sandru, Adelina Popa, Dan Paduraru, Alexandru Filipescu, Mara Carsote, Adina Ghemigian
Experimental and Therapeutic Medicine.2021;[Epub] CrossRef
- Biomarkers of response to treatment in acromegaly

- Endocrine Research
- Therapeutic Effect of a Novel Chimeric Molecule Targeting Both Somatostatin and Dopamine Receptors on Growth Hormone-Secreting Pituitary Adenomas
- Jean Kim, Ju Hun Oh, Heather Harlem, Michael D. Culler, Cheol Ryong Ku, Eun Jig Lee
- Endocrinol Metab. 2020;35(1):177-187. Published online March 19, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.1.177
- 4,491 View
- 106 Download
- 4 Web of Science
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
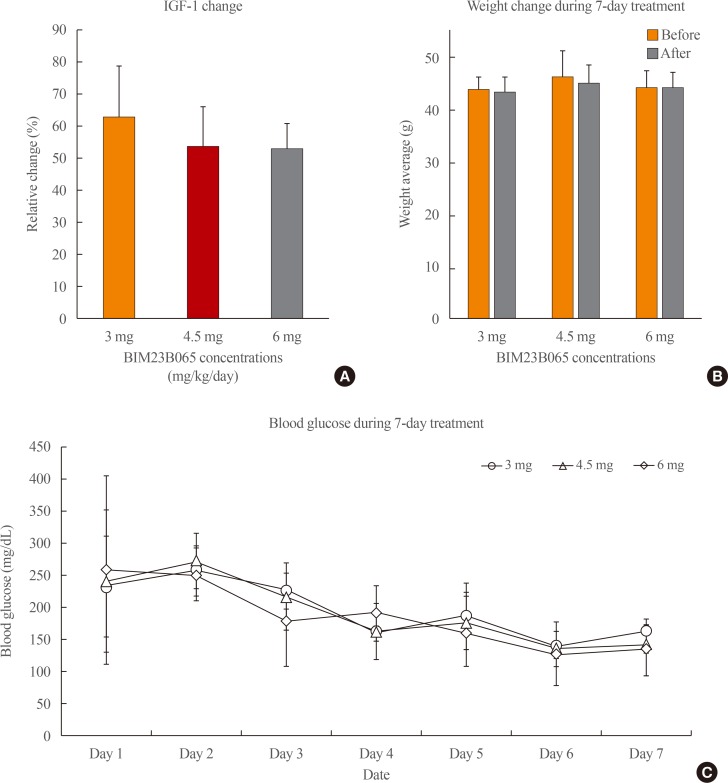
ePub Background Acromegaly is a rare disease primarily caused by growth hormone (GH)-secreting pituitary adenomas, and its treatment is costly. Moreover, some patients are unresponsive to treatment. Hence, there are increasing efforts to develop new drugs with improved effectiveness for this disease. BIM23B065 is a novel chimeric molecule that acts on both somatostatin and dopamine receptors. This study aimed to investigate the effects of BIM23B065 compared with those of a somatostatin receptor analog and a dopamine agonist.
Methods The effects of BIM23B065 on the proliferation, GH and insulin-like growth factor-1 (IGF-1) levels, and extracellular signal-regulated kinase (ERK) 1/2 and cyclic AMP response element binding (CREB) phosphorylation of GH3 cells were investigated with MTS assay, enzyme-linked immunosorbent assay, and Western blotting, respectively. The dosage and treatment duration of BIM23B065 were tested in animal models of GH-secreting pituitary adenoma. The effect of BIM23B065 (3 mg/kg/day) on changes in IGF-1 levels before and after treatment was further investigated.
Results In vitro , BIM23B065 treatment decreased GH release in the culture media and downregulated ERK 1/2 and CREB phosphorylation to 22% and 26%, respectively.In vivo , IGF-1 expression decreased to 50 % after 4 weeks of treatment with BIM23B065 using an osmotic pump implant. Moreover, magnetic resonance imaging results showed that the tumor size decreased significantly following treatment with BIM23B065 for 4 weeks.Conclusion The novel chimeric molecule was effective in decreasing IGF-1 and GH levels and may serve as an effective therapeutic agent for acromegaly.
-
Citations
Citations to this article as recorded by- Pituitary Tumorigenesis—Implications for Management
Rodanthi Vamvoukaki, Maria Chrysoulaki, Grigoria Betsi, Paraskevi Xekouki
Medicina.2023; 59(4): 812. CrossRef - Current and Emerging Medical Therapies in Pituitary Tumors
Nicolas Sahakian, Frédéric Castinetti, Thierry Brue, Thomas Cuny
Journal of Clinical Medicine.2022; 11(4): 955. CrossRef - Characterization of the ability of a, second-generation SST-DA chimeric molecule, TBR-065, to suppress GH secretion from human GH-secreting adenoma cells
Thomas Cuny, Thomas Graillon, Célines Defilles, Rakesh Datta, Shengwen Zhang, Dominique Figarella-Branger, Henry Dufour, Grégory Mougel, Thierry Brue, Tanya Landsman, Heather A. Halem, Michael D. Culler, Anne Barlier, Alexandru Saveanu
Pituitary.2021; 24(3): 351. CrossRef - Efficacy of a Novel Second-Generation Somatostatin-Dopamine Chimera (TBR-065) in Human Medullary Thyroid Cancer: A Preclinical Study
Alessandra Dicitore, Maria Celeste Cantone, Germano Gaudenzi, Davide Saronni, Silvia Carra, Maria Orietta Borghi, Manuela Albertelli, Diego Ferone, Leo J. Hofland, Luca Persani, Giovanni Vitale
Neuroendocrinology.2021; 111(10): 937. CrossRef - Emerging drugs for the treatment of acromegaly
Claudia Campana, Giuliana Corica, Federica Nista, Francesco Cocchiara, Giulia Graziani, Keyvan Khorrami, Marta Franco, Mara Boschetti, Diego Ferone, Federico Gatto
Expert Opinion on Emerging Drugs.2020; 25(4): 409. CrossRef
- Pituitary Tumorigenesis—Implications for Management

- Endocrine Research
- Transcriptome Analysis Identifies an Attenuated Local Immune Response in Invasive Nonfunctioning Pituitary Adenomas
- Yong Hwy Kim, Jung Hee Kim
- Endocrinol Metab. 2019;34(3):314-322. Published online September 26, 2019
- DOI: https://doi.org/10.3803/EnM.2019.34.3.314
- Correction in: Endocrinol Metab 2020;35(4):965
- 5,256 View
- 58 Download
- 12 Web of Science
- 10 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub Background Invasive nonfunctioning pituitary adenomas (NFPAs) remain challenging due to their high complication rate and poor prognosis. We aimed to identify the distinctive molecular signatures of invasive NFPAs, compared with noninvasive NFPAs, using gene expression profiling by RNA sequencing.
Methods We obtained frozen fresh tissue samples from 14 patients with NFPAs who underwent primary transsphenoidal surgery. Three non-invasive and 11 invasive NFPAs were used for RNA sequencing. The bioinformatics analysis included differential gene expression, gene ontology analysis, and pathway analysis.
Results A total of 700 genes were differentially expressed (59 up-regulated and 641 down-regulated genes) between invasive and non-invasive NFPAs (false discovery rate <0.1, and |fold change| ≥2). Using the down-regulated genes in invasive NFPAs, gene ontology enrichment analyses and pathway analyses demonstrated that the local immune response was attenuated and that transforming growth factor-β (TGF-β) RII-initiated TGF-β signaling was down-regulated in invasive NFPAs. The overexpression of claudin-9 (
CLDN9 ) and the down-regulation of insulin-like growth factor-binding protein 5 (IGFBP5 ), death-associated protein kinase 1 (DAPK1 ), and tissue inhibitor of metalloproteinase-3 (TIMP3 ) may be related with invasiveness in NFPAs.Conclusion Invasive NFPAs harbor different gene expression profiles relative to noninvasive NFPAs. In particular, local suppression of the immune response and TGF-β signaling can make PAs prone to invasiveness.
-
Citations
Citations to this article as recorded by- Transcriptome of GH-producing pituitary neuroendocrine tumours and models are significantly affected by somatostatin analogues
Rihards Saksis, Olesja Rogoza, Helvijs Niedra, Kaspars Megnis, Ilona Mandrika, Inga Balcere, Liva Steina, Janis Stukens, Austra Breiksa, Jurijs Nazarovs, Jelizaveta Sokolovska, Ilze Konrade, Raitis Peculis, Vita Rovite
Cancer Cell International.2023;[Epub] CrossRef - PD-L1 Expression in Pituitary Neuroendocrine Tumors/Pituitary Adenomas
Giulia Cossu, Stefano La Rosa, Jean Philippe Brouland, Nelly Pitteloud, Ethan Harel, Federico Santoni, Maxime Brunner, Roy Thomas Daniel, Mahmoud Messerer
Cancers.2023; 15(18): 4471. CrossRef - Aggressive PitNETs and Potential Target Therapies: A Systematic Review of Molecular and Genetic Pathways
Simona Serioli, Ludovico Agostini, Alberto Pietrantoni, Federico Valeri, Flavia Costanza, Sabrina Chiloiro, Barbara Buffoli, Amedeo Piazza, Pietro Luigi Poliani, Maria Peris-Celda, Federica Iavarone, Simona Gaudino, Marco Gessi, Giovanni Schinzari, Pier P
International Journal of Molecular Sciences.2023; 24(21): 15719. CrossRef - Muti-omics integration analysis revealed molecular network alterations in human nonfunctional pituitary neuroendocrine tumors in the framework of 3P medicine
Siqi Wen, Chunling Li, Xianquan Zhan
EPMA Journal.2022; 13(1): 9. CrossRef - A systematic review of molecular alterations in invasive non-functioning pituitary adenoma
Nazanin Hosseinkhan, Maryam Honardoost, Zahra Emami, Sara Cheraghi, Nahid Hashemi-Madani, Mohammad E. Khamseh
Endocrine.2022; 77(3): 500. CrossRef - Medication for Acromegaly Reduces Expression of MUC16, MACC1 and GRHL2 in Pituitary Neuroendocrine Tumour Tissue
Rihards Saksis, Ivars Silamikelis, Pola Laksa, Kaspars Megnis, Raitis Peculis, Ilona Mandrika, Olesja Rogoza, Ramona Petrovska, Inga Balcere, Ilze Konrade, Liva Steina, Janis Stukens, Austra Breiksa, Jurijs Nazarovs, Jelizaveta Sokolovska, Valdis Pirags,
Frontiers in Oncology.2021;[Epub] CrossRef - Large Scale Molecular Studies of Pituitary Neuroendocrine Tumors: Novel Markers, Mechanisms and Translational Perspectives
Raitis Peculis, Helvijs Niedra, Vita Rovite
Cancers.2021; 13(6): 1395. CrossRef - Nkx3-1 and Fech genes might be switch genes involved in pituitary non-functioning adenoma invasiveness
Nasibeh Khayer, Maryam Jalessi, Amin Jahanbakhshi, Alireza Tabib khooei, Mehdi Mirzaie
Scientific Reports.2021;[Epub] CrossRef - The tumour microenvironment of pituitary neuroendocrine tumours
Pedro Marques, Ashley B. Grossman, Márta Korbonits
Frontiers in Neuroendocrinology.2020; 58: 100852. CrossRef - The Progress of Immunotherapy in Refractory Pituitary Adenomas and Pituitary Carcinomas
Congxin Dai, Siyu Liang, Bowen Sun, Jun Kang
Frontiers in Endocrinology.2020;[Epub] CrossRef
- Transcriptome of GH-producing pituitary neuroendocrine tumours and models are significantly affected by somatostatin analogues

- Bone Metabolism
- Testosterone Replacement and Bone Mineral Density in Male Pituitary Tumor Patients
- Min Jeong Lee, Hyoung Kyu Ryu, So-Yeon An, Ja Young Jeon, Ji In Lee, Yoon-Sok Chung
- Endocrinol Metab. 2014;29(1):48-53. Published online March 14, 2014
- DOI: https://doi.org/10.3803/EnM.2014.29.1.48
- 3,502 View
- 32 Download
- 11 Web of Science
- 11 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader Background Hypopituitarism is associated with osteoporosis and osteopenia especially when hypogonadotropic hypogonadism is present. Despite hypopituitarism being an important cause of secondary osteoporosis, osteoporosis in patients receiving surgery for pituitary tumors in Korea has not been studied. In this study, we evaluated the effects of testosterone replacement therapy (TRT) on bone mineral density (BMD) in postoperative hypogonadal patients with pituitary tumors.
Methods To examine the effect of TRT on BMD, we performed a retrospective observational study in 21 postoperative male patients who underwent pituitary tumor surgery between 2003 and 2012 at the Ajou University Hospital. Testosterone was replaced in postoperative hypogonadal patients by regular intramuscular injection, daily oral medication, or application of transdermal gel. BMD (g/cm2) measurements of central skeletal sites (lumbar spine, femoral neck, and total femur) were obtained using dual-energy X-ray absorptiometry (GE Lunar). For lumbar spine BMD, L1 to L4 values were chosen for analysis. Femur neck and total femur were also analyzed.
Results During the follow-up period (mean, 56 months; range, 12 to 99 months) serum testosterone levels increased with the administration of TRT (
P =0.007). There was significant improvement (4.56%±9.81%) in the lumbar spine BMD compared to baseline BMD. There were no significant changes in the femur neck BMD or total femur BMD. We did not find any statistically significant relationships between changes in testosterone levels and BMD using Spearman correlation analysis.Conclusion Our results indicated that TRT used in the postoperative period for hypogonadal pituitary tumor surgery patients may have beneficial effects on the BMD of the spine.
-
Citations
Citations to this article as recorded by- Testosterone supplementation and bone parameters: a systematic review and meta-analysis study
G. Corona, W. Vena, A. Pizzocaro, V. A. Giagulli, D. Francomano, G. Rastrelli, G. Mazziotti, A. Aversa, A. M. Isidori, R. Pivonello, L. Vignozzi, E. Mannucci, M. Maggi, A. Ferlin
Journal of Endocrinological Investigation.2022; 45(5): 911. CrossRef - Physiological testosterone replacement effects on male aged rats with orchiectomy-induced osteoporosis in advanced stage: a tomographic and biomechanical pilot study
Vinícius de Paiva Gonçalves, Adriana Alicia Cabrera-Ortega, Jhonatan de Souza Carvalho, Dania Ramadan, Luís Carlos Spolidorio
The Aging Male.2021; 24(1): 139. CrossRef - Androgens and Androgen Receptor Actions on Bone Health and Disease: From Androgen Deficiency to Androgen Therapy
Jia-Feng Chen, Pei-Wen Lin, Yi-Ru Tsai, Yi-Chien Yang, Hong-Yo Kang
Cells.2019; 8(11): 1318. CrossRef - Testosterone and male rejuvenation
Sevann Helo, Peyton Thomas, Nicholas N. Tadros
Panminerva Medica.2019;[Epub] CrossRef - Systemic Non-Reproductive Effects of Sex Steroids in Adult Males and Females
Syed Imran Ali Shah
Human Physiology.2018; 44(1): 83. CrossRef - Benefits and Health Implications of Testosterone Therapy in Men With Testosterone Deficiency
Abdulmaged M. Traish
Sexual Medicine Reviews.2018; 6(1): 86. CrossRef - Multiple Fractures in Patient with Graves' Disease Accompanied by Isolated Hypogonadotropic Hypogonadism
Hyon-Seung Yi, Ji Min Kim, Sang Hyeon Ju, Younghak Lee, Hyun Jin Kim, Koon Soon Kim
Journal of Bone Metabolism.2016; 23(1): 40. CrossRef - Severity and pattern of bone mineral loss in endocrine causes of osteoporosis as compared to age-related bone mineral loss
D Dutta, P Dharmshaktu, A Aggarwal, K Gaurav, R Bansal, N Devru, UC Garga, B Kulshreshtha
Journal of Postgraduate Medicine.2016; 62(3): 162. CrossRef - Articles in 'Endocrinology and Metabolism' in 2014
Won-Young Lee
Endocrinology and Metabolism.2015; 30(1): 47. CrossRef - Bone health in hypogonadal men
Michael S. Irwig
Current Opinion in Urology.2014; 24(6): 608. CrossRef - Testosterone Replacement Therapy and Bone Mineral Density in Men with Hypogonadism
Se Hwa Kim
Endocrinology and Metabolism.2014; 29(1): 30. CrossRef
- Testosterone supplementation and bone parameters: a systematic review and meta-analysis study

- Adrenal gland
- The Molecular Pathogenesis of Pituitary Adenomas: An Update
- Xiaobing Jiang, Xun Zhang
- Endocrinol Metab. 2013;28(4):245-254. Published online December 12, 2013
- DOI: https://doi.org/10.3803/EnM.2013.28.4.245
- 4,076 View
- 43 Download
- 37 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader Pituitary tumors represent the most common intracranial neoplasms accompanying serious morbidity through mass effects and inappropriate secretion of pituitary hormones. Understanding the etiology of pituitary tumorigenesis will facilitate the development of satisfactory treatment for pituitary adenomas. Although the pathogenesis of pituitary adenomas is largely unknown, considerable evidence indicates that the pituitary tumorigenesis is a complex process involving multiple factors, including genetic and epigenetic changes. This review summarized the recent progress in the study of pituitary tumorigenesis, focusing on the role of tumor suppressor genes, oncogenes and microRNAs.
-
Citations
Citations to this article as recorded by- Plurihormonal Pituitary Neuroendocrine Tumors: Clinical Relevance of Immunohistochemical Analysis
Roxana-Ioana Dumitriu-Stan, Iulia-Florentina Burcea, Ramona Dobre, Valeria Nicoleta Nastase, Raluca Amalia Ceausu, Marius Raica, Catalina Poiana
Diagnostics.2024; 14(2): 170. CrossRef - LncRNA MYMLR promotes pituitary adenoma development by upregulating carbonyl reductase 1 via sponging miR-197-3p
Tuo Wang, Ping Mao, Yan Zhang, Bo Cui, Mao-De Wang, Ya Li, Ke Gao
Anti-Cancer Drugs.2022; 33(10): 1058. CrossRef - Transcriptomic Profiles of Normal Pituitary Cells and Pituitary Neuroendocrine Tumor Cells
Jun Y. Oh, Robert C. Osorio, Jangham Jung, Luis Carrete, Nikita Choudhary, Meeki Lad, Atul Saha, Manish K. Aghi
Cancers.2022; 15(1): 110. CrossRef - EMT-Related Markers in Serum Exosomes are Potential Diagnostic Biomarkers for Invasive Pituitary Adenomas
Kelin Chen, Guoge Li, Xixiong Kang, Pinan Liu, Lingye Qian, Yijun Shi, Rasha Alsamani Osman, Zhijun Yang, Guojun Zhang
Neuropsychiatric Disease and Treatment.2021; Volume 17: 3769. CrossRef - The Role of Long Noncoding RNAs in the Biology of Pituitary Adenomas
Ozal Beylerli, Ilgiz Gareev, Valentin Pavlov, Xin Chen, Shiguang Zhao
World Neurosurgery.2020; 137: 252. CrossRef - Sellar Tumors
Katherine E. Schwetye, Sonika M. Dahiya
Surgical Pathology Clinics.2020; 13(2): 305. CrossRef - Genomic and molecular characterization of pituitary adenoma pathogenesis: review and translational opportunities
Mazin Elsarrag, Parantap D. Patel, Ajay Chatrath, Davis Taylor, John A. Jane
Neurosurgical Focus.2020; 48(6): E11. CrossRef - Metabolic profiling reveals distinct metabolic alterations in different subtypes of pituitary adenomas and confers therapeutic targets
Jie Feng, Hua Gao, Qi Zhang, Yang Zhou, Chuzhong Li, Sida Zhao, Lichuan Hong, Jinjin Yang, Shuyu Hao, Wan Hong, Zhengping Zhuang, Guowang Xu, Yazhuo Zhang
Journal of Translational Medicine.2019;[Epub] CrossRef - Quantitative Analysis of Proteome in Non-functional Pituitary Adenomas: Clinical Relevance and Potential Benefits for the Patients
Tingting Cheng, Ya Wang, Miaolong Lu, Xiaohan Zhan, Tian Zhou, Biao Li, Xianquan Zhan
Frontiers in Endocrinology.2019;[Epub] CrossRef - circOMA1-Mediated miR-145-5p Suppresses Tumor Growth of Nonfunctioning Pituitary Adenomas by Targeting TPT1
Qiu Du, Bin Hu, Yajuan Feng, Zongming Wang, Xin Wang, Dimin Zhu, Yonghong Zhu, Xiaobing Jiang, Haijun Wang
The Journal of Clinical Endocrinology & Metabolism.2019; 104(6): 2419. CrossRef - Differential Expression of HMGA1 and HMGA2 in pituitary neuroendocrine tumors
Sérgio Portovedo, Nadja Gaido, Bruno de Almeida Nunes, Ana Giselia Nascimento, Allysson Rocha, Marcelo Magalhães, Gilvan Cortes Nascimento, Denise Pires de Carvalho, Paula Soares, Christina Takiya, Manuel dos Santos Faria, Leandro Miranda-Alves
Molecular and Cellular Endocrinology.2019; 490: 80. CrossRef - Double Pituitary Adenomas with Synchronous Somatotroph and Corticotroph Clinical Presentation of Acromegaly and Cushing's Disease
Naomi Collazo-Gutiérrez, Orlando de Jesús, Maria Villamil-Jarauta, Milliette Alvarado, Loida González, Margarita Ramírez, Victor J. Carlo-Chevere
World Neurosurgery.2019; 132: 161. CrossRef - Association of ApoE haplotype with clinical evidence of pituitary adenoma
Agne Sidaraite, Alvita Vilkeviciute, Brigita Glebauskiene, Loresa Kriauciuniene, Dalia Zaliuniene, Rasa Liutkeviciene
Gene.2019; 706: 154. CrossRef - Next-generation sequencing of microRNAs reveals a unique expression pattern in different types of pituitary adenomas
Zongze He, Longyi Chen, Xiao Hu, Jian Tang, Linfu He, Junting Hu, Fan Fei, Qi Wang
Endocrine Journal.2019; 66(8): 709. CrossRef - Growth hormone and prolactin-staining tumors causing acromegaly: a retrospective review of clinical presentations and surgical outcomes
Jonathan Rick, Arman Jahangiri, Patrick M. Flanigan, Ankush Chandra, Sandeep Kunwar, Lewis Blevins, Manish K. Aghi
Journal of Neurosurgery.2019; 131(1): 147. CrossRef - Study of major genetic factors involved in pituitary tumorigenesis and their impact on clinical and biological characteristics of sporadic somatotropinomas and non-functioning pituitary adenomas
R.K. Foltran, P.V.G.H. Amorim, F.H. Duarte, I.P.P. Grande, A.C.T.B. Freire, F.P. Frassetto, J.B. Dettoni, V.A. Alves, I. Castro, E.B. Trarbach, M.D. Bronstein, R.S. Jallad
Brazilian Journal of Medical and Biological Research.2018;[Epub] CrossRef - Detection of circulating tumor cells in patients with pituitary tumors
Gao Hua, He Yanjiao, Liu Qian, Wang Jichao, Zhang Yazhuo
BMC Cancer.2018;[Epub] CrossRef - Plurihormonal ACTH-GH Pituitary Adenoma: Case Report and Systematic Literature Review
Elena Roca, Pier Paolo Mattogno, Teresa Porcelli, Luigi Poliani, Francesco Belotti, Alberto Schreiber, Filippo Maffezzoni, Marco Maria Fontanella, Francesco Doglietto
World Neurosurgery.2018; 114: e158. CrossRef - The role of galectin-3 in the tumorigenesis and progression of pituitary tumors
Bo Diao, Ying Liu, Guo‑Zheng Xu, Yi Zhang, Jun Xie, Jie Gong
Oncology Letters.2018;[Epub] CrossRef - Programmed cell senescence: role of IL-6 in the pituitary
Melanie Sapochnik, Mariana Fuertes, Eduardo Arzt
Journal of Molecular Endocrinology.2017; 58(4): R241. CrossRef - Selective molecular biomarkers to predict biologic behavior in pituitary tumors
Aydin Sav, Fabio Rotondo, Luis V. Syro, Meric A. Altinoz, Kalman Kovacs
Expert Review of Endocrinology & Metabolism.2017; 12(3): 177. CrossRef - MicroRNA-200b inhibits pituitary tumor cell proliferation and invasion by targeting PKCα
Yuanchuan Wang, Xiaohong Yin, Long Zhao, Shun Li, Jie Duan, Renzhao Kuang, Junwei Duan
Experimental and Therapeutic Medicine.2017; 14(2): 1706. CrossRef - Increased expression of the microRNA 106b~25 cluster and its host gene MCM7 in corticotroph pituitary adenomas is associated with tumor invasion and Crooke’s cell morphology
Filip Garbicz, Dawid Mehlich, Beata Rak, Emir Sajjad, Maria Maksymowicz, Wiktor Paskal, Grzegorz Zieliński, Paweł K. Włodarski
Pituitary.2017; 20(4): 450. CrossRef - Bromocriptine Induces Autophagy-Dependent Cell Death in Pituitary Adenomas
Xin Geng, Lixin Ma, Zefu Li, Zhenzhu Li, Jianmin Li, Meng Li, Qingbo Wang, Zheng Chen, Qikai Sun
World Neurosurgery.2017; 100: 407. CrossRef - Biomarkers of pituitary carcinomas
Aydin Sav, Fabio Rotondo, Luis V. Syro, Antonio Di Ieva, Michael D. Cusimano, Kalman Kovacs
Expert Review of Endocrinology & Metabolism.2016; 11(3): 253. CrossRef - MicroRNAs in the pituitary
Erica Gentilin, Ettore degli Uberti, Maria Chiara Zatelli
Best Practice & Research Clinical Endocrinology & Metabolism.2016; 30(5): 629. CrossRef - Molecular markers in pituitary tumors
Asha M. Robertson, Anthony P. Heaney
Current Opinion in Endocrinology, Diabetes & Obesity.2016; 23(4): 324. CrossRef - MicroRNA-106b promotes pituitary tumor cell proliferation and invasion through PI3K/AKT signaling pathway by targeting PTEN
Kai Zhou, Tingrong Zhang, YanDong Fan, Serick, Guojia Du, Pengfei Wu, Dangmurenjiafu Geng
Tumor Biology.2016; 37(10): 13469. CrossRef - Isolated double adrenocorticotropic hormone-secreting pituitary adenomas: A case report and review of the literature
JIUJUN PU, ZHIMING WANG, HUI ZHOU, AILING ZHONG, KAI JIN, LUNLIANG RUAN, GANG YANG
Oncology Letters.2016; 12(1): 585. CrossRef - Progress in Endocrine Neoplasia
Samuel A. Wells
Clinical Cancer Research.2016; 22(20): 4981. CrossRef - Pituitary adenomas: historical perspective, surgical management and future directions
Debebe Theodros, Mira Patel, Jacob Ruzevick, Michael Lim, Chetan Bettegowda
CNS Oncology.2015; 4(6): 411. CrossRef - Epidrug mediated re-expression of miRNA targeting the HMGA transcripts in pituitary cells
Mark O. Kitchen, Kiren Yacqub-Usman, Richard D. Emes, Alan Richardson, Richard N. Clayton, William E. Farrell
Pituitary.2015; 18(5): 674. CrossRef - Pituitary Adenoma and the Chemokine Network: A Systemic View
Fabio Grizzi, Elena Monica Borroni, Alessandro Vacchini, Dorina Qehajaj, Manuela Liguori, Sanja Stifter, Maurizio Chiriva-Internati, Antonio Di Ieva
Frontiers in Endocrinology.2015;[Epub] CrossRef - MiR-132, miR-15a and miR-16 synergistically inhibit pituitary tumor cell proliferation, invasion and migration by targeting Sox5
Wang Renjie, Liang Haiqian
Cancer Letters.2015; 356(2): 568. CrossRef - Brief Review of Articles in 'Endocrinology and Metabolism' in 2013
Won-Young Lee
Endocrinology and Metabolism.2014; 29(3): 251. CrossRef - Epigenetics of pituitary tumours
William E. Farrell
Current Opinion in Endocrinology, Diabetes & Obesity.2014; 21(4): 299. CrossRef - The Role of Genetic and Epigenetic Changes in Pituitary Tumorigenesis
Hidenori FUKUOKA, Yutaka TAKAHASHI
Neurologia medico-chirurgica.2014; 54(12): 943. CrossRef
- Plurihormonal Pituitary Neuroendocrine Tumors: Clinical Relevance of Immunohistochemical Analysis

- Adrenal gland
- Characteristics of Acromegaly in Korea with a Literature Review
- Jae Won Hong, Cheol Ryong Ku, Sun Ho Kim, Eun Jig Lee
- Endocrinol Metab. 2013;28(3):164-168. Published online September 13, 2013
- DOI: https://doi.org/10.3803/EnM.2013.28.3.164
- 3,665 View
- 50 Download
- 12 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader Acromegaly is a slowly progressive disease caused by excessive growth hormone (GH), which is related to a GH secreting pituitary tumor in most cases. Herein, we describe the epidemiology, clinical characteristics, and treatment of acromegaly in Korea with a literature review. The average annual incidence of acromegaly in Korea was 3.9 cases per million people, which was within the range of previous Western studies. The primary treatment for acromegaly was also transsphenoidal adenomectomy, which accounted for 90.4% of patients whose primary therapeutic options were known. The overall surgical remission rates were 89%, 87%, 64%, 70%, and 50% for modified Hardy classification I, II, IIIA, IIIB, and IV, respectively. An updated and larger study regarding the treatment outcome of medical/radiotherapy in Korean acromegalic patients is needed.
-
Citations
Citations to this article as recorded by- Risk of depression in patients with acromegaly in Korea (2006-2016): a nationwide population-based study
Shinje Moon, Sangmo Hong, Kyungdo Han, Cheol-Young Park
European Journal of Endocrinology.2023; 189(3): 363. CrossRef - Increased Risk of Hip Fracture in Patients with Acromegaly: A Nationwide Cohort Study in Korea
Jiwon Kim, Namki Hong, Jimi Choi, Ju Hyung Moon, Eui Hyun Kim, Eun Jig Lee, Sin Gon Kim, Cheol Ryong Ku
Endocrinology and Metabolism.2023; 38(6): 690. CrossRef - Selective screening of patients with associated somatic diseases as a method of early detection of acromegaly
M. B. Antsiferov, V. S. Pronin, T. M. Alekseeva, O. A. Ionova, E. Y. Martynova, Yu. E. Poteshkin, N. A. Chubrova, K. Y. Zherebchikova
Problems of Endocrinology.2021; 67(1): 20. CrossRef - Precision Therapy in Acromegaly Caused by Pituitary Tumors: How Close Is It to Reality?
Cheol Ryong Ku, Vladimir Melnikov, Zhaoyun Zhang, Eun Jig Lee
Endocrinology and Metabolism.2020; 35(2): 206. CrossRef - Patient Characteristics, Diagnostic Delays, Treatment Patterns, Treatment Outcomes, Comorbidities, and Treatment Costs of Acromegaly in China: A Nationwide Study
Xiaopeng Guo, Kailu Wang, Siyue Yu, Lu Gao, Zihao Wang, Huijuan Zhu, Bing Xing, Shuyang Zhang, Dong Dong
Frontiers in Endocrinology.2020;[Epub] CrossRef - Epidemiología de la acromegalia en Ecuador
Enrique López Gavilanez, Kempis Guerrero Franco, Narcisa Solórzano Zambrano, Manuel Navarro Chávez, Camilo López Estrella, Luis Vaca Burbano, Eduardo Marriott Díaz
Endocrinología y Nutrición.2016; 63(7): 333. CrossRef - A magnetic resonance imaging‐based classification system for indication of trans‐sphenoidal hypophysectomy in canine pituitary‐dependent hypercortisolism
A. Sato, T. Teshima, H. Ishino, Y. Harada, T. Yogo, N. Kanno, D. Hasegawa, Y. Hara
Journal of Small Animal Practice.2016; 57(5): 240. CrossRef - Epidemiology of acromegaly in Ecuador
Enrique López Gavilanez, Kempis Guerrero Franco, Narcisa Solórzano Zambrano, Manuel Navarro Chávez, Camilo López Estrella, Luis Vaca Burbano, Eduardo Marriott Díaz
Endocrinología y Nutrición (English Edition).2016; 63(7): 333. CrossRef - An Association Study Between Gene Polymorphisms of Folic Acid Metabolism Enzymes and Biochemical and Hormonal Parameters in Acromegaly
Aslı Tetik Vardarlı, Ayhan Zengi, Vildan Bozok Çetintaş, Muammer Karadeniz, Sadık Tamsel, Ali Şahin Küçükaslan, Timur Köse, Füsun Saygılı, Zuhal Eroglu
Genetic Testing and Molecular Biomarkers.2015; 19(8): 431. CrossRef - Diagnosis, treatment and clinical perspectives of acromegaly
Ferdinand Roelfsema, Gerrit van den Berg
Expert Review of Endocrinology & Metabolism.2015; 10(6): 619. CrossRef - Change in quality of life in patients with acromegaly after treatment with octreotide LAR: first application of AcroQoL in Korea
S. O. Chin, C. H. Chung, Y.-S. Chung, B.-J. Kim, H. Y. Kim, I.-J. Kim, J. G. Kim, M.-S. Kim, S.-Y. Kim, E. J. Lee, K. Y. Lee, S.-W. Kim
BMJ Open.2015; 5(6): e006898. CrossRef - Brief Review of Articles in 'Endocrinology and Metabolism' in 2013
Won-Young Lee
Endocrinology and Metabolism.2014; 29(3): 251. CrossRef
- Risk of depression in patients with acromegaly in Korea (2006-2016): a nationwide population-based study


 KES
KES

 First
First Prev
Prev



