Search
- Page Path
- HOME > Search
- Adrenal gland
- The Fascinating Interplay between Growth Hormone, Insulin-Like Growth Factor-1, and Insulin
- Eline C. Nijenhuis-Noort, Kirsten A. Berk, Sebastian J. C. M. M. Neggers, Aart J. van der Lely
- Endocrinol Metab. 2024;39(1):83-89. Published online January 9, 2024
- DOI: https://doi.org/10.3803/EnM.2024.101
- 1,652 View
- 124 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
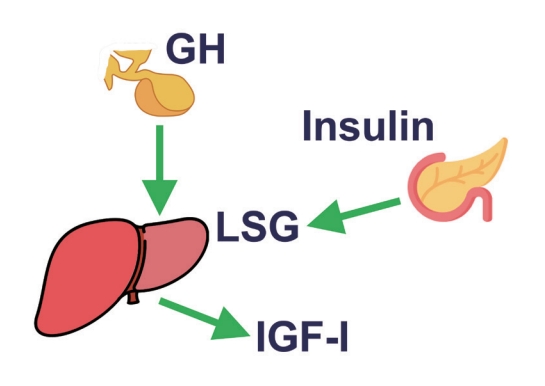
ePub - This review intends to provide the reader with a practical overview of several (patho)physiological conditions in which knowledge of the interplay between growth hormone (GH), insulin-like growth factor-1 (IGF-1), and insulin is important. This might help treating physicians in making the right decisions on how to intervene and improve metabolism for the benefit of patients, and to understand why and how metabolism responds in their specific cases. We will specifically address the interplay between GH, IGF-1, and insulin in type 1 and 2 diabetes mellitus, liver cirrhosis, and acromegaly as examples in which this knowledge is truly necessary.
-
Citations
Citations to this article as recorded by- IGF-1 and IGF-2 as Molecules Linked to Causes and Consequences of Obesity from Fetal Life to Adulthood: A Systematic Review
Justyna Szydlowska-Gladysz, Adrianna Edyta Gorecka, Julia Stepien, Izabela Rysz, Iwona Ben-Skowronek
International Journal of Molecular Sciences.2024; 25(7): 3966. CrossRef
- IGF-1 and IGF-2 as Molecules Linked to Causes and Consequences of Obesity from Fetal Life to Adulthood: A Systematic Review

- Calcium & bone metabolism
- Acromegaly and Bone: An Update
- Andrea Giustina
- Endocrinol Metab. 2023;38(6):655-666. Published online December 22, 2023
- DOI: https://doi.org/10.3803/EnM.2023.601
- 1,323 View
- 109 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
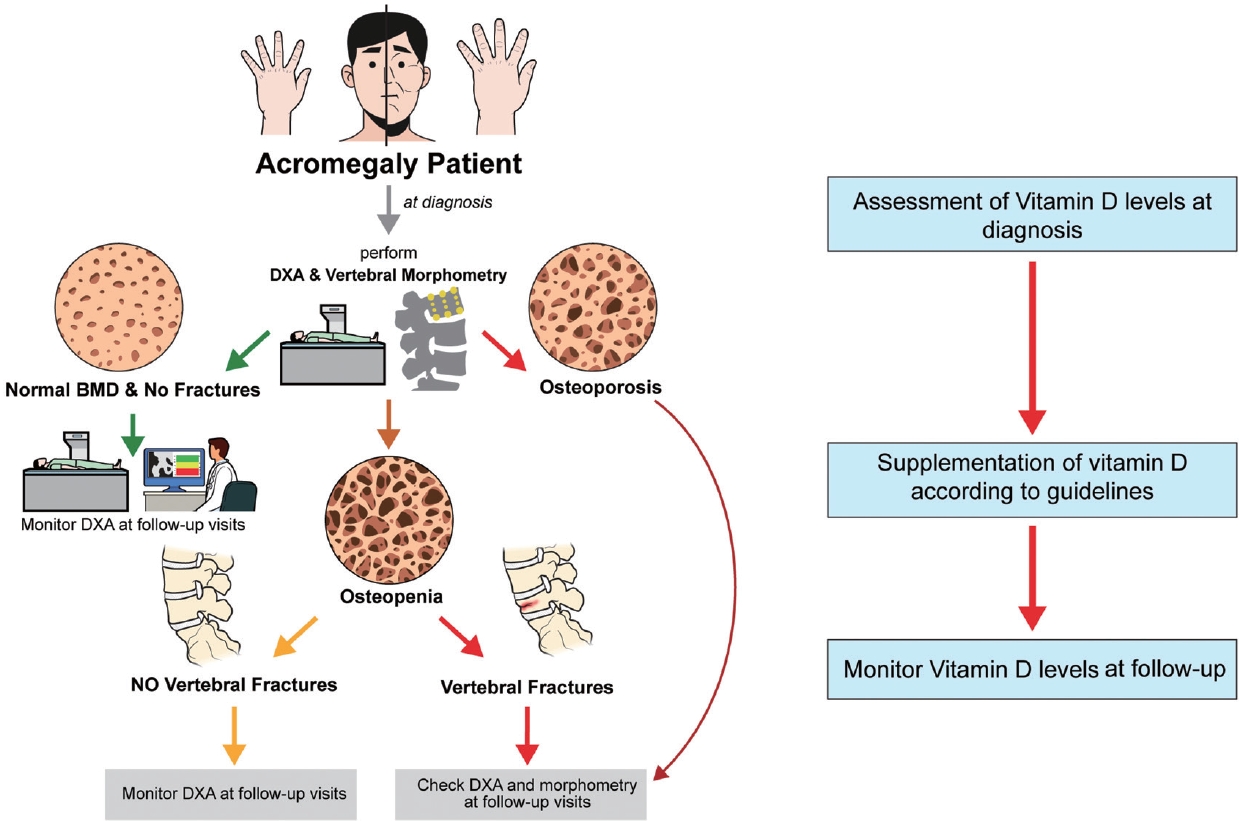
ePub - Since our discovery in 2006 that acromegaly is associated with an increased risk of vertebral fractures, many authors have confirmed this finding in both cross-sectional and prospective studies. Due to the high epidemiological and clinical impact of this newly discovered comorbidity of acromegaly, this topic has progressively become more important and prominent over the years, and the pertinent literature has been enriched by new findings on the pathophysiology and treatment. The aim of this narrative review was to discuss these novel findings, integrating them with the seminal observations, in order to give the reader an updated view of how the field of acromegaly and bone is developing, from strong clinical observations to a mechanistic understanding and possible prevention and treatment.
-
Citations
Citations to this article as recorded by- New insights into the vitamin D/PTH axis in endocrine-driven metabolic bone diseases
Luigi di Filippo, John P. Bilezikian, Ernesto Canalis, Umberto Terenzi, Andrea Giustina
Endocrine.2024;[Epub] CrossRef
- New insights into the vitamin D/PTH axis in endocrine-driven metabolic bone diseases

- Clinical Study
- Associations of GNAS Mutations with Surgical Outcomes in Patients with Growth Hormone-Secreting Pituitary Adenoma
- Hyein Jung, Kyungwon Kim, Daham Kim, Ju Hyung Moon, Eui Hyun Kim, Se Hoon Kim, Cheol Ryong Ku, Eun Jig Lee
- Endocrinol Metab. 2021;36(2):342-350. Published online March 23, 2021
- DOI: https://doi.org/10.3803/EnM.2020.875

- 4,346 View
- 143 Download
- 4 Web of Science
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
The guanine nucleotide-binding protein, alpha stimulating (GNAS) gene has been associated with growth hormone (GH)-secreting pituitary adenoma. We investigated the prevalence of GNAS mutations in Korean patients with acromegaly and assessed whether mutation status correlated with biochemical or clinical characteristics.
Methods
We studied 126 patients with acromegaly who underwent surgery between 2005 and 2014 at Severance Hospital. We performed GNAS gene analysis and evaluated age, sex, hormone levels, postoperative biochemical remission, and immunohistochemical staining results of the tumor.
Results
GNAS mutations were present in 75 patients (59.5%). Patients with and without GNAS mutations showed similar age distribution and Knosp classification. The proportion of female patients was 76.5% and 48.0% in the GNAS-negative and GNAS-mutation groups, respectively (P=0.006). In immunohistochemical staining, the GNAS-mutation group showed higher GH expression in pituitary tumor tissues than the mutation-negative group (98.7% vs. 92.2%, P=0.015). Patients with GNAS mutations had higher preoperative insulin-like growth factor-1 levels (791.3 ng/mL vs. 697.0 ng/mL, P=0.045) and lower immediate postoperative basal (0.9 ng/mL vs. 1.0 ng/mL, P=0.191) and nadir GH levels (0.3 ng/mL vs. 0.6 ng/mL, P=0.012) in oral glucose tolerance tests. Finally, the GNAS-mutation group showed significantly higher surgical remission rates than the mutation-negative group, both at 1 week and 6 months after surgical resection (70.7% vs. 54.9%, P=0.011; 85.3% vs. 82.4%, P=0.007, respectively).
Conclusion
GNAS mutations in GH-secreting pituitary tumors are associated with higher preoperative insulin-like growth factor-1 levels and surgical remission rates and lower immediate postoperative nadir GH levels. Thus, GNAS mutation status can predict surgical responsiveness in patients with acromegaly. -
Citations
Citations to this article as recorded by- Genetic diagnosis in acromegaly and gigantism: From research to clinical practice
Claudia Ramírez-Rentería, Laura C. Hernández-Ramírez
Best Practice & Research Clinical Endocrinology & Metabolism.2024; : 101892. CrossRef - CD8/PD-L1 immunohistochemical reactivity and gene alterations in cutaneous squamous cell carcinoma
Haruto Nishida, Yoshihiko Kondo, Takahiro Kusaba, Kazuhiro Kawamura, Yuzo Oyama, Tsutomu Daa, Avaniyapuram Kannan Murugan
PLOS ONE.2023; 18(2): e0281647. CrossRef - Dynamic monitoring of circulating tumor DNA to analyze genetic characteristics and resistance profile of lorlatinib in ALK positive previously treated NSCLC
Xiya Ma, Kun Zhang, Jing Xu, Hongjun Gao, Shaoxing Yang, Haifeng Qin, Hong Wang, Fang Gao, Xiaoqing Liu
Thoracic Cancer.2023; 14(20): 1980. CrossRef - Multiomics Approach to Acromegaly: Unveiling Translational Insights for Precision Medicine
Kyungwon Kim, Cheol Ryong Ku, Eun Jig Lee
Endocrinology and Metabolism.2023; 38(5): 463. CrossRef - Hotspots of Somatic Genetic Variation in Pituitary Neuroendocrine Tumors
Mariana Torres-Morán, Alexa L. Franco-Álvarez, Rosa G. Rebollar-Vega, Laura C. Hernández-Ramírez
Cancers.2023; 15(23): 5685. CrossRef
- Genetic diagnosis in acromegaly and gigantism: From research to clinical practice

- Hypothalamus and Pituitary gland
- Reference Ranges of Serum Insulin-Like Growth Factor-I and Insulin-Like Growth Factor Binding Protein-3: Results from a Multicenter Study in Healthy Korean Adults
- In-Kyung Jeong, Jong Kyu Byun, Junghyun Noh, Sang Wan Kim, Yoon-Sok Chung, Tae Sun Park, Sung-Woon Kim
- Endocrinol Metab. 2020;35(4):954-959. Published online December 1, 2020
- DOI: https://doi.org/10.3803/EnM.2020.785
- 4,418 View
- 125 Download
- 2 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
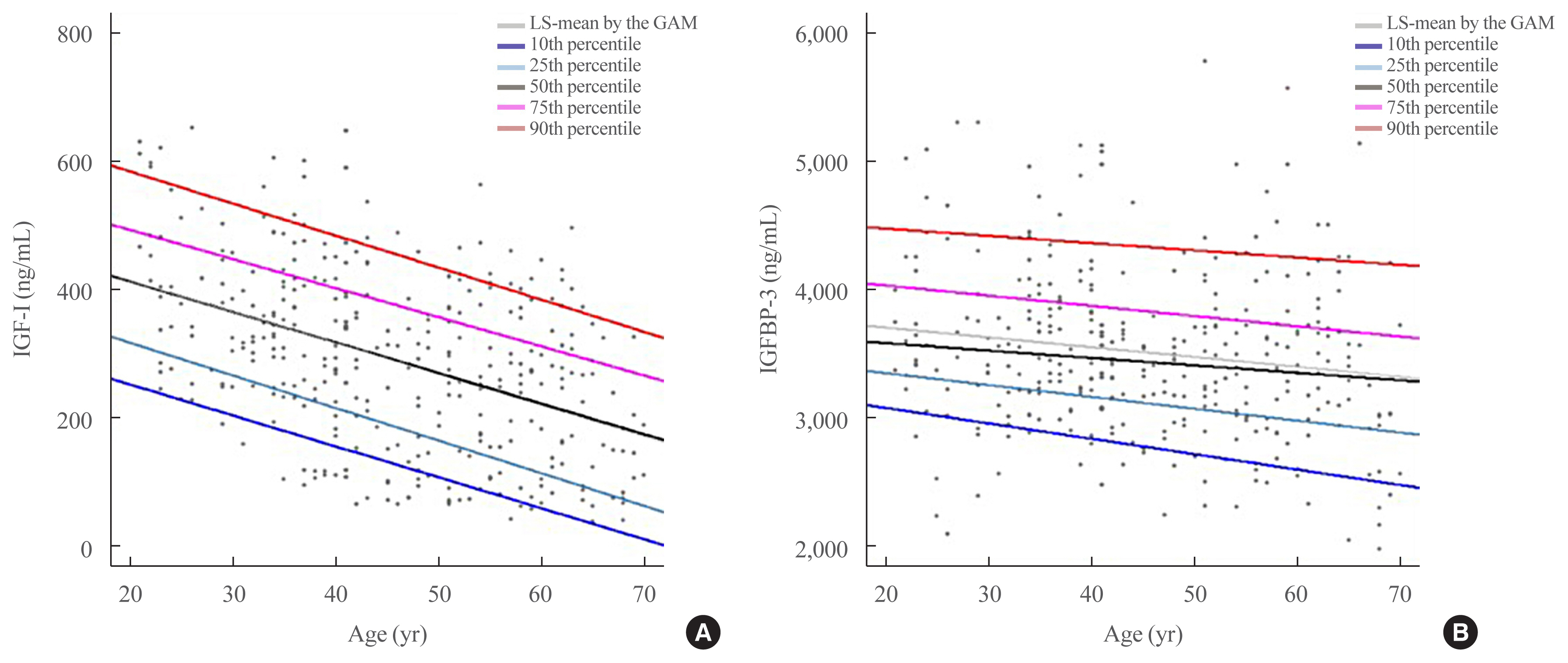
ePub - Insulin-like growth factor-I (IGF-I) plays a pivotal role in the diagnosis and treatment of growth hormone (GH) excess or deficiency. The GH study group of the Korean Endocrine Society aims to establish the Korean reference ranges of serum IGF-I and insulin-like growth factor binding protein-3 (IGFBP-3) and assess the relationship between IGF-I and IGFBP-3 and clinical parameters. Fasting serum was collected from healthy Korean adults at health promotion centers of five hospitals nationwide. Serum IGF-I and IGFBP-3 were measured via an immunoradiometric assay using a DSL kit (Diagnostic Systems Laboratories). Serum samples from 354 subjects (180 male, 174 female) were analyzed based on sex at 10-year intervals from 21 to 70 years. IGF-I levels were inversely correlated with age. After adjustment of age, the IGF-I/IGFBP-3 ratio was significantly negatively associated with blood pressure and free thyroxine and positively associated with weight, hemoglobin, creatinine, alanine transferase, fasting glucose, and thyroid stimulating hormone. Therefore, age- and sex-specific reference ranges of serum IGF-I and IGFBP-3 can be efficient in evaluating GH excess or deficiency in Korean population.
-
Citations
Citations to this article as recorded by- Capillary blood as a complementary matrix for doping control purposes. Application to the definition of the individual longitudinal profile of IGF-1
Carlotta Stacchini, Francesco Botrè, Xavier de la Torre, Monica Mazzarino
Journal of Pharmaceutical and Biomedical Analysis.2023; 227: 115274. CrossRef
- Capillary blood as a complementary matrix for doping control purposes. Application to the definition of the individual longitudinal profile of IGF-1

- Hypothalamus and Pituitary gland
- Establishment of Reference Intervals for Serum Insulin-Like Growth Factor I in Korean Adult Population
- Boyeon Kim, Yonggeun Cho, Cheol Ryong Ku, Sang-Guk Lee, Kyung-A Lee, Jeong-Ho Kim
- Endocrinol Metab. 2020;35(4):960-964. Published online November 20, 2020
- DOI: https://doi.org/10.3803/EnM.2020.732

- 4,768 View
- 107 Download
- 4 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Appropriate reference intervals of serum insulin-like growth factor I (IGF-I) is important for diagnosing and monitoring patients with growth hormone-related diseases. To establish reference intervals, adult individuals (n=1,334, 680 men and 654 women) were divided into six age groups (20–29, 30–39, 40–49, 50–59, 60–69, ≥70). Serum IGF-I was measured by chemiluminescence immunoassay (Liaison). Concordance of patient classification based on reference intervals, manufacturer’s intervals, and standard deviation score (SDS) was evaluated. New reference intervals had higher upper and lower limits than those specified by the manufacturer. The agreement between classification using new reference interval and the manufacturer’s reference interval, and that using new reference interval and SDS was 75.0% (weighted kappa, 0.17), 91.9% (weighted kappa, 0.51) in men and 91.0% (weighted kappa, 0.41), 92.5% (weighted kappa, 0.53) in women, respectively. Reference intervals should be established not only based on age and sex, but also on ethnicity and assay method.
-
Citations
Citations to this article as recorded by- Challenges of insulin-like growth factor-1 testing
Rongrong Huang, Junyan Shi, Ruhan Wei, Jieli Li
Critical Reviews in Clinical Laboratory Sciences.2024; : 1. CrossRef - Male idiopathic hypogonadotropic hypogonadism: Serum insulin-like growth factor-1 and oestradiol levels
Nastaran Salehisedeh, Amir Parhizkar, Parichehreh Yaghmaei, Marjan Sabbaghian
Journal of Human Reproductive Sciences.2022; 15(4): 351. CrossRef - Revisiting the Role of Insulin-like Growth Factor-1 Measurement After Surgical Treatment of Acromegaly
In-Ho Jung, Seonah Choi, Cheol Ryong Ku, Sang-Guk Lee, Eun Jig Lee, Sun Ho Kim, Eui Hyun Kim
The Journal of Clinical Endocrinology & Metabolism.2021; 106(7): e2589. CrossRef
- Challenges of insulin-like growth factor-1 testing

- Endocrine Research
- Therapeutic Effect of a Novel Chimeric Molecule Targeting Both Somatostatin and Dopamine Receptors on Growth Hormone-Secreting Pituitary Adenomas
- Jean Kim, Ju Hun Oh, Heather Harlem, Michael D. Culler, Cheol Ryong Ku, Eun Jig Lee
- Endocrinol Metab. 2020;35(1):177-187. Published online March 19, 2020
- DOI: https://doi.org/10.3803/EnM.2020.35.1.177
- 4,496 View
- 106 Download
- 4 Web of Science
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub Background Acromegaly is a rare disease primarily caused by growth hormone (GH)-secreting pituitary adenomas, and its treatment is costly. Moreover, some patients are unresponsive to treatment. Hence, there are increasing efforts to develop new drugs with improved effectiveness for this disease. BIM23B065 is a novel chimeric molecule that acts on both somatostatin and dopamine receptors. This study aimed to investigate the effects of BIM23B065 compared with those of a somatostatin receptor analog and a dopamine agonist.
Methods The effects of BIM23B065 on the proliferation, GH and insulin-like growth factor-1 (IGF-1) levels, and extracellular signal-regulated kinase (ERK) 1/2 and cyclic AMP response element binding (CREB) phosphorylation of GH3 cells were investigated with MTS assay, enzyme-linked immunosorbent assay, and Western blotting, respectively. The dosage and treatment duration of BIM23B065 were tested in animal models of GH-secreting pituitary adenoma. The effect of BIM23B065 (3 mg/kg/day) on changes in IGF-1 levels before and after treatment was further investigated.
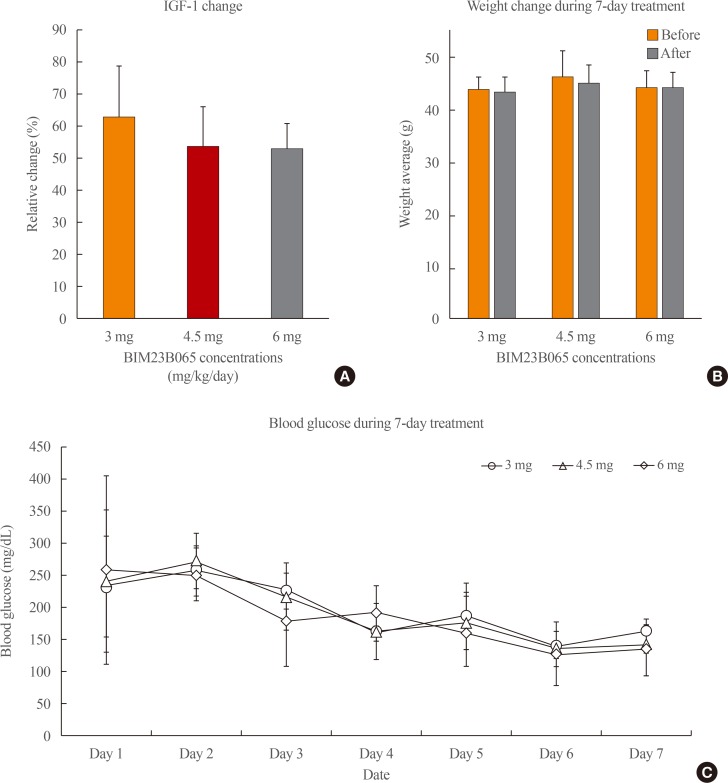
Results In vitro , BIM23B065 treatment decreased GH release in the culture media and downregulated ERK 1/2 and CREB phosphorylation to 22% and 26%, respectively.In vivo , IGF-1 expression decreased to 50 % after 4 weeks of treatment with BIM23B065 using an osmotic pump implant. Moreover, magnetic resonance imaging results showed that the tumor size decreased significantly following treatment with BIM23B065 for 4 weeks.Conclusion The novel chimeric molecule was effective in decreasing IGF-1 and GH levels and may serve as an effective therapeutic agent for acromegaly.
-
Citations
Citations to this article as recorded by- Pituitary Tumorigenesis—Implications for Management
Rodanthi Vamvoukaki, Maria Chrysoulaki, Grigoria Betsi, Paraskevi Xekouki
Medicina.2023; 59(4): 812. CrossRef - Current and Emerging Medical Therapies in Pituitary Tumors
Nicolas Sahakian, Frédéric Castinetti, Thierry Brue, Thomas Cuny
Journal of Clinical Medicine.2022; 11(4): 955. CrossRef - Characterization of the ability of a, second-generation SST-DA chimeric molecule, TBR-065, to suppress GH secretion from human GH-secreting adenoma cells
Thomas Cuny, Thomas Graillon, Célines Defilles, Rakesh Datta, Shengwen Zhang, Dominique Figarella-Branger, Henry Dufour, Grégory Mougel, Thierry Brue, Tanya Landsman, Heather A. Halem, Michael D. Culler, Anne Barlier, Alexandru Saveanu
Pituitary.2021; 24(3): 351. CrossRef - Efficacy of a Novel Second-Generation Somatostatin-Dopamine Chimera (TBR-065) in Human Medullary Thyroid Cancer: A Preclinical Study
Alessandra Dicitore, Maria Celeste Cantone, Germano Gaudenzi, Davide Saronni, Silvia Carra, Maria Orietta Borghi, Manuela Albertelli, Diego Ferone, Leo J. Hofland, Luca Persani, Giovanni Vitale
Neuroendocrinology.2021; 111(10): 937. CrossRef - Emerging drugs for the treatment of acromegaly
Claudia Campana, Giuliana Corica, Federica Nista, Francesco Cocchiara, Giulia Graziani, Keyvan Khorrami, Marta Franco, Mara Boschetti, Diego Ferone, Federico Gatto
Expert Opinion on Emerging Drugs.2020; 25(4): 409. CrossRef
- Pituitary Tumorigenesis—Implications for Management

- Adrenal gland
- Multiple Myeloma in a Patient with Acromegaly
- Yu Mi Kang, Jong Han Choi, Min Jung Lee, Ari Ahn, Chan-Jeoung Park, Kiju Chang, Seyoung Seo, Sun In Hong, Min-Seon Kim
- Endocrinol Metab. 2015;30(1):110-115. Published online March 27, 2015
- DOI: https://doi.org/10.3803/EnM.2015.30.1.110
- 5,006 View
- 36 Download
- 2 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader Acromegaly is a slowly progressing condition resulting from excess growth hormone (GH), generally caused by a GH-secreting pituitary adenoma. Cancer is the third most common cause of mortality in patients with acromegaly, and insulin-like growth factor 1 (IGF-1) is known to influence tumor formation by increasing cell proliferation and inhibiting apoptosis. Multiple myeloma (MM) is a plasma cell neoplasm, and previous studies have suggested the possible role of IGF-1 in its development of MM. However, no cases of acromegaly accompanied with MM have been reported in Asia to date. We here report the case of a 58-year-old woman with acromegaly accompanied with MM who presented with longstanding acromegalic manifestations resulting from a GH-secreting pituitary adenoma and also exhibited anemia, a reversed albumin/globulin ratio, and plasmacytosis on bone marrow examination. Because IGF-1 has been suggested to play an important role in the development and progression of MM, the patient promptly underwent surgical removal of the pituitary adenoma via a transsphenoidal approach. Since there is currently no consensus on therapeutic guidelines and suggested prognosis for MM with acromegaly, long-term follow-up of such cases is needed.
-
Citations
Citations to this article as recorded by- Co-Occurrence of Acromegaly and Hematological Disorders: A Myth or Common Pathogenic Mechanism
Prakamya Gupta, Pinaki Dutta
Integrative Medicine International.2017; 4(1-2): 94. CrossRef
- Co-Occurrence of Acromegaly and Hematological Disorders: A Myth or Common Pathogenic Mechanism

- Adrenal gland
- Acromegaly with Normal Insulin-Like Growth Factor-1 Levels and Congestive Heart Failure as the First Clinical Manifestation
- Hyae Min Lee, Sun Hee Lee, In-Ho Yang, In Kyoung Hwang, You Cheol Hwang, Kyu Jeung Ahn, Ho Yeon Chung, Hui-Jeong Hwang, In-Kyung Jeong
- Endocrinol Metab. 2015;30(3):395-401. Published online December 9, 2014
- DOI: https://doi.org/10.3803/EnM.2015.30.3.395
- 4,215 View
- 44 Download
- 7 Web of Science
- 4 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader The leading cause of morbidity and mortality in patients with acromegaly is cardiovascular complications. Myocardial exposure to excessive growth hormone can cause ventricular hypertrophy, hypertension, arrhythmia, and diastolic dysfunction. However, congestive heart failure as a result of systolic dysfunction is observed only rarely in patients with acromegaly. Most cases of acromegaly exhibit high levels of serum insulin-like growth factor-1 (IGF-1). Acromegaly with normal IGF-1 levels is rare and difficult to diagnose. Here, we report a rare case of an acromegalic patient whose first clinical manifestation was severe congestive heart failure, despite normal IGF-1 levels. We diagnosed acromegaly using a glucose-loading growth hormone suppression test. Cardiac function and myocardial hypertrophy improved 6 months after transsphenoidal resection of a pituitary adenoma.
-
Citations
Citations to this article as recorded by- Risk of Neurodegenerative Diseases in Patients With Acromegaly
Sangmo Hong, Kyungdo Han, Kyung-Soo Kim, Cheol-Young Park
Neurology.2022;[Epub] CrossRef - Levels of Serum IGF-1, HCY, and Plasma BNP in Patients with Chronic Congestive Heart Failure and Their Relationship with Cardiac Function and Short-Term Prognosis
Zhengyi Hu, Leifang Mao, Ling Wang, Weiguo Li
Evidence-Based Complementary and Alternative Medicine.2022; 2022: 1. CrossRef - Neurofibromatosis Type 1 with Concurrent Multiple Endocrine Disorders: Adenomatous Goiter, Primary Hyperparathyroidism, and Acromegaly
Shigemitsu Yasuda, Ikuo Inoue, Akira Shimada
Internal Medicine.2021; 60(15): 2451. CrossRef - Metformin stimulates IGFBP-2 gene expression through PPARalpha in diabetic states
Hye Suk Kang, Ho-Chan Cho, Jae-Ho Lee, Goo Taeg Oh, Seung-Hoi Koo, Byung-Hyun Park, In-Kyu Lee, Hueng-Sik Choi, Dae-Kyu Song, Seung-Soon Im
Scientific Reports.2016;[Epub] CrossRef
- Risk of Neurodegenerative Diseases in Patients With Acromegaly


 KES
KES

 First
First Prev
Prev



