Articles
- Page Path
- HOME > Endocrinol Metab > Volume 38(6); 2023 > Article
-
Review ArticleCalcium & bone metabolism Acromegaly and Bone: An Update
 Keypoint
Keypoint
Since acromegaly's association with increased vertebral fracture risk was discovered in 2006, substantial research has validated this link, highlighting its epidemiological and clinical significance. This review integrates recent findings with initial observations to offer an updated view on how acromegaly affects bone health. It covers the clinical impacts and underlying mechanisms, and explores prevention and treatment approaches. -
Andrea Giustina

-
Endocrinology and Metabolism 2023;38(6):655-666.
DOI: https://doi.org/10.3803/EnM.2023.601
Published online: December 22, 2023
Institute of Endocrine and Metabolic Sciences, San Raffaele Hospital, San Raffaele Vita Salute University, Milan, Italy
- Corresponding author: Andrea Giustina. Division of Endocrinology, San Raffaele Hospital, San Raffaele Vita Salute University, via Olgettina 60, Milano 20132, Italy Tel: +39-0226435136, Fax: +39-0226435136, E-mail: giustina.andrea@hsr.it
• Received: October 28, 2023 • Revised: November 23, 2023 • Accepted: November 30, 2023
Copyright © 2023 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
- Since our discovery in 2006 that acromegaly is associated with an increased risk of vertebral fractures, many authors have confirmed this finding in both cross-sectional and prospective studies. Due to the high epidemiological and clinical impact of this newly discovered comorbidity of acromegaly, this topic has progressively become more important and prominent over the years, and the pertinent literature has been enriched by new findings on the pathophysiology and treatment. The aim of this narrative review was to discuss these novel findings, integrating them with the seminal observations, in order to give the reader an updated view of how the field of acromegaly and bone is developing, from strong clinical observations to a mechanistic understanding and possible prevention and treatment.
- In acromegaly, uncontrolled and excessive growth hormone (GH) production [1], usually caused by a pituitary adenoma [2], results in increased levels of insulin-like growth factor 1 (IGF-1) [3] and triggers the occurrence of systemic complications, mainly due to diagnostic delay with consequent impairment of quality of life and survival [4]. In fact, besides abnormal growth of bone and soft tissue, which determines the classical features of this condition, in patients with acromegaly, an increased prevalence of complications—mainly cardio-metabolic and respiratory—is observed [5-7]. The treatment of acromegaly is often challenging, and GH and/or IGF-1 levels cannot be normalized in some cases [8,9] even with multidisciplinary management at pituitary tumors centers of excellence (PTCOEs) [10,11]. In fact, patients diagnosed too late may often present with large pituitary adenomas that cannot be entirely removed by neurosurgeons [12], and subsequent medical and radiation treatment may not achieve optimal biochemical and tumor mass control [13]. In patients in whom biochemical control is not obtained, but also sometimes in apparently controlled patients, clinical disease progression may be observed [14]. Clearly, besides optimization of treatment of the underlying disease following the most current guidelines [15], early diagnosis, appropriate follow-up, and the prevention and treatment of acromegaly complications constitute a key strategy for preserving quality of life and reducing the mortality rate of affected patients [16].
- Interestingly, GH and IGF-1 are among the most relevant hormonal modulators of skeletal metabolism [17], causing an increase in bone turnover when produced in excess [18] and thereby leading to clinically relevant structural damage both at cortical and trabecular bone levels [19]. Therefore, in acromegaly an increase in fracture risk, particularly at the vertebral level [20], has been progressively and consistently reported in the last 15 years [21,22], and this now represents one of the most relevant systemic complications of the disease [23]. In fact, since the first report of excess morphometric fractures in postmenopausal women with acromegaly [21], many subsequent papers—including several from our group [24,25]—have confirmed, extended, analyzed the underlying mechanism(s) and possible protective effect of treatment, making this one of the hottest topics in the area of acromegaly management [26]. Due to the current clinical impact of bone involvement in acromegaly and its increasingly wide literature coverage, it was felt necessary to critically review and update the emerging evidence on the pathophysiology, clinical history and presentation, diagnosis and treatment of acromegaly-related osteopathy through this paper. More recent methods for bone evaluation will also be preliminarily and briefly reviewed in this paper, as they have been demonstrated to show the potential of leading to a better understanding of the pathophysiology and possibly prediction and prevention of skeletal damage in acromegaly.
INTRODUCTION
- It is now widely accepted that in most secondary forms of osteoporosis [27-30], and specifically in pituitary-driven skeletal diseases [31-34], bone mineral density (BMD) measured with dual-energy X-ray absorptiometry (DXA) may not be a reliable predictor of fracture risk as opposed to postmenopausal osteoporosis [35]. The current thinking attributes this discrepancy to a prevalent impact of pituitary and other hormone hyperproduction on bone quality with respect to bone quantity [36]. Therefore, new tools such as vertebral morphometry for detecting subclinical or previously undiagnosed vertebral fractures (VFs) and methods for assessing bone quality have become fundamental in the modern approach to bone comorbidities in patients with pituitary adenomas, as in those with acromegaly [37]. These methods are briefly reviewed below.
- Assessment of morphometric vertebral fractures
- VFs have emerged in the last decades as a hallmark of skeletal fragility and are associated with a high risk of subsequent clinical fractures, increased mortality, and decreased quality of life [38] in the general population. Since back pain is a very common symptom and is generally self-limiting after a VF, the clinical diagnosis, particularly in the case of not severe VFs may be overlooked. Therefore, a proactive radiological and morphometric diagnosis is currently the gold standard for investigating the prevalence and incidence of VFs in high-risk groups and clinical trials [39]. Morphometric VFs are diagnosed on thoracic-lumbar spine (LS) imaging (X-rays or magnetic resonance imaging) and on DXA through qualitative and quantitative vertebral shape assessment [40]. The classification of VFs as mild, moderate, or severe is based, according to Genant [41], on a height ratio decrease of 20%–25%, 25%–40%, and more than 40%, respectively. Very recently, in order to minimize radiological and repeated x-ray exposure of patients, an opportunistic approach to morphometry based on the evaluation of chest imaging performed on patients for other reasons, which we originally proposed [30,34] has become the preferred approach, particularly for research purposes but also in clinical practice.
- Assessment of bone quality
- Novel tools for assessing bone quality and resistance have recently emerged as having added value for predicting fractures in various forms of osteoporosis [42,43]. So far, some of these tools have also been tested as fracture predictors in osteopathies driven by pituitary hyperfunction [44] and specifically in acromegaly, since they could possibly overcome the abovementioned limitations of areal DXA evaluation in patients affected by this disease [45]. Briefly, bone quality in acromegaly has been assessed by high-resolution peripheral quantitative computed tomography (HR-pQCT), a three-dimensional noninvasive imaging technique that scans the extremities and can assess volumetric bone density and microarchitecture of cortical and cancellous bone [46]. HR-pQCT is still mostly used for research because it is expensive and not widely available [37]. Interesting results have also been obtained with high-resolution cone-beam computed tomography, which rapidly scans the extremities with a larger field of view but reduced image contrast because of artifacts, as compared to HR-pQCT [47]. Moreover, the most popular and routinely used method for assessing bone quality is undoubtedly the trabecular bone score (TBS), which is a parameter directly obtained from LS DXA scans that significantly correlates with direct assessments of bone microarchitecture, predicting fragility fracture risk in several conditions [45]. Finally, bone microindentation, a micro-invasive method that measures bone material strength by a probe on the tibial surface, has also been tested in acromegaly [48].
METHODS FOR THE EVALUATION OF BONE HEALTH
- GH may have direct effects on skeletal metabolism by stimulating the local synthesis of IGF-1 in bone [17]; however, this predominantly occurs under endogenous parathyroid hormone control [49]. Inasmuch, GH may also indirectly affect bone by increasing the level of circulating IGF-1 [18]. Overall, despite GH being by definition a bone anabolic hormone [49], its persistent elevation and that of IGF-1 result in an increased bone turnover with deterioration of bone status [50].
- Direct GH effects on bone
- GH stimulates osteoblastogenesis as opposed to adipogenesis [51]. In fact, the expression of fetal antigen-1 is downregulated by GH; this also increases the expression of bone morphogenetic proteins, which together with Wnt play a positive role in osteoblastogenesis [52]. GH also directly stimulates the carboxylation of osteocalcin, which is an anabolic bone marker, and the production and accumulation of osteocalcin in the bone matrix [17].
- Indirect GH effects on bone
- Most of the systemic effects of GH are mediated by the GH-stimulated hepatic production of IGF-1 with variable efficiency based on different polymorphisms of the GH receptor [53], which seems to be key for cortical bone health, while locally produced IGF-I in the skeleton may be more important for trabecular bone structure [49]. IGF-I may also favor osteoblastogenesis through the stabilization of beta-catenin [54], a key component of the Wnt canonical signaling pathway [49]. Expression of the receptor activator of nuclear factor κB ligand (RANKL) is stimulated by IGF-1, which through this pathway decreases the expression of osteoprotegerin, the RANKL decoy receptor, which regulates osteoclastogenesis [55]. Finally, IGF-1 also stimulates the activity and expression of 1α-hydroxylase increasing serum levels of 1,25-dihydroxyvitamin D [56] concomitantly with GH-mediated effects on vitamin D-binding protein (VDBP) production [57].
PATHOPHYSIOLOGY OF BONE FRAGILITY IN ACROMEGALY
- Conventional tools
- One of the historical problems in the field of the clinical impact of GH excess on bone has been the use of conventional tools for assessing bone health. In fact, as a result of evaluating traditional bone markers and bone densitometry through DXA, acromegaly was barely included among the causes of secondary osteoporosis due to the inconsistency in the evidence of altered parameters in different studies [58,59]. Based on these misleading data and on the pathophysiological concept of GH as an anabolic hormone [49] it is unsurprising that the paradoxical possibility that acromegaly patients could even be protected against osteoporosis was initially considered [60]. Therefore, we will rapidly review the historical evidence regarding classical bone turnover markers (BTMs) and DXA in order to highlight the limitations that were overcome by introducing novel methods of skeletal assessment in this area, the results of which will be reviewed in the second part of this section.
- Bone turnover has been shown to be increased in acromegaly in a meta-analysis finding significant differences in standardized mean levels of classical BTMs of bone formation and resorption between acromegaly and control subjects [50]. Moreover, various studies on BTMs supported the notion of the occurrence of an uncoupling of bone turnover in acromegaly with an excess resorption versus bone formation [61]. However, likely due to huge inter-individual variability in BTM assays [61], despite these quite consistent findings with important pathophysiological insights, no practical implications were inferred from the above data in clinical practice. As well, the introduction of possibly novel BTMs has also not had a relevant clinical impact so far. In fact, low sclerostin and high Dickkopf-1 (DKK-1) levels were found to be associated with altered bone structure in acromegaly [62-64] and inconsistently with either GH/IGF-1 levels or with VF risk [65,66].
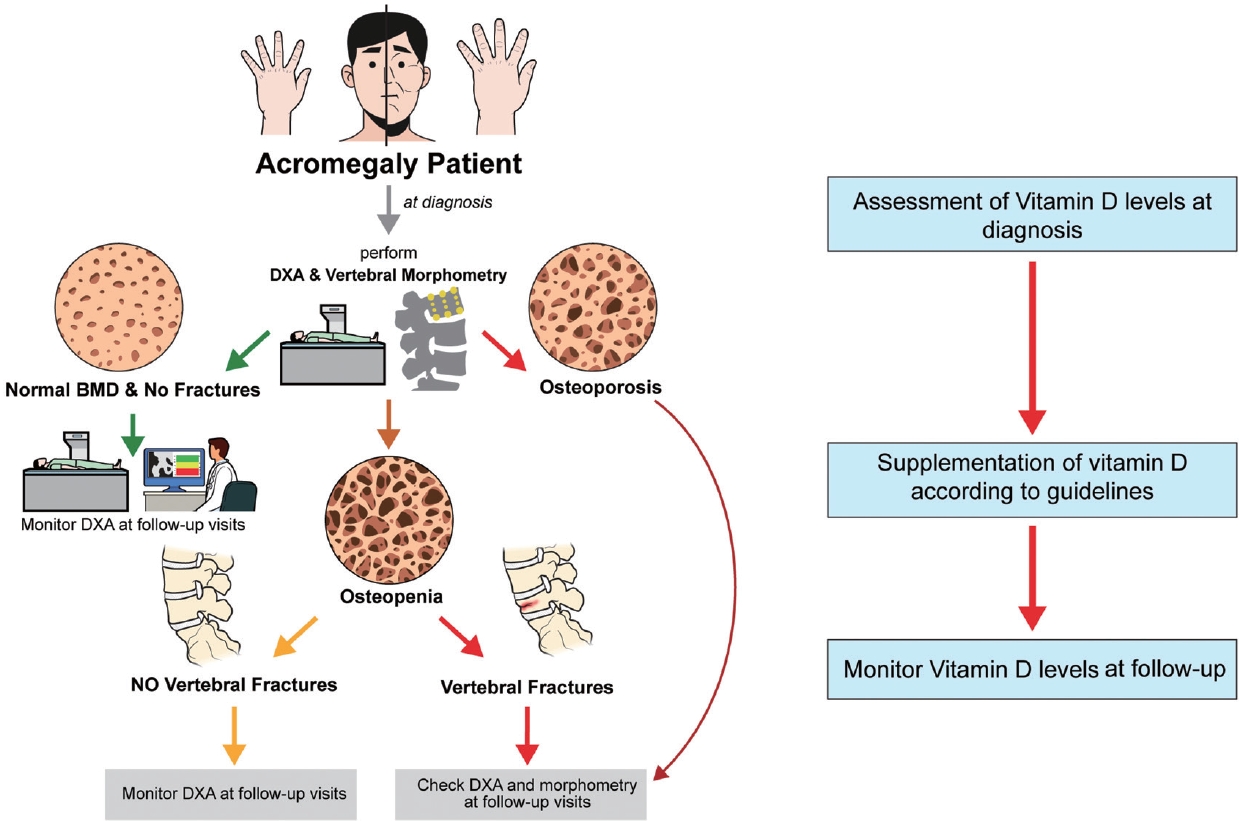
- DXA BMD of the LS may be overestimated in acromegaly due to the very frequent concomitant joint degenerative disorders, which are characterized by osteophytes and facet joint hypertrophy in the presence of bone enlargement [18]. BMD in the LS was reported to be decreased, normal, or increased in acromegaly [58,59,66], whereas it was reported to be increased in the total hip and femoral neck [67]. This reflects what has already been reported in other endocrinopathies, in which it is well known that DXA BMD does not predict fracture risk in a reliable fashion [68-70]. In biochemically controlled patients, VF progression was not found to be associated with BMD results or with prospective changes in BMD [71]. However, a decrease in femoral neck BMD was reported in acromegaly patients with incident VFs [72]. Therefore, DXA BMD testing may still have a role in fracture prediction in such patients at baseline and during follow-up (Fig. 1) [72].
- Assessment of bone quality in acromegaly
- Several studies have shown lower TBS in patients with acromegaly as compared to control subjects in the absence of significant difference in BMD [73], regardless of the presence of VFs [74] and not necessarily influenced by disease activity [75,76]. HR-pQCT bone microarchitecture data in acromegaly were significantly associated with disease activity, while altered microstructure was also found in eugonadal patients [46]. Interestingly, recent HR-pQCT studies also found increased cortical porosity and decreased cortical bone strength [77]. On the other hand, CBCT studies reported significantly lower bone volume/trabecular volume ratio, greater mean trabecular separation, and cortical porosity in fractured versus. non-fractured acromegaly patients [47]. Bone microindentation studies also found lower bone material strength index values in biochemically controlled patients with acromegaly as compared to healthy controls [48]. In fact, this finding may be consistent with persistently altered material properties of cortical bone even after biochemical normalization. Interestingly, hip structure analysis recently also revealed a persistently significant decrease in cortical thickness, which may imply endocortical trabecularization and may potentially explain the increased incident VF risk following effective treatment [78]. In contrast VFs in active acromegaly may be caused by impaired trabecular structure, which may improve after treatment [78].
ASSESSMENT OF BONE HEALTH IN ACROMEGALY
Bone turnover markers
DXA bone mineral density
- Morphometric vertebral fractures
- Since our seminal observation in 2005 [21], many authors from different areas of the world have confirmed that thoracic and LS fragility VFs constitute one of the most epidemiologically and clinically relevant comorbidities of acromegaly, with a reported prevalence of up to 60% of patients (increased 3- to 8-fold as compared to the general population) [79-81], affecting slightly more males than females, and being associated with biochemical activity of the disease but not with DXA BMD [21-23].
- In a 3-year controlled perspective study 42% of acromegaly versus 3.8% of non-acromegaly subjects were found to be affected by incident VFs [72]. VF progression was also frequently reported in biochemically normalized patients, particularly in males with more than two VFs at enrollment [71].
- Hypogonadism, a decrease in femoral neck BMD, and prevalent VFs at the beginning of follow-up may predict incident VF occurrence only in patients with controlled/cured acromegaly, whereas in patients with active acromegaly, the incident fracture risk was only significantly associated with active disease duration [72].
- The incidence of VFs was also significantly increased in patients who were enrolled with active disease versus those with controlled acromegaly at study entry [72]. Therefore, biochemical control appears to be an important protective factor for skeletal damage. VFs may occur early in the natural disease history [25] and therefore normalization of biochemical activity should be mandatorily achieved as quickly as possible after the diagnosis, although this may not be enough to avoid fractures due to the known issue of diagnostic delay [24,82]. Additionally, concomitant diabetes mellitus, another common comorbidity of acromegaly [83], as well as a cause of bone fragility per se [84], was reported to increase the risk of fracture in active disease [85]. Moreover, in patients with active acromegaly on different medical treatments, incident VFs were more frequent in those with active disease independently of the type of therapy [86].
- Recently, we showed that prevalence of radiological thoracic VFs was associated with elevated presurgical random GH levels in patients recently diagnosed with acromegaly [25]. Moreover, another recent study suggested that a diagnostic delay >10 years may be a quite strong predictor of incident VFs [24].
- Therefore, more recent guidelines suggest that vertebral morphometry should be performed at diagnosis in all patients with acromegaly (Fig. 1) [4]. Morphometry should also be repeated during follow-up, particularly in patients with active disease or untreated hypogonadism and densitometric osteoporosis or prevalent VFs (Fig. 1) [72].
- Clinical fractures in acromegaly
- Recently, in a Korean population-based cohort prospective study including more than 1,700 patients with acromegaly followed up for more than 8 years, the risk of clinical vertebral and hip fractures was evaluated in comparison with 8,885 age- and sex-matched controls [87]. Interestingly, patients with acromegaly were found to have significantly higher risks of clinical vertebral (hazard ratio [HR], 2.09) and hip (HR, 2.52) fractures than controls. As reported for morphometric VFs, clinical VFs also occurred early during follow-up [87]. Interestingly, the multivariable-adjusted HRs were higher for clinical hip fractures (ranging from 2.29 to 3.36) than for VFs (from 1.69 to 2.70) [87].
CLINICAL PRESENTATION OF BONE INVOLVEMENT IN ACROMEGALY
Prevalence
Incidence
Predictors
- Estrogens in postmenopausal women and androgens in hypogonadal males
- Both postmenopausal females [21] and males [22] with acromegaly have been reported to be at increased risk of VFs. In males, hypogonadism has been reported to have a negative impact on fracture occurrence, although to a lesser degree than active disease duration [22]. Therefore, although specific data are missing, based on bone protective effects of sex hormones in postmenopausal and male osteoporosis [88,89] either estrogen or androgen replacement should be considered in hypogonadal subjects if not contraindicated, particularly in case of subjects already bearing a prevalent morphometric VF, which—as mentioned above—is a strong predictor of subsequent fractures. In this light, the possible IGF-1-lowering effect of estrogens and estrogen receptor modulators [90] may also facilitate the optimization of biochemical control, which may synergize with the correction of hypogonadism in lowering fracture risk.
- Vitamin D and anti-osteoporotic agents
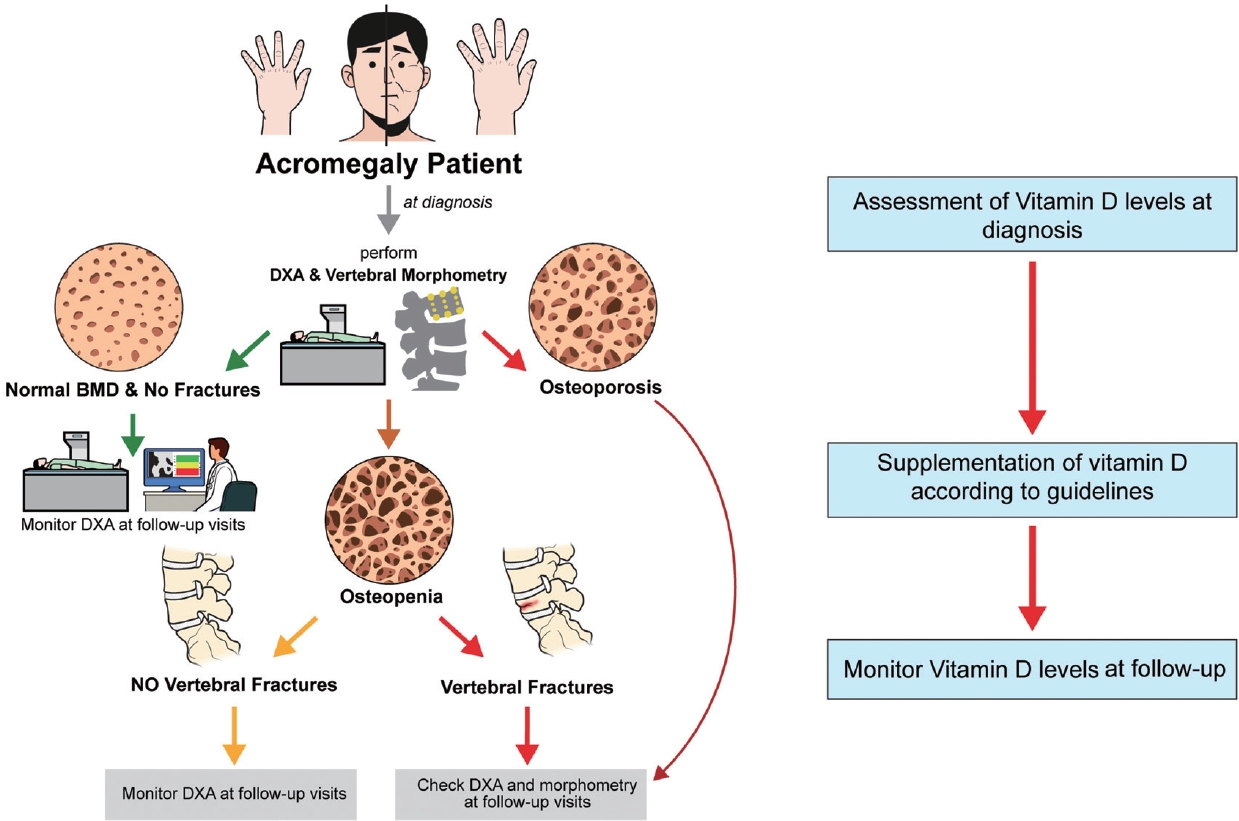
- Vitamin D is a crucial hormone with skeletal and extraskeletal effects [91]. The most relevant skeletal action of vitamin D is an increase in intestinal calcium absorption and bone mineralization [92]. Its deficiency, when severe, may cause rickets in children and osteomalacia in adults. Less severe deficiency of vitamin D increases osteoporosis and fracture risk in the general population [93].
- Active acromegaly and treatment with somatostatin receptor ligands (SRLs; as a possible result of induced malabsorption of lipophilic substances [94]) have been reported to be risk factors for vitamin D deficiency [95,96], with significant positive correlation between IGF-1 and 25 hydroxy-vitamin D (25OHD) levels in biochemically active patients [97]. Additionally, the reported increase in acromegaly of VDBP [57] is likely to cause a decrease in free vitamin D levels. Moreover, in many countries where food is not supplemented with vitamin D [98] a high prevalence of hypovitaminosis D in the general population is observed, particularly in the elderly [99]. Finally, whereas supplementation with vitamin D, which is in general based on oral cholecalciferol [100] administration, in the whole population has not been proven to be protective against bone events [101] it has been found to have positive effects on bone in subjects with poor vitamin D status and high fracture risk [102] such as those with excess glucocorticoids [103].
- Recently, in a prospective study, active acromegaly patients showed a lower response in terms of serum free vitamin D levels to a single dose (150,000 IU) of oral cholecalciferol (and delta 25OHD was negatively correlated with biochemical activity of the disease) as compared to similar number of controls matched for age, sex, and body mass index [104]. These data suggest that active acromegaly may cause a state of relative vitamin D resistance, as already observed in Cushing’s disease [105].
- Moreover, also very recently, in an Italian longitudinal, retrospective, and multicenter study, acromegaly patients supplemented with cholecalciferol based on general population guidelines [100] (based on clinical judgment at a median dose of 8,500 IU/week) were reported to have a lower incidence of VFs (14.3%) after a median follow-up of 94 months than untreated patients [26]. The final median 25OHD level was significantly lower in patients with incident VFs (28.6 ng/mL) than in patients without incident VFs (34.2 ng/mL). The fracture-protective effect of cholecalciferol was confirmed in logistic regression analysis (odds ratio, 0.16; 95% CI, 0.03 to 0.79) [26]. These findings suggest that at least in regions with a high prevalence of vitamin D deficiency, such as Italy [106], 25OHD should be measured in all acromegaly patients and in case of poor vitamin D status, supplementation with a standard dose of cholecalciferol should be started (Fig. 1).
- As a matter of fact, this study was the first to show at least an association between the long-term use of standard doses [100] of a bone-active hormone such as cholecalciferol and a reduction in incident VFs, in acromegaly [26]. This is good news, since methods of preventing VFs in acromegaly—besides improving biochemical control (see below)—remain an open issue.
- In fact, evidence on potential effects of bone-sparing agents in acromegaly-related osteopathy is limited (the abovementioned study on cholecalciferol was carried out in patients untreated with bone-active agents [26]) and based on retrospective observational data [107]. It was hypothesized that since the biochemical mechanism underlying the increased risk of fractures in acromegaly is suspected to be an increase in bone turnover, the use of anti-resorptive drugs could have been the most beneficial treatment [108]. However, it was recently retrospectively observed that any type of bone-sparing therapy (either anti-resorptive with bisphosphonates and denosumab or anabolic with teriparatide) was able to lower the incidence of VFs only in biochemically active patients [107]. Nevertheless, despite the lack of specific evidence and guidelines on anti-osteoporotic treatment in acromegaly-related osteopathy, it has to be noted that if a bone-active treatment is felt to be indicated in a patient with acromegaly, a combination with cholecalciferol should be used, as in the general population [109].
- Moreover, since cholecalciferol administration was also reported to be safe in patients with acromegaly [26], as in the general population [100] it could be useful to include in guidelines that vitamin D assessment and administration may be managed at every endocrine center (Fig. 1) while the indication and choice of specific anti-osteoporotic therapies should be responsibility of the bone expert within the PTCOE [110,111].
- Finally, since GH deficiency has been found to be a strong risk factor for prevalent [112] and incident [113] VFs, the use of GH replacement therapy should be considered in patients with acromegaly and prevalent VFs made GH-deficient by treatment [114].
- Acromegaly treatment
- Medical therapy has become an important option over the years as the second- or third-line treatment of acromegaly, but can also be used in some instances as primary therapy [115-117]. First-generation SRLs such as octreotide or lanreotide are generally used as first-line medical therapy, particularly in Europe [118,119], whereas high doses of octreotide or lanreotide [120,121], pegvisomant [122,123], or a second-generation SRL (pasireotide) [124] according to guidelines [125] may be used as second-line therapy in patients partially or not at all controlled with first-generation SRLs [126]. The medical therapy of acromegaly has been reported to have positive impact on the cardiorespiratory complications of acromegaly [127], likely via improved biochemical control [128]. Recently, the impact of medical therapies for acromegaly on skeletal fragility has also been analyzed in depth [129,130].
- In cross-sectional [86] and prospective studies [129], obtaining biochemical disease control by either first-generation SRLs or pegvisomant was linked to a decreased VF risk. Recently, it was reported that patients treated with pasireotide showed decreased incidence of VFs as compared to those treated with pegvisomant, independently of IGF-1 values and the degree of biochemical control [130], suggesting that pasireotide may potentially directly and positively influence bone metabolism by decreasing both GH and systemic and local IGF-1 levels [130].
- On the other hand, the decrease in incident VFs under treatment with pegvisomant was closely related to biochemical disease control [129]. The observed gap in VF prevention in favor of pasireotide may be due to possible detrimental effects of high circulating GH on bone due to the mechanism of action of pegvisomant [131].
- Overall, although the acromegaly treatment-specific effects on skeletal end-points in acromegaly are not yet fully elucidated, the data seem to suggest that improvement of bone status may be mediated by IGF-1 reduction/normalization under pegvisomant whereas it could be partly independent from systemic IGF-1 under pasireotide. Therefore, since second-line medical therapy with pasireotide may be more effective in preventing VFs than pegvisomant, it can be inferred that bone status may also influence the choice of treatment, as already proposed for tumor dimensions and diabetes mellitus [125]. In fact, it can be hypothesized that pasireotide could preferentially be used in subjects with persistently active disease and high risk of VFs.
TREATMENT OF ACROMEGALY-RELATED OSTEOPATHY
- Due to the insufficient fracture predictivity of DXA BMD, vertebral morphometry, possibly in combination with bone quality assessment, currently appears mandatory in the evaluation of bone status in patients with acromegaly (Fig. 1) and due to the high epidemiological relevance of VFs in acromegaly, guidelines with these recommendations should be implemented not only in PTCOEs at diagnosis and follow-up, particularly in the case of patients with prevalent VFs. The involvement of both trabecular and cortical bone in the detrimental skeletal effect of excess GH and IGF-1 has been reported; thus, explaining the recently reported increased prevalence of both VFs and clinical hip fractures. Moreover, cortical bone damage could also explain the persistence of increased fracture risk in biochemically controlled acromegaly. The use of cholecalciferol has been associated with decreased risk of incident VFs in acromegaly [26], whereas modernly defined tight biochemical control [131] and the use of pasireotide in active acromegaly patients may also be important protective factors against skeletal events in this clinical setting [130].
CONCLUSIONS
-
CONFLICTS OF INTEREST
Andrea Giustina is consultant for Abiogen, Ipsen, Pizer, and Recordati.
Article information
Fig. 1.Proposed diagnostic and follow-up approach to bone comorbidities in acromegaly. DXA, dual-energy X-ray absorptiometry; BMD, bone mineral density.

- 1. Giustina A, Veldhuis JD. Pathophysiology of the neuroregulation of growth hormone secretion in experimental animals and the human. Endocr Rev 1998;19:717–97.ArticlePubMed
- 2. Melmed S, Kaiser UB, Lopes MB, Bertherat J, Syro LV, Raverot G, et al. Clinical biology of the pituitary adenoma. Endocr Rev 2022;43:1003–37.ArticlePubMedPMCPDF
- 3. Colao A, Grasso LF, Giustina A, Melmed S, Chanson P, Pereira AM, et al. Acromegaly. Nat Rev Dis Primers 2019;5:20.ArticlePubMedPDF
- 4. Giustina A, Barkan A, Beckers A, Biermasz N, Biller BM, Boguszewski C, et al. A consensus on the diagnosis and treatment of acromegaly comorbidities: an update. J Clin Endocrinol Metab 2020;105:dgz096.PubMed
- 5. Frara S, Maffezzoni F, Mazziotti G, Giustina A. Current and emerging aspects of diabetes mellitus in acromegaly. Trends Endocrinol Metab 2016;27:470–83.ArticlePubMed
- 6. Giustina A, Boni E, Romanelli G, Grassi V, Giustina G. Cardiopulmonary performance during exercise in acromegaly, and the effects of acute suppression of growth hormone hypersecretion with octreotide. Am J Cardiol 1995;75:1042–7.ArticlePubMed
- 7. Davi’ MV, Dalle Carbonare L, Giustina A, Ferrari M, Frigo A, Lo Cascio V, et al. Sleep apnoea syndrome is highly prevalent in acromegaly and only partially reversible after biochemical control of the disease. Eur J Endocrinol 2008;159:533–40.ArticlePubMed
- 8. Giustina A, di Filippo L, Uygur MM, Frara S. Modern approach to resistant acromegaly. Endocrine 2023;80:303–7.ArticlePubMedPDF
- 9. Gola M, Bonadonna S, Mazziotti G, Amato G, Giustina A. Resistance to somatostatin analogs in acromegaly: an evolving concept? J Endocrinol Invest 2006;29:86–93.ArticlePubMedPDF
- 10. Giustina A, Barkhoudarian G, Beckers A, Ben-Shlomo A, Biermasz N, Biller B, et al. Multidisciplinary management of acromegaly: a consensus. Rev Endocr Metab Disord 2020;21:667–78.PubMedPMC
- 11. Casanueva FF, Barkan AL, Buchfelder M, Klibanski A, Laws ER, Loeffler JS, et al. Criteria for the definition of pituitary tumor centers of excellence (PTCOE): a Pituitary Society statement. Pituitary 2017;20:489–98.ArticlePubMedPMCPDF
- 12. Giustina A, Bronstein MD, Chanson P, Petersenn S, Casanueva FF, Sert C, et al. International multicenter validation study of the SAGIT instrument in acromegaly. J Clin Endocrinol Metab 2021;106:3555–68.ArticlePubMedPDF
- 13. Mazziotti G, Giustina A. Effects of lanreotide SR and Autogel on tumor mass in patients with acromegaly: a systematic review. Pituitary 2010;13:60–7.ArticlePubMedPDF
- 14. Frara S, Maffezzoni F, Mazziotti G, Giustina A. The modern criteria for medical management of acromegaly. Prog Mol Biol Transl Sci 2016;138:63–83.ArticlePubMed
- 15. Fleseriu M, Biller BM, Freda PU, Gadelha MR, Giustina A, Katznelson L, et al. A Pituitary Society update to acromegaly management guidelines. Pituitary 2021;24:1–13.ArticlePubMedPMCPDF
- 16. Melmed S, Casanueva FF, Klibanski A, Bronstein MD, Chanson P, Lamberts SW, et al. A consensus on the diagnosis and treatment of acromegaly complications. Pituitary 2013;16:294–302.ArticlePubMedPMC
- 17. Giustina A, Mazziotti G, Canalis E. Growth hormone, insulin-like growth factors, and the skeleton. Endocr Rev 2008;29:535–59.ArticlePubMedPMC
- 18. Mazziotti G, Frara S, Giustina A. Pituitary diseases and bone. Endocr Rev 2018;39:440–88.ArticlePubMed
- 19. Ribeiro de Moura C, Campos Lopes S, Monteiro AM. Determinants of skeletal fragility in acromegaly: a systematic review and meta-analysis. Pituitary 2022;25:780–94.ArticlePubMedPDF
- 20. Giustina A. Acromegaly and vertebral fractures: facts and questions. Trends Endocrinol Metab 2020;31:274–5.ArticlePubMed
- 21. Bonadonna S, Mazziotti G, Nuzzo M, Bianchi A, Fusco A, De Marinis L, et al. Increased prevalence of radiological spinal deformities in active acromegaly: a cross-sectional study in postmenopausal women. J Bone Miner Res 2005;20:1837–44.ArticlePubMed
- 22. Mazziotti G, Bianchi A, Bonadonna S, Cimino V, Patelli I, Fusco A, et al. Prevalence of vertebral fractures in men with acromegaly. J Clin Endocrinol Metab 2008;93:4649–55.ArticlePubMed
- 23. Mazziotti G, Maffezzoni F, Frara S, Giustina A. Acromegalic osteopathy. Pituitary 2017;20:63–9.ArticlePubMedPDF
- 24. Chiloiro S, Giampietro A, Gagliardi I, Bondanelli M, Veleno M, Ambrosio MR, et al. Impact of the diagnostic delay of acromegaly on bone health: data from a real life and long term follow-up experience. Pituitary 2022;25:831–41.ArticlePubMedPMCPDF
- 25. Frara S, Melin Uygur M, di Filippo L, Doga M, Losa M, Santoro S, et al. High prevalence of vertebral fractures associated with preoperative GH levels in patients with recent diagnosis of acromegaly. J Clin Endocrinol Metab 2022;107:e2843–50.ArticlePubMedPDF
- 26. Chiloiro S, Frara S, Gagliardi I, Bianchi A, Giampietro A, Medici M, et al. Cholecalciferol use is associated with a decreased risk of incident morphometric vertebral fractures in acromegaly. J Clin Endocrinol Metab 2023 Aug 22 [Epub]. https://doi.org/10.1210/clinem/dgad493.Article
- 27. Mancini T, Mazziotti G, Doga M, Carpinteri R, Simetovic N, Vescovi PP, et al. Vertebral fractures in males with type 2 diabetes treated with rosiglitazone. Bone 2009;45:784–8.ArticlePubMed
- 28. Pedersini R, Monteverdi S, Mazziotti G, Amoroso V, Roca E, Maffezzoni F, et al. Morphometric vertebral fractures in breast cancer patients treated with adjuvant aromatase inhibitor therapy: a cross-sectional study. Bone 2017;97:147–52.ArticlePubMed
- 29. Torti C, Mazziotti G, Soldini PA, Foca E, Maroldi R, Gotti D, et al. High prevalence of radiological vertebral fractures in HIV-infected males. Endocrine 2012;41:512–7.ArticlePubMedPDF
- 30. di Filippo L, Formenti AM, Doga M, Pedone E, Rovere-Querini P, Giustina A. Radiological thoracic vertebral fractures are highly prevalent in COVID-19 and predict disease outcomes. J Clin Endocrinol Metab 2021;106:e602–14.ArticlePubMedPDF
- 31. Mazziotti G, Porcelli T, Mormando M, De Menis E, Bianchi A, Mejia C, et al. Vertebral fractures in males with prolactinoma. Endocrine 2011;39:288–93.ArticlePubMedPDF
- 32. Mazziotti G, Mancini T, Mormando M, De Menis E, Bianchi A, Doga M, et al. High prevalence of radiological vertebral fractures in women with prolactin-secreting pituitary adenomas. Pituitary 2011;14:299–306.ArticlePubMedPDF
- 33. Frara S, di Filippo L, Doga M, Loli P, Casanueva FF, Giustina A. Novel approaches to bone comorbidity in Cushing’s disease: an update. Pituitary 2022;25:754–9.ArticlePubMedPDF
- 34. Frara S, Losa M, Doga M, Formenti AM, Mortini P, Mazziotti G, et al. High prevalence of radiological vertebral fractures in patients with TSH-secreting pituitary adenoma. J Endocr Soc 2018;2:1089–99.ArticlePubMedPMC
- 35. Angeli A, Guglielmi G, Dovio A, Capelli G, de Feo D, Giannini S, et al. High prevalence of asymptomatic vertebral fractures in post-menopausal women receiving chronic glucocorticoid therapy: a cross-sectional outpatient study. Bone 2006;39:253–9.ArticlePubMed
- 36. Claessen KM, Mazziotti G, Biermasz NR, Giustina A. Bone and joint disorders in acromegaly. Neuroendocrinology 2016;103:86–95.ArticlePubMedPDF
- 37. Uygur MM, Frara S, di Filippo L, Giustina A. New tools for bone health assessment in secreting pituitary adenomas. Trends Endocrinol Metab 2023;34:231–42.ArticlePubMed
- 38. Skjodt MK, Abrahamsen B. New insights in the pathophysiology, epidemiology, and response to treatment of osteoporotic vertebral fractures. J Clin Endocrinol Metab 2023;108:e1175–85.ArticlePubMedPDF
- 39. Formenti AM, Dalla Volta A, di Filippo L, Berruti A, Giustina A. Effects of medical treatment of prostate cancer on bone health. Trends Endocrinol Metab 2021;32:135–58.ArticlePubMed
- 40. Formenti AM, Tecilazich F, Giubbini R, Giustina A. Risk of vertebral fractures in hypoparathyroidism. Rev Endocr Metab Disord 2019;20:295–302.ArticlePubMedPDF
- 41. Griffith JF, Genant HK. New advances in imaging osteoporosis and its complications. Endocrine 2012;42:39–51.ArticlePubMedPDF
- 42. Bhattacharya S, Nagendra L, Chandran M, Kapoor N, Patil P, Dutta D, et al. Trabecular bone score in adults with type 1 diabetes: a meta-analysis. Osteoporos Int 2023 Oct 11 [Epub]. https://doi.org/10.1007/s00198-023-06935-z.Article
- 43. Song A, Chen R, Guan W, Yu W, Yang Y, Wang J, et al. Trabecular bone score as a more sensitive tool to evaluate bone involvement in MEN1-related primary hyperparathyroidism. J Clin Endocrinol Metab 2023 Aug 4 [Epub]. https://doi.org/10.1210/clinem/dgad460.Article
- 44. Boro H, Mannar V, Malhotra R, Alam S, Khatiwada S, Kubihal S, et al. Trabecular bone score and bone mineral density as indices of skeletal fragility in endogenous Cushing’s syndrome. Clin Endocrinol (Oxf) 2023;99:253–61.ArticlePubMed
- 45. Bioletto F, Barale M, Prencipe N, Berton AM, Parasiliti-Caprino M, Gasco V, et al. Trabecular bone score as an index of bone fragility in patients with acromegaly: a systematic review and meta-analysis. Neuroendocrinology 2023;113:395–405.ArticlePubMedPDF
- 46. Madeira M, Neto LV, de Paula Paranhos Neto F, Barbosa Lima IC, Carvalho de Mendonca LM, Gadelha MR, et al. Acromegaly has a negative influence on trabecular bone, but not on cortical bone, as assessed by high-resolution peripheral quantitative computed tomography. J Clin Endocrinol Metab 2013;98:1734–41.ArticlePubMed
- 47. Maffezzoni F, Maddalo M, Frara S, Mezzone M, Zorza I, Baruffaldi F, et al. High-resolution-cone beam tomography analysis of bone microarchitecture in patients with acromegaly and radiological vertebral fractures. Endocrine 2016;54:532–42.ArticlePubMedPDF
- 48. Malgo F, Hamdy NA, Rabelink TJ, Kroon HM, Claessen KM, Pereira AM, et al. Bone material strength index as measured by impact microindentation is altered in patients with acromegaly. Eur J Endocrinol 2017;176:339–47.ArticlePubMed
- 49. Canalis E, Giustina A, Bilezikian JP. Mechanisms of anabolic therapies for osteoporosis. N Engl J Med 2007;357:905–16.ArticlePubMed
- 50. Mazziotti G, Biagioli E, Maffezzoni F, Spinello M, Serra V, Maroldi R, et al. Bone turnover, bone mineral density, and fracture risk in acromegaly: a meta-analysis. J Clin Endocrinol Metab 2015;100:384–94.ArticlePubMedPDF
- 51. Menagh PJ, Turner RT, Jump DB, Wong CP, Lowry MB, Yakar S, et al. Growth hormone regulates the balance between bone formation and bone marrow adiposity. J Bone Miner Res 2010;25:757–68.ArticlePubMedPMC
- 52. Li H, Bartold PM, Zhang CZ, Clarkson RW, Young WG, Waters MJ. Growth hormone and insulin-like growth factor I induce bone morphogenetic proteins 2 and 4: a mediator role in bone and tooth formation? Endocrinology 1998;139:3855–62.ArticlePubMed
- 53. Bianchi A, Giustina A, Cimino V, Pola R, Angelini F, Pontecorvi A, et al. Influence of growth hormone receptor d3 and full-length isoforms on biochemical treatment outcomes in acromegaly. J Clin Endocrinol Metab 2009;94:2015–22.ArticlePubMed
- 54. Playford MP, Bicknell D, Bodmer WF, Macaulay VM. Insulin-like growth factor 1 regulates the location, stability, and transcriptional activity of beta-catenin. Proc Natl Acad Sci U S A 2000;97:12103–8.PubMedPMC
- 55. Zhao HY, Liu JM, Ning G, Zhao YJ, Chen Y, Sun LH, et al. Relationships between insulin-like growth factor-I (IGF-I) and OPG, RANKL, bone mineral density in healthy Chinese women. Osteoporos Int 2008;19:221–6.ArticlePubMedPDF
- 56. Halhali A, Diaz L, Sanchez I, Garabedian M, Bourges H, Larrea F. Effects of IGF-I on 1,25-dihydroxyvitamin D(3) synthesis by human placenta in culture. Mol Hum Reprod 1999;5:771–6.ArticlePubMed
- 57. Mazziotti G, Maffezzoni F, Giustina A. Vitamin D-binding protein: one more piece in the puzzle of acromegalic osteopathy? Endocrine 2016;52:183–6.ArticlePubMedPDF
- 58. Spinal and peripheral bone mineral densities in acromegaly: the effects of excess growth hormone and hypogonadism. Ann Intern Med 1989;111:567–73.ArticlePubMed
- 59. Zgliczynski W, Kochman M, Misiorowski W, Zdunowski P. In acromegaly, increased bone mineral density (BMD) is determined by GH-excess, gonadal function and gender. Neuro Endocrinol Lett 2007;28:621–8.PubMed
- 60. Chiodini I, Trischitta V, Carnevale V, Liuzzi A, Scillitani A. Bone mineral density in acromegaly: does growth hormone excess protect against osteoporosis? J Endocrinol Invest 2001;24:288–91.ArticlePubMedPDF
- 61. Szulc P. Biochemical bone turnover markers in hormonal disorders in adults: a narrative review. J Endocrinol Invest 2020;43:1409–27.ArticlePubMedPDF
- 62. Ueland T, Stilgren L, Bollerslev J. Bone matrix levels of Dickkopf and sclerostin are positively correlated with bone mass and strength in postmenopausal osteoporosis. Int J Mol Sci 2019;20:2896.ArticlePubMedPMC
- 63. Silva PP, Pereira RM, Takayama L, Borba CG, Duarte FH, Trarbach EB, et al. Impaired bone microarchitecture in premenopausal women with acromegaly: the possible role of Wnt signaling. J Clin Endocrinol Metab 2021;106:2690–706.ArticlePubMedPDF
- 64. Uygur MM, Yazici DD, Bugdayci O, Yavuz DG. Prevalence of vertebral fractures and serum sclerostin levels in acromegaly. Endocrine 2021;73:667–73.ArticlePubMedPDF
- 65. Claessen KM, Pelsma IC, Kroon HM, van Lierop AH, Pereira AM, Biermasz NR, et al. Low sclerostin levels after long-term remission of acromegaly. Endocrine 2022;75:228–38.ArticlePubMedPDF
- 66. Ho PJ, Fig LM, Barkan AL, Shapiro B. Bone mineral density of the axial skeleton in acromegaly. J Nucl Med 1992;33:1608–12.PubMed
- 67. Lesse GP, Fraser WD, Farquharson R, Hipkin L, Vora JP. Gonadal status is an important determinant of bone density in acromegaly. Clin Endocrinol (Oxf) 1998;48:59–65.ArticlePubMedPDF
- 68. di Filippo L, Doga M, Resmini E, Giustina A. Hyperprolactinemia and bone. Pituitary 2020;23:314–21.ArticlePubMedPDF
- 69. Frara S, Allora A, di Filippo L, Formenti AM, Loli P, Polizzi E, et al. Osteopathy in mild adrenal Cushing’s syndrome and Cushing disease. Best Pract Res Clin Endocrinol Metab 2021;35:101515.ArticlePubMed
- 70. Mazziotti G, Formenti AM, Frara S, Roca E, Mortini P, Berruti A, et al. Management of endocrine disease: risk of overtreatment in patients with adrenal insufficiency: current and emerging aspects. Eur J Endocrinol 2017;177:R231–48.ArticlePubMed
- 71. Pelsma IC, Biermasz NR, Pereira AM, van Furth WR, Appelman-Dijkstra NM, Kloppenburg M, et al. Progression of vertebral fractures in long-term controlled acromegaly: a 9-year follow-up study. Eur J Endocrinol 2020;183:427–37.ArticlePubMed
- 72. Mazziotti G, Bianchi A, Porcelli T, Mormando M, Maffezzoni F, Cristiano A, et al. Vertebral fractures in patients with acromegaly: a 3-year prospective study. J Clin Endocrinol Metab 2013;98:3402–10.ArticlePubMed
- 73. Hong AR, Kim JH, Kim SW, Kim SY, Shin CS. Trabecular bone score as a skeletal fragility index in acromegaly patients. Osteoporos Int 2016;27:1123–9.ArticlePubMedPDF
- 74. Kuzma M, Vanuga P, Sagova I, Pavai D, Jackuliak P, Killinger Z, et al. Non-invasive DXA-derived bone structure assessment of acromegaly patients: a cross-sectional study. Eur J Endocrinol 2019;180:201–11.ArticlePubMed
- 75. Calatayud M, Perez-Olivares Martin L, Librizzi MS, Lora Pablos D, Gonzalez Mendez V, Aramendi Ramos M, et al. Trabecular bone score and bone mineral density in patients with long-term controlled acromegaly. Clin Endocrinol (Oxf) 2021;95:58–64.ArticlePubMedPDF
- 76. Sala E, Malchiodi E, Carosi G, Verrua E, Cairoli E, Ferrante E, et al. Spine bone texture assessed by trabecular bone score in active and controlled acromegaly: a prospective study. J Endocr Soc 2021;5:bvab090.ArticlePubMedPMCPDF
- 77. Duan L, Yang S, Wang LJ, Zhang Y, Li R, Yang H, et al. The negative impacts of acromegaly on bone microstructure not fully reversible. Front Endocrinol (Lausanne) 2021;12:738895.ArticlePubMedPMC
- 78. Heck A, Godang K, Lekva T, Markussen KN, De Vincentis S, Ueland T, et al. Endocortical trabecularization in acromegaly: the cause for the paradoxically increased vertebral fracture risk? JBMR Plus 2023;7:e10787.ArticlePubMedPMC
- 79. Sorohan MC, Poiana C. Vertebral fractures in acromegaly: a systematic review. J Clin Med 2022;12:164.ArticlePubMedPMC
- 80. Kuzma M, Vanuga P, Sagova I, Pavai D, Jackuliak P, Killinger Z, et al. Vertebral fractures occur despite control of acromegaly and are predicted by cortical volumetric bone mineral density. J Clin Endocrinol Metab 2021;106:e5088–96.PubMed
- 81. Sardella C, Urbani C, Marconcini G, Cappellani D, Manetti L, De Liperi A, et al. Conventional X-rays in the diagnosis and follow-up of vertebral fractures in patients with acromegaly: a real-life study. J Endocrinol Invest 2023 Sep 5 [Epub]. https://doi.org/10.1007/s40618-023-02148-7.Article
- 82. Giustina A. Acromegaly: reducing diagnostic delay. Recenti Prog Med 2016;107:450–1.PubMed
- 83. Chiloiro S, Giampietro A, Visconti F, Rossi L, Donfrancesco F, Fleseriu CM, et al. Glucose metabolism outcomes in acromegaly patients on treatment with pasireotide-LAR or pasireotide-LAR plus Pegvisomant. Endocrine 2021;73:658–66.ArticlePubMedPMCPDF
- 84. Mazziotti G, Bilezikian J, Canalis E, Cocchi D, Giustina A. New understanding and treatments for osteoporosis. Endocrine 2012;41:58–69.ArticlePubMedPDF
- 85. Mazziotti G, Gola M, Bianchi A, Porcelli T, Giampietro A, Cimino V, et al. Influence of diabetes mellitus on vertebral fractures in men with acromegaly. Endocrine 2011;40:102–8.ArticlePubMedPDF
- 86. Chiloiro S, Mormando M, Bianchi A, Giampietro A, Milardi D, Bima C, et al. Prevalence of morphometric vertebral fractures in “difficult” patients with acromegaly with different biochemical outcomes after multimodal treatment. Endocrine 2018;59:449–53.ArticlePubMedPDF
- 87. Kwon H, Han KD, Kim BS, Moon SJ, Park SE, Rhee EJ, et al. Acromegaly and the long-term fracture risk of the vertebra and hip: a national cohort study. Osteoporos Int 2023;34:1591–600.ArticlePubMedPDF
- 88. Cipriani C, Pepe J, Bertoldo F, Bianchi G, Cantatore FP, Corrado A, et al. The epidemiology of osteoporosis in Italian postmenopausal women according to the National Bone Health Alliance (NBHA) diagnostic criteria: a multicenter cohort study. J Endocrinol Invest 2018;41:431–8.ArticlePubMedPDF
- 89. Valenti G, Bossoni S, Giustina A, Maugeri D, Motta M, Vigna GB, et al. Consensus document on substitution therapy with testosterone in hypoandrogenic elderly men. Aging Clin Exp Res 2002;14:439–64.ArticlePubMedPDF
- 90. Cozzi R, Attanasio R, Oppizzi G, Orlandi P, Giustina A, Lodrini S, et al. Effects of tamoxifen on GH and IGF-I levels in acromegaly. J Endocrinol Invest 1997;20:445–51.ArticlePubMedPDF
- 91. Bouillon R, Marcocci C, Carmeliet G, Bikle D, White JH, Dawson-Hughes B, et al. Skeletal and extraskeletal actions of vitamin D: current evidence and outstanding questions. Endocr Rev 2019;40:1109–51.ArticlePubMedPMC
- 92. Giustina A, di Filippo L, Allora A, Bikle DD, Cavestro GM, Feldman D, et al. Vitamin D and malabsorptive gastrointestinal conditions: a bidirectional relationship? Rev Endocr Metab Disord 2023;24:121–38.ArticlePubMedPMCPDF
- 93. Giustina A, Adler RA, Binkley N, Bollerslev J, Bouillon R, Dawson-Hughes B, et al. Consensus statement from 2nd International Conference on Controversies in Vitamin D. Rev Endocr Metab Disord 2020;21:89–116.ArticlePubMedPMCPDF
- 94. Halupczok-Zyla J, Jawiarczyk-Przybylowska A, Bolanowski M. Patients with active acromegaly are at high risk of 25(OH)D deficiency. Front Endocrinol (Lausanne) 2015;6:89.PubMedPMC
- 95. Jawiarczyk-Przybylowska A, Halupczok-Zyla J, Kolackov K, Gojny L, Zembska A, Bolanowski M. Association of vitamin D receptor polymorphisms with activity of acromegaly, vitamin D status and risk of osteoporotic fractures in acromegaly patients. Front Endocrinol (Lausanne) 2019;10:643.PubMedPMC
- 96. Legovini P, De Menis E, Breda F, Billeci D, Carteri A, Pavan P, et al. Long-term effects of octreotide on markers of bone metabolism in acromegaly: evidence of increased serum parathormone concentrations. J Endocrinol Invest 1997;20:434–8.ArticlePubMedPDF
- 97. Ajmal A, Haghshenas A, Attarian S, Barake M, Tritos NA, Klibanski A, et al. The effect of somatostatin analogs on vitamin D and calcium concentrations in patients with acromegaly. Pituitary 2014;17:366–73.ArticlePubMedPDF
- 98. Giustina A, Bouillon R, Binkley N, Sempos C, Adler RA, Bollerslev J, et al. Controversies in vitamin D: a statement from the third International Conference. JBMR Plus 2020;4:e10417.ArticlePubMedPMCPDF
- 99. Giustina A, Bouillon R, Dawson-Hughes B, Ebeling PR, Lazaretti-Castro M, Lips P, et al. Vitamin D in the older population: a consensus statement. Endocrine 2023;79:31–44.ArticlePubMedPMCPDF
- 100. Ebeling PR, Adler RA, Jones G, Liberman UA, Mazziotti G, Minisola S, et al. Management of endocrine disease: therapeutics of vitamin D. Eur J Endocrinol 2018;179:R239–59.ArticlePubMed
- 101. LeBoff MS, Chou SH, Ratliff KA, Cook NR, Khurana B, Kim E, et al. Supplemental vitamin D and incident fractures in midlife and older adults. N Engl J Med 2022;387:299–309.ArticlePubMedPMC
- 102. Bilezikian JP, Formenti AM, Adler RA, Binkley N, Bouillon R, Lazaretti-Castro M, et al. Vitamin D: dosing, levels, form, and route of administration: does one approach fit all? Rev Endocr Metab Disord 2021;22:1201–18.ArticlePubMedPMCPDF
- 103. Mazziotti G, Formenti AM, Adler RA, Bilezikian JP, Grossman A, Sbardella E, et al. Glucocorticoid-induced osteoporosis: pathophysiological role of GH/IGF-I and PTH/VITAMIN D axes, treatment options and guidelines. Endocrine 2016;54:603–11.ArticlePubMedPDF
- 104. Manelli F, Giustina A. Glucocorticoid-induced osteoporosis. Trends Endocrinol Metab 2000;11:79–85.ArticlePubMed
- 105. Canalis E, Bilezikian JP, Angeli A, Giustina A. Perspectives on glucocorticoid-induced osteoporosis. Bone 2004;34:593–8.ArticlePubMed
- 106. Puig-Domingo M, Marazuela M, Yildiz BO, Giustina A. COVID-19 and endocrine and metabolic diseases: an updated statement from the European Society of Endocrinology. Endocrine 2021;72:301–16.ArticlePubMedPMCPDF
- 107. Mazziotti G, Battista C, Maffezzoni F, Chiloiro S, Ferrante E, Prencipe N, et al. Treatment of acromegalic osteopathy in real-life clinical practice: the BAAC (Bone Active Drugs in Acromegaly) Study. J Clin Endocrinol Metab 2020;105:dgaa363.ArticlePubMedPDF
- 108. Mazziotti G, Chiavistelli S, Giustina A. Pituitary diseases and bone. Endocrinol Metab Clin North Am 2015;44:171–80.ArticlePubMed
- 109. di Filippo L, Ulivieri FM, Nuti R, Giustina A. Use of vitamin D with anti-osteoporotic drugs: are available clinical trials telling us the whole story? Endocrine 2023 Oct 10 [Epub]. https://doi.org/10.1007/s12020-023-03551-z.Article
- 110. Frara S, Rodriguez-Carnero G, Formenti AM, Martinez-Olmos MA, Giustina A, Casanueva FF. Pituitary tumors centers of excellence. Endocrinol Metab Clin North Am 2020;49:553–64.ArticlePubMed
- 111. Giustina A, Uygur MM, Frara S, Barkan A, Biermasz NR, Chanson P, et al. Pilot study to define criteria for pituitary tumors centers of excellence (PTCOE): results of an audit of leading international centers. Pituitary 2023;26:583–96.ArticlePubMedPMCPDF
- 112. Mazziotti G, Bianchi A, Bonadonna S, Nuzzo M, Cimino V, Fusco A, et al. Increased prevalence of radiological spinal deformities in adult patients with GH deficiency: influence of GH replacement therapy. J Bone Miner Res 2006;21:520–8.ArticlePubMed
- 113. Mazziotti G, Doga M, Frara S, Maffezzoni F, Porcelli T, Cerri L, et al. Incidence of morphometric vertebral fractures in adult patients with growth hormone deficiency. Endocrine 2016;52:103–10.ArticlePubMedPDF
- 114. Formenti AM, Maffezzoni F, Doga M, Mazziotti G, Giustina A. Growth hormone deficiency in treated acromegaly and active Cushing’s syndrome. Best Pract Res Clin Endocrinol Metab 2017;31:79–90.ArticlePubMed
- 115. Giustina A, Chanson P, Kleinberg D, Bronstein MD, Clemmons DR, Klibanski A, et al. Expert consensus document: a consensus on the medical treatment of acromegaly. Nat Rev Endocrinol 2014;10:243–8.ArticlePubMedPDF
- 116. Melmed S, Colao A, Barkan A, Molitch M, Grossman AB, Kleinberg D, et al. Guidelines for acromegaly management: an update. J Clin Endocrinol Metab 2009;94:1509–17.ArticlePubMed
- 117. Giustina A, Bronstein MD, Casanueva FF, Chanson P, Ghigo E, Ho KK, et al. Current management practices for acromegaly: an international survey. Pituitary 2011;14:125–33.ArticlePubMedPDF
- 118. Giustina A, Karamouzis I, Patelli I, Mazziotti G. Octreotide for acromegaly treatment: a reappraisal. Expert Opin Pharmacother 2013;14:2433–47.ArticlePubMed
- 119. Ambrosio MR, Franceschetti P, Bondanelli M, Doga M, Maffei P, Baldelli R, et al. Efficacy and safety of the new 60-mg formulation of the long-acting somatostatin analog lanreotide in the treatment of acromegaly. Metabolism 2002;51:387–93.ArticlePubMed
- 120. Giustina A, Mazziotti G, Cannavo S, Castello R, Arnaldi G, Bugari G, et al. High-dose and high-frequency lanreotide autogel in acromegaly: a randomized, multicenter study. J Clin Endocrinol Metab 2017;102:2454–64.ArticlePubMed
- 121. Giustina A, Bonadonna S, Bugari G, Colao A, Cozzi R, Cannavo S, et al. High-dose intramuscular octreotide in patients with acromegaly inadequately controlled on conventional somatostatin analogue therapy: a randomised controlled trial. Eur J Endocrinol 2009;161:331–8.ArticlePubMed
- 122. Giustina A, Arnaldi G, Bogazzi F, Cannavo S, Colao A, De Marinis L, et al. Pegvisomant in acromegaly: an update. J Endocrinol Invest 2017;40:577–89.ArticlePubMedPMCPDF
- 123. Giustina A. Optimal use of pegvisomant in acromegaly: are we getting there? Endocrine 2015;48:3–8.ArticlePubMedPDF
- 124. Maffezzoni F, Formenti AM, Mazziotti G, Frara S, Giustina A. Current and future medical treatments for patients with acromegaly. Expert Opin Pharmacother 2016;17:1631–42.ArticlePubMed
- 125. Melmed S, Bronstein MD, Chanson P, Klibanski A, Casanueva FF, Wass JA, et al. A consensus statement on acromegaly therapeutic outcomes. Nat Rev Endocrinol 2018;14:552–61.ArticlePubMedPMCPDF
- 126. Chiloiro S, Giampietro A, Mirra F, Donfrancesco F, Tartaglione T, Mattogno PP, et al. Pegvisomant and pasireotide LAR as second line therapy in acromegaly: clinical effectiveness and predictors of response. Eur J Endocrinol 2021;184:217–29.ArticlePubMed
- 127. Maison P, Tropeano AI, Macquin-Mavier I, Giustina A, Chanson P. Impact of somatostatin analogs on the heart in acromegaly: a metaanalysis. J Clin Endocrinol Metab 2007;92:1743–7.ArticlePubMed
- 128. De Marinis L, Bianchi A, Mazziotti G, Mettimano M, Milardi D, Fusco A, et al. The long-term cardiovascular outcome of different GH-lowering treatments in acromegaly. Pituitary 2008;11:13–20.ArticlePubMedPDF
- 129. Chiloiro S, Mazziotti G, Giampietro A, Bianchi A, Frara S, Mormando M, et al. Effects of pegvisomant and somatostatin receptor ligands on incidence of vertebral fractures in patients with acromegaly. Pituitary 2018;21:302–8.ArticlePubMedPDF
- 130. Chiloiro S, Giampietro A, Frara S, Bima C, Donfrancesco F, Fleseriu CM, et al. Effects of pegvisomant and pasireotide LAR on vertebral fractures in acromegaly resistant to first-generation SRLs. J Clin Endocrinol Metab 2020;105:dgz 054.ArticlePDF
- 131. Giustina A, Biermasz N, Casanueva FF, Fleseriu M, Mortini P, Strasburger C, et al. Consensus on criteria for acromegaly diagnosis and remission. Pituitary 2023 Nov 3 [Epub]. https://doi.org/10.1007/s11102-023-01360-1.Article
References
Figure & Data
References
Citations
Citations to this article as recorded by 

- New insights into the vitamin D/PTH axis in endocrine-driven metabolic bone diseases
Luigi di Filippo, John P. Bilezikian, Ernesto Canalis, Umberto Terenzi, Andrea Giustina
Endocrine.2024;[Epub] CrossRef

 KES
KES
 PubReader
PubReader ePub Link
ePub Link Cite
Cite