Search
- Page Path
- HOME > Search
- Calcium & Bone Metabolism
- Development of a Spine X-Ray-Based Fracture Prediction Model Using a Deep Learning Algorithm
- Sung Hye Kong, Jae-Won Lee, Byeong Uk Bae, Jin Kyeong Sung, Kyu Hwan Jung, Jung Hee Kim, Chan Soo Shin
- Endocrinol Metab. 2022;37(4):674-683. Published online August 5, 2022
- DOI: https://doi.org/10.3803/EnM.2022.1461
- 3,938 View
- 212 Download
- 13 Web of Science
- 15 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
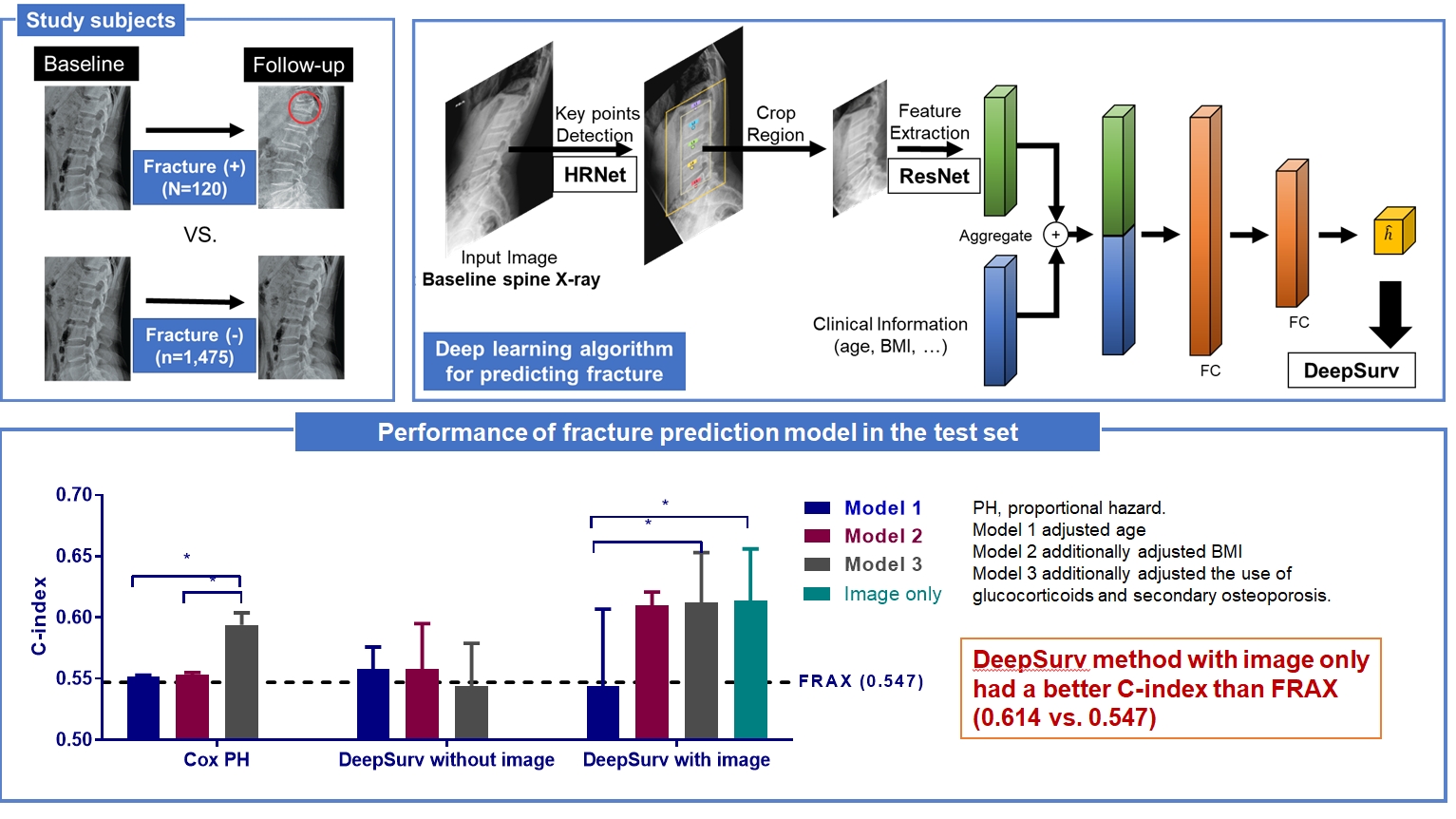
Since image-based fracture prediction models using deep learning are lacking, we aimed to develop an X-ray-based fracture prediction model using deep learning with longitudinal data.
Methods
This study included 1,595 participants aged 50 to 75 years with at least two lumbosacral radiographs without baseline fractures from 2010 to 2015 at Seoul National University Hospital. Positive and negative cases were defined according to whether vertebral fractures developed during follow-up. The cases were divided into training (n=1,416) and test (n=179) sets. A convolutional neural network (CNN)-based prediction algorithm, DeepSurv, was trained with images and baseline clinical information (age, sex, body mass index, glucocorticoid use, and secondary osteoporosis). The concordance index (C-index) was used to compare performance between DeepSurv and the Fracture Risk Assessment Tool (FRAX) and Cox proportional hazard (CoxPH) models.
Results
Of the total participants, 1,188 (74.4%) were women, and the mean age was 60.5 years. During a mean follow-up period of 40.7 months, vertebral fractures occurred in 7.5% (120/1,595) of participants. In the test set, when DeepSurv learned with images and clinical features, it showed higher performance than FRAX and CoxPH in terms of C-index values (DeepSurv, 0.612; 95% confidence interval [CI], 0.571 to 0.653; FRAX, 0.547; CoxPH, 0.594; 95% CI, 0.552 to 0.555). Notably, the DeepSurv method without clinical features had a higher C-index (0.614; 95% CI, 0.572 to 0.656) than that of FRAX in women.
Conclusion
DeepSurv, a CNN-based prediction algorithm using baseline image and clinical information, outperformed the FRAX and CoxPH models in predicting osteoporotic fracture from spine radiographs in a longitudinal cohort. -
Citations
Citations to this article as recorded by- Automated detection of vertebral fractures from X-ray images: A novel machine learning model and survey of the field
Li-Wei Cheng, Hsin-Hung Chou, Yu-Xuan Cai, Kuo-Yuan Huang, Chin-Chiang Hsieh, Po-Lun Chu, I-Szu Cheng, Sun-Yuan Hsieh
Neurocomputing.2024; 566: 126946. CrossRef - Application of radiomics model based on lumbar computed tomography in diagnosis of elderly osteoporosis
Baisen Chen, Jiaming Cui, Chaochen Li, Pengjun Xu, Guanhua Xu, Jiawei Jiang, Pengfei Xue, Yuyu Sun, Zhiming Cui
Journal of Orthopaedic Research.2024;[Epub] CrossRef - Machine Learning and Deep Learning in Spinal Injury: A Narrative Review of Algorithms in Diagnosis and Prognosis
Satoshi Maki, Takeo Furuya, Masahiro Inoue, Yasuhiro Shiga, Kazuhide Inage, Yawara Eguchi, Sumihisa Orita, Seiji Ohtori
Journal of Clinical Medicine.2024; 13(3): 705. CrossRef - A CT-based Deep Learning Model for Predicting Subsequent Fracture Risk in Patients with Hip Fracture
Yisak Kim, Young-Gon Kim, Jung-Wee Park, Byung Woo Kim, Youmin Shin, Sung Hye Kong, Jung Hee Kim, Young-Kyun Lee, Sang Wan Kim, Chan Soo Shin
Radiology.2024;[Epub] CrossRef - A Novel QCT-Based Deep Transfer Learning Approach for Predicting Stiffness Tensor of Trabecular Bone Cubes
Pengwei Xiao, Tinghe Zhang, Yufei Huang, Xiaodu Wang
IRBM.2024; 45(2): 100831. CrossRef - Deep learning in the radiologic diagnosis of osteoporosis: a literature review
Yu He, Jiaxi Lin, Shiqi Zhu, Jinzhou Zhu, Zhonghua Xu
Journal of International Medical Research.2024;[Epub] CrossRef - Development and Validation of a Convolutional Neural Network Model to Predict a Pathologic Fracture in the Proximal Femur Using Abdomen and Pelvis CT Images of Patients With Advanced Cancer
Min Wook Joo, Taehoon Ko, Min Seob Kim, Yong-Suk Lee, Seung Han Shin, Yang-Guk Chung, Hong Kwon Lee
Clinical Orthopaedics & Related Research.2023; 481(11): 2247. CrossRef - Automated Opportunistic Trabecular Volumetric Bone Mineral Density Extraction Outperforms Manual Measurements for the Prediction of Vertebral Fractures in Routine CT
Sophia S. Goller, Jon F. Rischewski, Thomas Liebig, Jens Ricke, Sebastian Siller, Vanessa F. Schmidt, Robert Stahl, Julian Kulozik, Thomas Baum, Jan S. Kirschke, Sarah C. Foreman, Alexandra S. Gersing
Diagnostics.2023; 13(12): 2119. CrossRef - Machine learning‐based prediction of osteoporosis in postmenopausal women with clinical examined features: A quantitative clinical study
Kainat A. Ullah, Faisal Rehman, Muhammad Anwar, Muhammad Faheem, Naveed Riaz
Health Science Reports.2023;[Epub] CrossRef - Skeletal Fracture Detection with Deep Learning: A Comprehensive Review
Zhihao Su, Afzan Adam, Mohammad Faidzul Nasrudin, Masri Ayob, Gauthamen Punganan
Diagnostics.2023; 13(20): 3245. CrossRef - Deep learning system for automated detection of posterior ligamentous complex injury in patients with thoracolumbar fracture on MRI
Sang Won Jo, Eun Kyung Khil, Kyoung Yeon Lee, Il Choi, Yu Sung Yoon, Jang Gyu Cha, Jae Hyeok Lee, Hyunggi Kim, Sun Yeop Lee
Scientific Reports.2023;[Epub] CrossRef - Vertebra Segmentation Based Vertebral Compression Fracture Determination from Reconstructed Spine X-Ray Images
Srinivasa Rao Gadu, Chandra Sekhar Potala
International Journal of Electrical and Electronics Research.2023; 11(4): 1225. CrossRef - Computer Vision in Osteoporotic Vertebral Fracture Risk Prediction: A Systematic Review
Anthony K. Allam, Adrish Anand, Alex R. Flores, Alexander E. Ropper
Neurospine.2023; 20(4): 1112. CrossRef - A Meaningful Journey to Predict Fractures with Deep Learning
Jeonghoon Ha
Endocrinology and Metabolism.2022; 37(4): 617. CrossRef - New Horizons: Artificial Intelligence Tools for Managing Osteoporosis
Hans Peter Dimai
The Journal of Clinical Endocrinology & Metabolism.2022;[Epub] CrossRef
- Automated detection of vertebral fractures from X-ray images: A novel machine learning model and survey of the field

- Miscellaneous
- Machine Learning Applications in Endocrinology and Metabolism Research: An Overview
- Namki Hong, Heajeong Park, Yumie Rhee
- Endocrinol Metab. 2020;35(1):71-84. Published online March 19, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.1.71
- 15,569 View
- 205 Download
- 13 Web of Science
- 13 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
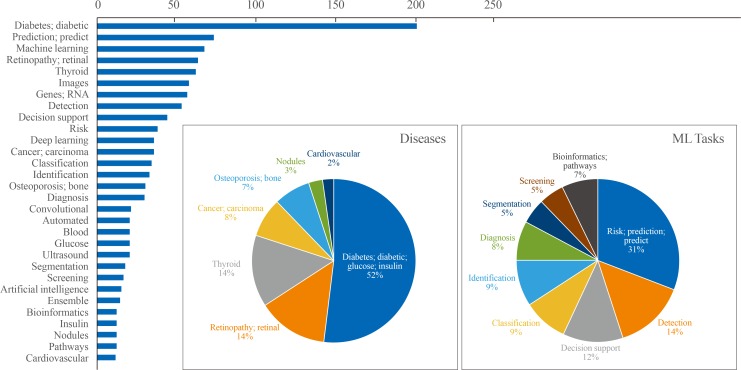
ePub Machine learning (ML) applications have received extensive attention in endocrinology research during the last decade. This review summarizes the basic concepts of ML and certain research topics in endocrinology and metabolism where ML principles have been actively deployed. Relevant studies are discussed to provide an overview of the methodology, main findings, and limitations of ML, with the goal of stimulating insights into future research directions. Clear, testable study hypotheses stem from unmet clinical needs, and the management of data quality (beyond a focus on quantity alone), open collaboration between clinical experts and ML engineers, the development of interpretable high-performance ML models beyond the black-box nature of some algorithms, and a creative environment are the core prerequisites for the foreseeable changes expected to be brought about by ML and artificial intelligence in the field of endocrinology and metabolism, with actual improvements in clinical practice beyond hype. Of note, endocrinologists will continue to play a central role in these developments as domain experts who can properly generate, refine, analyze, and interpret data with a combination of clinical expertise and scientific rigor.
-
Citations
Citations to this article as recorded by- Artificial Intelligence for Predicting and Diagnosing Complications of Diabetes
Jingtong Huang, Andrea M. Yeung, David G. Armstrong, Ashley N. Battarbee, Jorge Cuadros, Juan C. Espinoza, Samantha Kleinberg, Nestoras Mathioudakis, Mark A. Swerdlow, David C. Klonoff
Journal of Diabetes Science and Technology.2023; 17(1): 224. CrossRef - Expressions of Cushing’s syndrome in multiple endocrine neoplasia type 1
William F. Simonds
Frontiers in Endocrinology.2023;[Epub] CrossRef - Application of machine learning and artificial intelligence in the diagnosis and classification of polycystic ovarian syndrome: a systematic review
Francisco J. Barrera, Ethan D.L. Brown, Amanda Rojo, Javier Obeso, Hiram Plata, Eddy P. Lincango, Nancy Terry, René Rodríguez-Gutiérrez, Janet E. Hall, Skand Shekhar
Frontiers in Endocrinology.2023;[Epub] CrossRef - Predictors of rituximab effect on modified Rodnan skin score in systemic sclerosis: a machine-learning analysis of the DesiReS trial
Satoshi Ebata, Koji Oba, Kosuke Kashiwabara, Keiko Ueda, Yukari Uemura, Takeyuki Watadani, Takemichi Fukasawa, Shunsuke Miura, Asako Yoshizaki-Ogawa, Asano Yoshihide, Ayumi Yoshizaki, Shinichi Sato
Rheumatology.2022; 61(11): 4364. CrossRef - Automating and improving cardiovascular disease prediction using Machine learning and EMR data features from a regional healthcare system
Qi Li, Alina Campan, Ai Ren, Wael E. Eid
International Journal of Medical Informatics.2022; 163: 104786. CrossRef - An Interactive Online App for Predicting Diabetes via Machine Learning from Environment-Polluting Chemical Exposure Data
Rosy Oh, Hong Kyu Lee, Youngmi Kim Pak, Man-Suk Oh
International Journal of Environmental Research and Public Health.2022; 19(10): 5800. CrossRef - Ensemble blood glucose prediction in diabetes mellitus: A review
M.Z. Wadghiri, A. Idri, Touria El Idrissi, Hajar Hakkoum
Computers in Biology and Medicine.2022; 147: 105674. CrossRef - The maze runner: navigating through basic kinetics to AI models of human metabolism pathology
Arina V. Martyshina, Oksana M. Tilinova, Anastasia A. Simanova, Olga S. Knyazeva, Irina V. Dokukina
Procedia Computer Science.2022; 213: 271. CrossRef - Applications of Machine Learning in Bone and Mineral Research
Sung Hye Kong, Chan Soo Shin
Endocrinology and Metabolism.2021; 36(5): 928. CrossRef - Facial Recognition Intensity in Disease Diagnosis Using Automatic Facial Recognition
Danning Wu, Shi Chen, Yuelun Zhang, Huabing Zhang, Qing Wang, Jianqiang Li, Yibo Fu, Shirui Wang, Hongbo Yang, Hanze Du, Huijuan Zhu, Hui Pan, Zhen Shen
Journal of Personalized Medicine.2021; 11(11): 1172. CrossRef - The Application of Artificial Intelligence and Machine Learning in Pituitary Adenomas
Congxin Dai, Bowen Sun, Renzhi Wang, Jun Kang
Frontiers in Oncology.2021;[Epub] CrossRef - Real World Data and Artificial Intelligence in Diabetology
Kwang Joon Kim
The Journal of Korean Diabetes.2020; 21(3): 140. CrossRef - A Novel Detection Framework for Detecting Abnormal Human Behavior
Chengfei Wu, Zixuan Cheng, Yi-Zhang Jiang
Mathematical Problems in Engineering.2020; 2020: 1. CrossRef
- Artificial Intelligence for Predicting and Diagnosing Complications of Diabetes

- Miscellaneous
- Medical Big Data Is Not Yet Available: Why We Need Realism Rather than Exaggeration
- Hun-Sung Kim, Dai-Jin Kim, Kun-Ho Yoon
- Endocrinol Metab. 2019;34(4):349-354. Published online December 23, 2019
- DOI: https://doi.org/10.3803/EnM.2019.34.4.349
- 5,847 View
- 140 Download
- 36 Web of Science
- 47 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub Most people are now familiar with the concepts of big data, deep learning, machine learning, and artificial intelligence (AI) and have a vague expectation that AI using medical big data can be used to improve the quality of medical care. However, the expectation that big data could change the field of medicine is inconsistent with the current reality. The clinical meaningfulness of the results of research using medical big data needs to be examined. Medical staff needs to be clear about the purpose of AI that utilizes medical big data and to focus on the quality of this data, rather than the quantity. Further, medical professionals should understand the necessary precautions for using medical big data, as well as its advantages. No doubt that someday, medical big data will play an essential role in healthcare; however, at present, it seems too early to actively use it in clinical practice. The field continues to work toward developing medical big data and making it appropriate for healthcare. Researchers should continue to engage in empirical research to ensure that appropriate processes are in place to empirically evaluate the results of its use in healthcare.
-
Citations
Citations to this article as recorded by- Current status of remote collaborative care for hypertension in medically underserved areas
Seo Yeon Baik, Kyoung Min Kim, Hakyoung Park, Jiwon Shinn, Hun-Sung Kim
Cardiovascular Prevention and Pharmacotherapy.2024; 6(1): 33. CrossRef - Prediction of Cardiovascular Complication in Patients with Newly Diagnosed Type 2 Diabetes Using an XGBoost/GRU-ODE-Bayes-Based Machine-Learning Algorithm
Joonyub Lee, Yera Choi, Taehoon Ko, Kanghyuck Lee, Juyoung Shin, Hun-Sung Kim
Endocrinology and Metabolism.2024; 39(1): 176. CrossRef - Dark Data in Real-World Evidence: Challenges, Implications, and the Imperative of Data Literacy in Medical Research
Hun-Sung Kim
Journal of Korean Medical Science.2024;[Epub] CrossRef - A comparative analysis: health data protection laws in Malaysia, Saudi Arabia and EU General Data Protection Regulation (GDPR)
Jawahitha Sarabdeen, Mohamed Mazahir Mohamed Ishak
International Journal of Law and Management.2024;[Epub] CrossRef - Long-Term Risk of Cardiovascular Disease Among Type 2 Diabetes Patients According to Average and Visit-to-Visit Variations of HbA1c Levels During the First 3 Years of Diabetes Diagnosis
Hyunah Kim, Da Young Jung, Seung-Hwan Lee, Jae-Hyoung Cho, Hyeon Woo Yim, Hun-Sung Kim
Journal of Korean Medical Science.2023;[Epub] CrossRef - Comparison of cardiocerebrovascular disease incidence between angiotensin converting enzyme inhibitor and angiotensin receptor blocker users in a real-world cohort
Suehyun Lee, Hyunah Kim, Hyeon Woo Yim, Kim Hun-Sung, Ju Han Kim
Journal of Applied Biomedicine.2023; 21(1): 7. CrossRef - Multi-Omics and Management of Follicular Carcinoma of the Thyroid
Thifhelimbilu Emmanuel Luvhengo, Ifongo Bombil, Arian Mokhtari, Maeyane Stephens Moeng, Demetra Demetriou, Claire Sanders, Zodwa Dlamini
Biomedicines.2023; 11(4): 1217. CrossRef - Correlation analysis of cancer incidence after pravastatin treatment
Jin Yu, Raeun Kim, Jiwon Shinn, Man Young Park, Hun-Sung Kim
Cardiovascular Prevention and Pharmacotherapy.2023; 5(2): 61. CrossRef - A New Strategy for Evaluating the Quality of Laboratory Results for Big Data Research: Using External Quality Assessment Survey Data (2010–2020)
Eun-Jung Cho, Tae-Dong Jeong, Sollip Kim, Hyung-Doo Park, Yeo-Min Yun, Sail Chun, Won-Ki Min
Annals of Laboratory Medicine.2023; 43(5): 425. CrossRef - Weight loss and side-effects of liraglutide and lixisenatide in obesity and type 2 diabetes mellitus
Jeongmin Lee, Raeun Kim, Min-Hee Kim, Seung-Hwan Lee, Jae-Hyoung Cho, Jung Min Lee, Sang-Ah Jang, Hun-Sung Kim
Primary Care Diabetes.2023; 17(5): 460. CrossRef - Cohort profile for development of machine learning models to predict healthcare-related adverse events (Demeter): clinical objectives, data requirements for modelling and overview of data set for 2016–2018
Svetlana Artemova, Ursula von Schenck, Rui Fa, Daniel Stoessel, Hadiseh Nowparast Rostami, Pierre-Ephrem Madiot, Jean-Marie Januel, Daniel Pagonis, Caroline Landelle, Meghann Gallouche, Christophe Cancé, Frederic Olive, Alexandre Moreau-Gaudry, Sigurd Pri
BMJ Open.2023; 13(8): e070929. CrossRef - The Present and Future of Artificial Intelligence-Based Medical Image in Diabetes Mellitus: Focus on Analytical Methods and Limitations of Clinical Use
Ji-Won Chun, Hun-Sung Kim
Journal of Korean Medical Science.2023;[Epub] CrossRef - Construction and application on the training course of information literacy for clinical nurses
Chao Wu, Yinjuan Zhang, Jing Wu, Linyuan Zhang, Juan Du, Lu Li, Nana Chen, Liping Zhu, Sheng Zhao, Hongjuan Lang
BMC Medical Education.2023;[Epub] CrossRef - Lightweight Histological Tumor Classification Using a Joint Sparsity-Quantization Aware Training Framework
Dina Aboutahoun, Rami Zewail, Keiji Kimura, Mostafa I. Soliman
IEEE Access.2023; 11: 119342. CrossRef - Long-Term Cumulative Exposure to High γ-Glutamyl Transferase Levels and the Risk of Cardiovascular Disease: A Nationwide Population-Based Cohort Study
Han-Sang Baek, Bongseong Kim, Seung-Hwan Lee, Dong-Jun Lim, Hyuk-Sang Kwon, Sang-Ah Chang, Kyungdo Han, Jae-Seung Yun
Endocrinology and Metabolism.2023; 38(6): 770. CrossRef - Comorbidity Patterns and Management in Inpatients with Endocrine Diseases by Age Groups in South Korea: Nationwide Data
Sung-Soo Kim, Hun-Sung Kim
Journal of Personalized Medicine.2023; 14(1): 42. CrossRef - Angiotensin‐converting enzyme inhibitors versus angiotensin receptor blockers: New‐onset diabetes mellitus stratified by statin use
Juyoung Shin, Hyunah Kim, Hyeon Woo Yim, Ju Han Kim, Suehyun Lee, Hun‐Sung Kim
Journal of Clinical Pharmacy and Therapeutics.2022; 47(1): 97. CrossRef - Physician Knowledge Base: Clinical Decision Support Systems
Sira Kim, Eung-Hee Kim, Hun-Sung Kim
Yonsei Medical Journal.2022; 63(1): 8. CrossRef - Sodium-Glucose Cotransporter-2 Inhibitor-Related Diabetic Ketoacidosis: Accuracy Verification of Operational Definition
Dong Yoon Kang, Hyunah Kim, SooJeong Ko, HyungMin Kim, Jiwon Shinn, Min-Gyu Kang, Sun-ju Byeon, Jeong-Hee Choi, Soo-Yong Shin, Hun-Sung Kim
Journal of Korean Medical Science.2022;[Epub] CrossRef - Drug Repositioning: Exploring New Indications for Existing Drug-Disease Relationships
Hun-Sung Kim
Endocrinology and Metabolism.2022; 37(1): 62. CrossRef - A Study on Methodologies of Drug Repositioning Using Biomedical Big Data: A Focus on Diabetes Mellitus
Suehyun Lee, Seongwoo Jeon, Hun-Sung Kim
Endocrinology and Metabolism.2022; 37(2): 195. CrossRef - Development of a predictive model for the side effects of liraglutide
Jiyoung Min, Jiwon Shinn, Hun-Sung Kim
Cardiovascular Prevention and Pharmacotherapy.2022; 4(2): 87. CrossRef - Understanding and Utilizing Claim Data from the Korean National Health Insurance Service (NHIS) and Health Insurance Review & Assessment (HIRA) Database for Research
Dae-Sung Kyoung, Hun-Sung Kim
Journal of Lipid and Atherosclerosis.2022; 11(2): 103. CrossRef - The Impact of the Association between Cancer and Diabetes Mellitus on Mortality
Sung-Soo Kim, Hun-Sung Kim
Journal of Personalized Medicine.2022; 12(7): 1099. CrossRef - Development of Various Diabetes Prediction Models Using Machine Learning Techniques
Juyoung Shin, Jaewon Kim, Chanjung Lee, Joon Young Yoon, Seyeon Kim, Seungjae Song, Hun-Sung Kim
Diabetes & Metabolism Journal.2022; 46(4): 650. CrossRef - Characteristics of Glycemic Control and Long-Term Complications in Patients with Young-Onset Type 2 Diabetes
Han-sang Baek, Ji-Yeon Park, Jin Yu, Joonyub Lee, Yeoree Yang, Jeonghoon Ha, Seung Hwan Lee, Jae Hyoung Cho, Dong-Jun Lim, Hun-Sung Kim
Endocrinology and Metabolism.2022; 37(4): 641. CrossRef - Retrospective cohort analysis comparing changes in blood glucose level and body composition according to changes in thyroid‐stimulating hormone level
Hyunah Kim, Da Young Jung, Seung‐Hwan Lee, Jae‐Hyoung Cho, Hyeon Woo Yim, Hun‐Sung Kim
Journal of Diabetes.2022; 14(9): 620. CrossRef - Long-Term Changes in HbA1c According to Blood Glucose Control Status During the First 3 Months After Visiting a Tertiary University Hospital
Hyunah Kim, Da Young Jung, Seung-Hwan Lee, Jae-Hyoung Cho, Hyeon Woo Yim, Hun-Sung Kim
Journal of Korean Medical Science.2022;[Epub] CrossRef - Medication based machine learning to identify subpopulations of pediatric hemodialysis patients in an electronic health record database
Autumn M. McKnite, Kathleen M. Job, Raoul Nelson, Catherine M.T. Sherwin, Kevin M. Watt, Simon C. Brewer
Informatics in Medicine Unlocked.2022; 34: 101104. CrossRef - Improving Machine Learning Diabetes Prediction Models for the Utmost Clinical Effectiveness
Juyoung Shin, Joonyub Lee, Taehoon Ko, Kanghyuck Lee, Yera Choi, Hun-Sung Kim
Journal of Personalized Medicine.2022; 12(11): 1899. CrossRef - A Study on Weight Loss Cause as per the Side Effect of Liraglutide
Jin Yu, Jeongmin Lee, Seung-Hwan Lee, Jae-Hyung Cho, Hun-Sung Kim, Heng Zhou
Cardiovascular Therapeutics.2022; 2022: 1. CrossRef - Risk Classification and Subphenotyping of Acute Kidney Injury: Concepts and Methodologies
Javier A. Neyra, Jin Chen, Sean M. Bagshaw, Jay L. Koyner
Seminars in Nephrology.2022; 42(3): 151285. CrossRef - Estimation of sodium‐glucose cotransporter 2 inhibitor–related genital and urinary tract infections via electronic medical record–based common data model
SooJeong Ko, HyungMin Kim, Jiwon Shinn, Sun‐ju Byeon, Jeong‐Hee Choi, Hun‐Sung Kim
Journal of Clinical Pharmacy and Therapeutics.2021; 46(4): 975. CrossRef - Blood glucose levels and bodyweight change after dapagliflozin administration
Hyunah Kim, Seung‐Hwan Lee, Hyunyong Lee, Hyeon Woo Yim, Jae‐Hyoung Cho, Kun‐Ho Yoon, Hun‐Sung Kim
Journal of Diabetes Investigation.2021; 12(9): 1594. CrossRef - Artificial intelligence in healthcare: possibilities of patent protection
T. N. Erivantseva, Yu. V. Blokhina
FARMAKOEKONOMIKA. Modern Pharmacoeconomic and Pharmacoepidemiology.2021; 14(2): 270. CrossRef - Lack of Acceptance of Digital Healthcare in the Medical Market: Addressing Old Problems Raised by Various Clinical Professionals and Developing Possible Solutions
Jong Il Park, Hwa Young Lee, Hyunah Kim, Jisan Lee, Jiwon Shinn, Hun-Sung Kim
Journal of Korean Medical Science.2021;[Epub] CrossRef - Prospect of Artificial Intelligence Based on Electronic Medical Records
Suehyun Lee, Hun-Sung Kim
Journal of Lipid and Atherosclerosis.2021; 10(3): 282. CrossRef - Data Pseudonymization in a Range That Does Not Affect Data Quality: Correlation with the Degree of Participation of Clinicians
Soo-Yong Shin, Hun-Sung Kim
Journal of Korean Medical Science.2021;[Epub] CrossRef - Development of a Predictive Model for Glycated Hemoglobin Values and Analysis of the Factors Affecting It
HyeongKyu Park, Da Young Lee, So young Park, Jiyoung Min, Jiwon Shinn, Dae Ho Lee, Soon Hyo Kwon, Hun-Sung Kim, Nan Hee Kim
Cardiovascular Prevention and Pharmacotherapy.2021; 3(4): 106. CrossRef - Modeling of Changes in Creatine Kinase after HMG-CoA Reductase Inhibitor Prescription
Hun-Sung Kim, Jiyoung Min, Jiwon Shinn, Oak-Kee Hong, Jang-Won Son, Seong-Su Lee, Sung-Rae Kim, Soon Jib Yoo
Cardiovascular Prevention and Pharmacotherapy.2021; 3(4): 115. CrossRef - TRAINING IN BIG DATA TECHNOLOGIES OF MEDICAL UNIVERSITY STUDENTS
K.S ITINSON
AZIMUTH OF SCIENTIFIC RESEARCH: PEDAGOGY AND PSYCHOLOGY.2021;[Epub] CrossRef - Machine Learning Applications in Endocrinology and Metabolism Research: An Overview
Namki Hong, Heajeong Park, Yumie Rhee
Endocrinology and Metabolism.2020; 35(1): 71. CrossRef - Lessons from Use of Continuous Glucose Monitoring Systems in Digital Healthcare
Hun-Sung Kim, Kun-Ho Yoon
Endocrinology and Metabolism.2020; 35(3): 541. CrossRef - Apprehensions about Excessive Belief in Digital Therapeutics: Points of Concern Excluding Merits
Hun-Sung Kim
Journal of Korean Medical Science.2020;[Epub] CrossRef - Medical Ethics in the Era of Artificial Intelligence Based on Medical Big Data
Hae-Ran Na, Hun-Sung Kim
The Journal of Korean Diabetes.2020; 21(3): 126. CrossRef - Machine Learning Application in Diabetes and Endocrine Disorders
Namki Hong, Heajeong Park, Yumie Rhee
The Journal of Korean Diabetes.2020; 21(3): 130. CrossRef - Real World Data and Artificial Intelligence in Diabetology
Kwang Joon Kim
The Journal of Korean Diabetes.2020; 21(3): 140. CrossRef
- Current status of remote collaborative care for hypertension in medically underserved areas


 KES
KES

 First
First Prev
Prev



