Articles
- Page Path
- HOME > Endocrinol Metab > Volume 32(4); 2017 > Article
-
Review ArticleRecent Advances in Core Needle Biopsy for Thyroid Nodules
-
Chan Kwon Jung1
 , Jung Hwan Baek2
, Jung Hwan Baek2
-
Endocrinology and Metabolism 2017;32(4):407-412.
DOI: https://doi.org/10.3803/EnM.2017.32.4.407
Published online: December 14, 2017
1Department of Hospital Pathology, College of Medicine, The Catholic University of Korea, Seoul, Korea.
2Department of Radiology and Research Institute of Radiology, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea.
- Corresponding author: Jung Hwan Baek. Department of Radiology and Research Institute of Radiology, Asan Medical Center, University of Ulsan College of Medicine, 86 Asanbyeongwon-gil, Songpa-gu, Seoul 05505, Korea. Tel: +82-2-3010-4348, Fax: +82-2-476-0090, radbaek@naver.com
• Received: November 23, 2017 • Revised: November 30, 2017 • Accepted: December 4, 2017
Copyright © 2017 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
- Core needle biopsy (CNB) was introduced as an alternative diagnostic tool to fine-needle aspiration (FNA), and is increasingly being used in the preoperative assessment of thyroid nodules. CNB provides a definitive diagnosis in most cases, but it sometimes may be inconclusive. CNB has the advantage of enabling a histologic examination in relation to the surrounding thyroid tissue, immunohistochemistry, and molecular testing that can provide a more accurate assessment than FNA in selected cases. Nevertheless, CNB should be performed only by experienced experts in thyroid interventions to prevent complications because CNB needles are larger in caliber than FNA needles. As recent evidence has accumulated, and with improvements in the technique and devices for thyroid CNB, the Korean Society of Thyroid Radiology released its 2016 thyroid CNB guidelines and the Korean Endocrine Pathology Thyroid Core Needle Biopsy Study Group published a consensus statement on the pathology reporting system for thyroid CNB in 2015. This review presents the current consensus and recommendations regarding thyroid CNB, focusing on indications, complications, and pathologic classification and reporting.
- Fine-needle aspiration (FNA) cytology is the gold-standard diagnostic method for the preoperative diagnosis of thyroid nodules [12]. Although thyroid aspiration cytology using a large needle was initially introduced in the 1920s, FNA using a 23- to 27-gauge needle has been a mainstream diagnostic modality in the triage of thyroid nodules since the 1980s in most countries because of its diagnostic accuracy and safety [34567]. Core needle biopsy (CNB) of the thyroid was first introduced as an alternative to FNA in the 1990s [4] and is now widely used for the preoperative examination of thyroid nodules in Korea, with excellent results, because of advances in core needle devices (thinner needles and automatic devices), biopsy technology (new sampling technique) and ultrasound machine (high resolution) [8]. In this review, we present an overview of the indications, complications, and pathologic classification of thyroid CNB.
INTRODUCTION
- As evidence has accumulated regarding thyroid CNB, the Korean Society of Thyroid Radiology (KSThR) released its 2016 thyroid CNB guidelines [9]. These guidelines include recommendations regarding indications and complications, and are summarized in Table 1. A consensus statement on the pathology reporting system for thyroid CNB was published by the Korean Endocrine Pathology Thyroid Core Needle Biopsy Study Group in 2015 [10]. The six categorical diagnoses used in the Bethesda System for Reporting Thyroid Cytopathology (TBSRTC) were retained in the reporting system for thyroid CNB in order to ensure effective communication between pathologists and clinicians, with less likelihood of misinterpreting the pathologic results [110].
- The current thyroid guidelines of the American Association of Clinical Endocrinologists/American College of Endocrinology/Associazione Medici Endocrinologi, British Thyroid Association, and KSThR permit the limited use of CNB for thyroid nodules, whereas the American Thyroid Association does not recommend CNB [2911].
CURRENT GUIDELINES AND RECOMMENDATIONS
- The most widely accepted indications for CNB in the literature include previous FNA results of non-diagnostic [12131415] and atypia of undetermined significance [151617]. The tissue obtained by CNB provides more abundant material than FNA cytology, especially in cases with marked sclerosis and calcification. A recent meta-analysis of published data showed that the non-diagnostic and inconclusive rates of CNB were 5.5% (95% confidence interval [CI], 2.2% to 8.7%) and 8.0% (95% CI, 4.4% to 11.5%), respectively, whereas the non-diagnostic and inconclusive rates of FNA were 22.6% (95% CI, 12.2% to 33.0%) and 40.2% (95% CI, 25.1% to 55.3%), respectively [18]. In another meta-analysis of thyroid nodules with initially non-diagnostic FNA results, the non-diagnostic rate (6.4%; 95% CI, 3.3% to 16.1%) of follow-up CNB was significantly lower than that of repeated FNA (36.5%; 95% CI, 29.9% to 43.1%) [13]. In large cohort CNB studies, the false-negative rates of CNB ranged from 1% to 3% [192021].
- Although CNB may not be the method of choice for all thyroid nodules, previous studies have demonstrated the advantages of histologic diagnoses made using CNB specimens over the cytological diagnosis using FNA for several specific diseases. Malignant lymphoma, medullary thyroid carcinoma, anaplastic thyroid carcinoma, and parathyroid lesions can be confirmatively diagnosed with CNB based on the histologic morphology in conjunction with immunohistochemistry [222324]. While calcified nodules and degenerating nodules are often diagnosed as unsatisfactory with FNA because of the aspiration of acellular or paucicellular material, scant cellular nodules can be diagnosed as specific disease entities with CNB (Fig. 1) [252627].
- Follicular-patterned lesions include nodular hyperplasia, follicular adenoma, noninvasive follicular thyroid neoplasm with papillary-like nuclear features (NIFTP), the follicular variant of papillary thyroid carcinoma (PTC), and follicular thyroid carcinoma. While FNA and CNB cannot differentiate follicular adenoma from follicular carcinoma and NIFTP from the invasive encapsulated follicular variant of PTC because vascular or capsular invasion cannot be assessed without surgery, it is possible to divide these follicular-patterned lesions into almost certainly benign non-neoplastic lesions and follicular newoplasms with a risk of malignancy. NIFTP and the follicular variant of PTC are diagnosed based on nuclear features in conjunction with architectural atypia showing a follicular growth pattern. For the diagnosis of follicular neoplasm with CNB, it is recommended that the tissue sampling of CNB should include tumor tissue, the tumor capsule, and adjacent normal parenchyma (Fig. 2).
- A few studies have suggested that CNB is valuable as a first-line tool for diagnosing initially detected thyroid nodules [2128]. Suh et al. [21] reported that CNB had a non-diagnostic rate of 1.3% and an inconclusive result rate of 5.9%, a high diagnostic accuracy of 97.6%, and a complication rate of 0.2%.
INDICATIONS
- The current thyroid guidelines suggest that CNB is a safe, tolerable procedure [1529]. The complication rate is also acceptable (0% to 4.1%), with a low rate of major complications (0% to 1.9%). Recently a large-population single-center study showed no procedure-related deaths, and low rates of major (0.06%) and minor (0.79%) complications [30]. To minimize complications, CNB should be performed by well-trained doctors under real-time ultrasound monitoring. Moreover, knowledge of the neck anatomy, anatomical variations, and potential complications is also required for the safe performance of CNB [31].
SAFETY AND COMPLICATIONS
- The pathology reporting system of CNB is based on the six TBSRTC categories [10] and is summarized in Table 2. There is only one difference in terminology between the CNB reporting system and TBSRTC. The diagnostic category III for CNB is referred to as an “indeterminate lesion,” which corresponds to “atypia of undetermined significance” or “follicular lesion of undetermined significance” in TBSRTC. Subclassification based on the cytologic and/or architectural atypia in category III and IV was encouraged to enhance communication with clinicians and pathologists. Each diagnostic category in the TBSRTC has an implied risk of malignancy and clearly recommended clinical management steps regarding the triage of the patient for clinical follow-up, repeated FNA, or surgery. However, only limited data have been reported regarding the risk of malignancy in each diagnostic category of CNB.
PATHOLOGY REPORTING SYSTEM
- Thyroid CNB has been used as an alternative second option to FNA in patients with a previous non-diagnostic or indeterminate diagnosis, and is now suggested as a first-line tool for the diagnosis of thyroid nodule in selected cases. Recent studies have provided evidence for the efficacy and safety of thyroid CNB. Although the current thyroid CNB guidelines and pathologic reporting system are now widely available and affordable, further validation studies should be continued.
CONCLUSIONS
-
Acknowledgements
- This study was partially supported by a grant (HI16C2013) of the Korean Health Technology R&D Project, Ministry of Health and Welfare, Republic of Korea.
ACKNOWLEDGMENTS
-
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
Article information
- 1. Cibas ES, Ali SZ. The 2017 Bethesda system for reporting thyroid cytopathology. Thyroid 2017;27:1341–1346. ArticlePubMed
- 2. Haugen BR, Alexander EK, Bible KC, Doherty GM, Mandel SJ, Nikiforov YE, et al. 2015 American Thyroid Association management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: The American Thyroid Association Guidelines task force on thyroid nodules and differentiated thyroid cancer. Thyroid 2016;26:1–133. ArticlePubMedPMC
- 3. Jung CK, Hong S, Bychkov A, Kakudo K. The use of fine-needle aspiration (FNA) cytology in patients with thyroid nodules in Asia: a brief overview of studies from the Working Group of Asian Thyroid FNA Cytology. J Pathol Transl Med 2017;51:571–578. ArticlePubMedPMCPDF
- 4. Baek JH. Current status of core needle biopsy of the thyroid. Ultrasonography 2017;36:83–85. ArticlePubMedPMCPDF
- 5. Pitman MB, Abele J, Ali SZ, Duick D, Elsheikh TM, Jeffrey RB, et al. Techniques for thyroid FNA: a synopsis of the National Cancer Institute Thyroid Fine-Needle Aspiration State of the Science Conference. Diagn Cytopathol 2008;36:407–424. ArticlePubMed
- 6. Silverman JF, West RL, Finley JL, Larkin EW, Park HK, Swanson MS, et al. Fine-needle aspiration versus large-needle biopsy or cutting biopsy in evaluation of thyroid nodules. Diagn Cytopathol 1986;2:25–30. ArticlePubMed
- 7. Wang C, Vickery AL Jr, Maloof F. Needle biopsy of the thyroid. Surg Gynecol Obstet 1976;143:365–368. PubMed
- 8. Novoa E, Gurtler N, Arnoux A, Kraft M. Role of ultrasound-guided core-needle biopsy in the assessment of head and neck lesions: a meta-analysis and systematic review of the literature. Head Neck 2012;34:1497–1503. ArticlePubMed
- 9. Na DG, Baek JH, Jung SL, Kim JH, Sung JY, Kim KS, et al. Core needle biopsy of the thyroid: 2016 Consensus Statement and Recommendations from Korean Society of Thyroid Radiology. Korean J Radiol 2017;18:217–237. ArticlePubMedPMC
- 10. Jung CK, Min HS, Park HJ, Song DE, Kim JH, Park SY, et al. Pathology reporting of thyroid core needle biopsy: a proposal of the Korean Endocrine Pathology Thyroid Core Needle Biopsy Study Group. J Pathol Transl Med 2015;49:288–299. ArticlePubMedPMCPDF
- 11. Gharib H, Papini E, Garber JR, Duick DS, Harrell RM, Hegedus L, et al. American Association of Clinical Endocrinologists, American College of Endocrinology, and Associazione Medici Endocrinologi Medical Guidelines for clinical practice for the diagnosis and management of thyroid nodules: 2016 update. Endocr Pract 2016;22:622–639. ArticlePubMed
- 12. Yeon JS, Baek JH, Lim HK, Ha EJ, Kim JK, Song DE, et al. Thyroid nodules with initially nondiagnostic cytologic results: the role of core-needle biopsy. Radiology 2013;268:274–280. ArticlePubMed
- 13. Suh CH, Baek JH, Kim KW, Sung TY, Kim TY, Song DE, et al. The role of core-needle biopsy for thyroid nodules with initially nondiagnostic fine-needle aspiration results: a systematic review and meta-analysis. Endocr Pract 2016;22:679–688. ArticlePubMed
- 14. Choi SH, Baek JH, Lee JH, Choi YJ, Hong MJ, Song DE, et al. Thyroid nodules with initially non-diagnostic, fine-needle aspiration results: comparison of core-needle biopsy and repeated fine-needle aspiration. Eur Radiol 2014;24:2819–2826. ArticlePubMedPDF
- 15. Na DG, Kim JH, Sung JY, Baek JH, Jung KC, Lee H, et al. Core-needle biopsy is more useful than repeat fine-needle aspiration in thyroid nodules read as nondiagnostic or atypia of undetermined significance by the Bethesda system for reporting thyroid cytopathology. Thyroid 2012;22:468–475. ArticlePubMed
- 16. Choi YJ, Baek JH, Suh CH, Shim WH, Jeong B, Kim JK, et al. Core-needle biopsy versus repeat fine-needle aspiration for thyroid nodules initially read as atypia/follicular lesion of undetermined significance. Head Neck 2017;39:361–369. ArticlePubMed
- 17. Park KT, Ahn SH, Mo JH, Park YJ, Park DJ, Choi SI, et al. Role of core needle biopsy and ultrasonographic finding in management of indeterminate thyroid nodules. Head Neck 2011;33:160–165. ArticlePubMed
- 18. Suh CH, Baek JH, Lee JH, Choi YJ, Kim KW, Lee J, et al. The role of core-needle biopsy in the diagnosis of thyroid malignancy in 4580 patients with 4746 thyroid nodules: a systematic review and meta-analysis. Endocrine 2016;54:315–328. ArticlePubMedPDF
- 19. Paja M, del Cura JL, Zabala R, Corta I, Lizarraga A, Oleaga A, et al. Ultrasound-guided core-needle biopsy in thyroid nodules. A study of 676 consecutive cases with surgical correlation. Eur Radiol 2016;26:1–8. ArticlePubMedPDF
- 20. Sung JY, Na DG, Kim KS, Yoo H, Lee H, Kim JH, et al. Diagnostic accuracy of fine-needle aspiration versus core-needle biopsy for the diagnosis of thyroid malignancy in a clinical cohort. Eur Radiol 2012;22:1564–1572. ArticlePubMedPDF
- 21. Suh CH, Baek JH, Lee JH, Choi YJ, Kim JK, Sung TY, et al. The role of core-needle biopsy as a first-line diagnostic tool for initially detected thyroid nodules. Thyroid 2016;26:395–403. ArticlePubMed
- 22. Ha EJ, Baek JH, Lee JH, Kim JK, Song DE, Kim WB, et al. Core needle biopsy could reduce diagnostic surgery in patients with anaplastic thyroid cancer or thyroid lymphoma. Eur Radiol 2016;26:1031–1036. ArticlePubMedPDF
- 23. Ha EJ, Baek JH, Na DG, Kim JH, Kim JK, Min HS, et al. The role of core needle biopsy and its impact on surgical management in patients with medullary thyroid cancer: clinical experience at 3 medical institutions. AJNR Am J Neuroradiol 2015;36:1512–1517. ArticlePubMedPMC
- 24. Lee SH, Park GS, Jung SL, Kim MH, Bae JS, Lim DJ, et al. Core-needle biopsy for the preoperative diagnosis of follicular neoplasm in thyroid nodule screening: a validation study. Pathol Res Pract 2016;212:44–50.Article
- 25. Ha EJ, Baek JH, Lee JH, Lee HY, Song DE, Kim JK, et al. A focal marked hypoechogenicity within an isoechoic thyroid nodule: is it a focal malignancy or not? Acta Radiol 2015;56:814–819. ArticlePubMed
- 26. Ha EJ, Baek JH, Lee JH, Song DE, Kim JK, Shong YK, et al. Sonographically suspicious thyroid nodules with initially benign cytologic results: the role of a core needle biopsy. Thyroid 2013;23:703–708. ArticlePubMed
- 27. Ha EJ, Baek JH, Lee JH, Kim JK, Kim JK, Lim HK, et al. Core needle biopsy can minimise the non-diagnostic results and need for diagnostic surgery in patients with calcified thyroid nodules. Eur Radiol 2014;24:1403–1409. ArticlePubMedPDF
- 28. Trimboli P, Nasrollah N, Guidobaldi L, Taccogna S, Cicciarella Modica DD, Amendola S, et al. The use of core needle biopsy as first-line in diagnosis of thyroid nodules reduces false negative and inconclusive data reported by fine-needle aspiration. World J Surg Oncol 2014;12:61ArticlePubMedPMC
- 29. Baloch ZW, Cibas ES, Clark DP, Layfield LJ, Ljung BM, Pitman MB, et al. The National Cancer Institute Thyroid fine needle aspiration state of the science conference: a summation. Cytojournal 2008;5:6ArticlePubMedPMC
- 30. Ha EJ, Baek JH, Lee JH, Kim JK, Choi YJ, Sung TY, et al. Complications following US-guided core-needle biopsy for thyroid lesions: a retrospective study of 6,169 consecutive patients with 6,687 thyroid nodules. Eur Radiol 2017;27:1186–1194. ArticlePubMedPDF
- 31. Ha EJ, Baek JH, Lee JH. Ultrasonography-based thyroidal and perithyroidal anatomy and its clinical significance. Korean J Radiol 2015;16:749–766. ArticlePubMedPMC
References
Fig. 1

Examples of core needle biopsy (CNB) in thyroid nodules with initial non-diagnostic fine-needle aspiration cytology. (A) On the ultrasound findings of case 1, there is a well-defined hypoechoic solid nodule with rim calcification. (B) CNB of the nodule shows scanty cellular and sclerotic nodules (H&E stain, ×40). (C) A high-power view shows the typical histologic features of papillary carcinoma (H&E stain, ×1,000). (D) An ultrasound image from case 2 shows a 1.5-cm, ill-defined hypoechoic solid nodule with macrocalcifications. (E) CNB shows marked calcification sclerosis, and focally follicular proliferative lesions (H&E stain, ×40). (F) A high-power view of the follicular lesion shows the histologic findings of papillary carcinoma (H&E stain, ×1,000).

Fig. 2
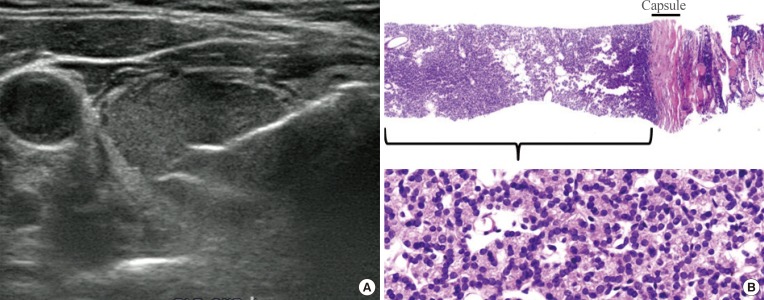
Current core needle biopsy technique. (A) The specimen notch shown in ultrasound should include the tumor tissue, tumor capsule, and adjacent normal parenchyma. (B) Histologic examination of the specimen shows the tumor tissue with a microfollicular growth pattern, tumor capsule, and surrounding thyroid tissue (H&E stain, ×40, upper; ×400, lower). This case should be diagnosed as a follicular neoplasm.

Table 1
![enm-32-407-i001.jpg]()
Consensus Statement and Recommendations on Thyroid CNB from the Korean Society of Thyroid Radiology [9]
Table 2
![enm-32-407-i002.jpg]()
Diagnostic Categories of Thyroid Core Needle Biopsy from the Korean Endocrine Pathology Thyroid Core Needle Biopsy Study Group [10]
Figure & Data
References
Citations
Citations to this article as recorded by 

- Parenchymal microcalcifications in the thyroid gland: Clinical significance and management strategy
Younghee Yim, Hye Sun Park, Jung Hwan Baek, Hyunju Yoo, Jin Yong Sung
Medicine.2023; 102(32): e34636. CrossRef - Strategies for Safe and Effective Core Needle Biopsy of Thyroid Nodules with Macrocalcification
Jae Ho Shin
International Journal of Thyroidology.2023; 16(2): 195. CrossRef - Utilidad de la biopsia con aguja gruesa ecoguiada en nódulos tiroideos con punción aspirativa con aguja fina no diagnóstica
R. Cortázar-García, M.D. Martín-Escalante, L. Robles-Cabeza, C. Martínez-Santos
Radiología.2022; 64(3): 195. CrossRef - Usefulness of ultrasound-guided core biopsy in thyroid nodules with inconclusive fine-needle aspiration biopsy findings
R. Cortázar-García, M.D. Martín-Escalante, L. Robles-Cabeza, C. Martínez-Santos
Radiología (English Edition).2022; 64(3): 195. CrossRef - Effectiveness of core needle biopsy in the diagnosis of thyroid lymphoma and anaplastic thyroid carcinoma: A systematic review and meta-analysis
Vincent Vander Poorten, Nathan Goedseels, Asterios Triantafyllou, Alvaro Sanabria, Paul M. Clement, Oded Cohen, Pawel Golusinski, Orlando Guntinas-Lichius, Cesare Piazza, Gregory W. Randolph, Alessandra Rinaldo, Ohad Ronen, Maria E. Cabanillas, Ashok R. S
Frontiers in Endocrinology.2022;[Epub] CrossRef - A Matched-Pair Analysis of Nuclear Morphologic Features Between Core Needle Biopsy and Surgical Specimen in Thyroid Tumors Using a Deep Learning Model
Faridul Haq, Andrey Bychkov, Chan Kwon Jung
Endocrine Pathology.2022; 33(4): 472. CrossRef - Intraoperative Assessment of High-Risk Thyroid Nodules Based on Electrical Impedance Measurements: A Feasibility Study
Jalil Beheshti Firoozabadi, Reihane Mahdavi, Khosro Shamsi, Hossein Ataee, Abdollah Shafiee, Hojat Ebrahiminik, Hossein Chegini, Parisa Hoseinpour, Afshin Moradi, Narges Yousefpour, Faeze Aghaei, Ali Fardoost, Alireza Ghelichli, Hadi Mokhtari Dowlatabad,
Diagnostics.2022; 12(12): 2950. CrossRef - Assessing the diagnostic performance of thyroid biopsy with recommendations for appropriate interpretation
Su Min Ha, Jung Hwan Baek, Dong Gyu Na, Chan-Kwon Jung, Chong Hyun Suh, Young Kee Shong, Tae Yon Sung, Dong Eun Song, Jeong Hyun Lee
Ultrasonography.2021; 40(2): 228. CrossRef - Update on the Evaluation of Thyroid Nodules
Victor J. Bernet, Ana-Maria Chindris
Journal of Nuclear Medicine.2021; 62(Supplement): 13S. CrossRef - Poorly Differentiated Thyroid Carcinoma: Single Centre Experience and Review of the Literature
Maria Bellini, Marco Biffoni, Renato Patrone, Maria Borcea, Maria Costanzo, Tiziana Garritano, Rossella Melcarne, Rosa Menditto, Alessio Metere, Chiara Scorziello, Marco Summa, Luca Ventrone, Vito D’Andrea, Laura Giacomelli
Journal of Clinical Medicine.2021; 10(22): 5258. CrossRef - Laser Ablation Versus Radiofrequency Ablation for Benign Non-Functioning Thyroid Nodules: Six-Month Results of a Randomized, Parallel, Open-Label, Trial (LARA Trial)
Roberto Cesareo, Claudio Maurizio Pacella, Valerio Pasqualini, Giuseppe Campagna, Mario Iozzino, Andrea Gallo, Angelo Lauria Pantano, Roberto Cianni, Claudio Pedone, Paolo Pozzilli, Chiara Taffon, Anna Crescenzi, Silvia Manfrini, Andrea Palermo
Thyroid.2020; 30(6): 847. CrossRef - 2019 Practice guidelines for thyroid core needle biopsy: a report of the Clinical Practice Guidelines Development Committee of the Korean Thyroid Association
Chan Kwon Jung, Jung Hwan Baek, Dong Gyu Na, Young Lyun Oh, Ka Hee Yi, Ho-Cheol Kang
Journal of Pathology and Translational Medicine.2020; 54(1): 64. CrossRef - Morphological and Molecular Assessment in Thyroid Cytology Using Cell-Capturing Scaffolds
Stefania Scarpino, Silvia Taccogna, Giuseppina Pepe, Enrico Papini, Martina D’Angelo, Federica Cascone, Daniele Nicoletti, Rinaldo Guglielmi, Andrea Palermo, Marcella Trombetta, Alberto Rainer, Chiara Taffon, Anna Crescenzi
Hormone and Metabolic Research.2020; 52(11): 803. CrossRef - European Thyroid Association Survey on Use of Minimally Invasive Techniques for Thyroid Nodules
Laszlo Hegedüs, Andrea Frasoldati, Roberto Negro, Enrico Papini
European Thyroid Journal.2020; 9(4): 194. CrossRef - Malignancy risk of initially benign thyroid nodules: validation with various Thyroid Imaging Reporting and Data System guidelines
Su Min Ha, Jung Hwan Baek, Young Jun Choi, Sae Rom Chung, Tae Yon Sung, Tae Yong Kim, Jeong Hyun Lee
European Radiology.2019; 29(1): 133. CrossRef - Tumor Volume Doubling Time in Active Surveillance of Papillary Thyroid Carcinoma
Hye-Seon Oh, Hyemi Kwon, Eyun Song, Min Ji Jeon, Tae Yong Kim, Jeong Hyun Lee, Won Bae Kim, Young Kee Shong, Ki-Wook Chung, Jung Hwan Baek, Won Gu Kim
Thyroid.2019; 29(5): 642. CrossRef - Risk of Malignancy According to the Sub-classification of Atypia of Undetermined Significance and Suspicious Follicular Neoplasm Categories in Thyroid Core Needle Biopsies
Sae Rom Chung, Jung Hwan Baek, Jeong Hyun Lee, Yu-Mi Lee, Tae-Yon Sung, Ki-Wook Chung, Suck Joon Hong, Min Ji Jeon, Tae Yong Kim, Young Kee Shong, Won Bae Kim, Won Gu Kim, Dong Eun Song
Endocrine Pathology.2019; 30(2): 146. CrossRef - Does Radiofrequency Ablation Induce Neoplastic Changes in Benign Thyroid Nodules: A Preliminary Study
Su Min Ha, Jun Young Shin, Jung Hwan Baek, Dong Eun Song, Sae Rom Chung, Young Jun Choi, Jeong Hyun Lee
Endocrinology and Metabolism.2019; 34(2): 169. CrossRef - Response: Long-Term Outcomes Following Thermal Ablation of Benign Thyroid Nodules as an Alternative to Surgery: The Importance of Controlling Regrowth (Endocrinol Metab 2019;34:117–23, Jung Suk Sim et al.)
Jung Suk Sim, Jung Hwan Baek
Endocrinology and Metabolism.2019; 34(3): 325. CrossRef - A Study on Head and Neck Malignant Lymphoma Diagnosed by Core Needle Biopsy
Keisuke Yamamoto, Tsuyoshi Okuni, Makoto Kurose, Akira Yorozu, Kizuku Owada, Ryoto Yajima, Ayumi Takahashi, Kazufumi Obata, Atsushi Kondo, Kenichi Takano
Practica Oto-Rhino-Laryngologica.2019; 112(9): 609. CrossRef - The Role of Core Needle Biopsy for the Evaluation of Thyroid Nodules with Suspicious Ultrasound Features
Sae Rom Chung, Jung Hwan Baek, Young Jun Choi, Tae-Yon Sung, Dong Eun Song, Tae Yong Kim, Jeong Hyun Lee
Korean Journal of Radiology.2019; 20(1): 158. CrossRef - Safety and Efficacy of Radiofrequency Ablation for Nonfunctioning Benign Thyroid Nodules in Children and Adolescents in 14 Patients over a 10-Year Period
Min Ji Hong, Jin Yong Sung, Jung Hwan Baek, Mi Su Je, Dong Whan Choi, Hyunju Yoo, Sae Jeong Yang, Sang Yu Nam, Eun Young Yoo
Journal of Vascular and Interventional Radiology.2019; 30(6): 900. CrossRef - Reliability of core needle biopsy as a second-line procedure in thyroid nodules with an indeterminate fine-needle aspiration report: a systematic review and meta-analysis
Pierpaolo Trimboli, Luca Giovanella
Ultrasonography.2018; 37(2): 121. CrossRef - The Significance of Having an Excellent Patient's Comfort with Thyroid Core Needle Biopsy
Pierpaolo Trimboli, Luca Giovanella
Endocrinology and Metabolism.2018; 33(1): 53. CrossRef - Ultrasonographic Echogenicity and Histopathologic Correlation of Thyroid Nodules in Core Needle Biopsy Specimens
Ji-hoon Kim, Dong Gyu Na, Hunkyung Lee
Korean Journal of Radiology.2018; 19(4): 673. CrossRef - A Comparison of Ultrasound-Guided Fine Needle Aspiration versus Core Needle Biopsy for Thyroid Nodules: Pain, Tolerability, and Complications
Eun Ji Jeong, Sae Rom Chung, Jung Hwan Baek, Young Jun Choi, Jae Kyun Kim, Jeong Hyun Lee
Endocrinology and Metabolism.2018; 33(1): 114. CrossRef

 KES
KES
 PubReader
PubReader Cite
Cite



