Articles
- Page Path
- HOME > Endocrinol Metab > Volume 30(4); 2015 > Article
-
Original ArticleClinical Study Economic Evaluation of Recombinant Human Thyroid Stimulating Hormone Stimulation vs. Thyroid Hormone Withdrawal Prior to Radioiodine Ablation for Thyroid Cancer: The Korean Perspective
- Seo Young Sohn1*, Hye Won Jang2*, Yoon Young Cho3, Sun Wook Kim3, Jae Hoon Chung3
-
Endocrinology and Metabolism 2015;30(4):531-542.
DOI: https://doi.org/10.3803/EnM.2015.30.4.531
Published online: December 31, 2015
1Division of Endocrinology, Department of Medicine, Myongji Hospital, Seonam University College of Medicine, Goyang, Korea.
2Department of Social and Preventive Medicine, Sungkyunkwan University School of Medicine, Seoul, Korea.
3Division of Endocrinology and Metabolism, Department of Medicine, Thyroid Center, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- Corresponding author: Jae Hoon Chung. Division of Endocrinology and Metabolism, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, 81 Irwon-ro, Gangnam-gu, Seoul 06351, Korea. Tel: +82-2-3410-3434, Fax: +82-2-3410-3849, thyroid@skku.edu
- *These authors contributed equally to this work.
Copyright © 2015 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/3.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- Previous studies have suggested that recombinant human thyroid stimulating hormone (rhTSH) stimulation is an acceptable alternative to thyroid hormone withdrawal (THW) when radioiodine remnant ablation is planned for thyroid cancer treatment, based on superior short-term quality of life with non-inferior remnant ablation efficacy. This study evaluated the cost-effectiveness of radioiodine remnant ablation using rhTSH, compared with the traditional preparation method which renders patients hypothyroid by THW, in Korean perspective.
-
Methods
- This economic evaluation considered the costs and benefits to the Korean public healthcare system. Clinical experts were surveyed regarding the current practice of radioiodine ablation in Korea and their responses helped inform assumptions used in a cost effectiveness model. Markov modelling with 17 weekly cycles was used to assess the incremental costs per quality-adjusted life year (QALY) associated with rhTSH. Clinical inputs were based on a multi-center, randomized controlled trial comparing remnant ablation success after rhTSH preparation with THW. The additional costs associated with rhTSH were considered relative to the clinical benefits and cost offsets.
-
Results
- The additional benefits of rhTSH (0.036 QALY) are achieved with an additional cost of Korean won ₩961,105, equating to cost per QALY of ₩26,697,361. Sensitivity analyses had only a modest impact upon cost-effectiveness, with one-way sensitivity results of approximately ₩33,000,000/QALY.
-
Conclusion
- The use of rhTSH is a cost-effective alternative to endogenous hypothyroid stimulation prior to radioiodine ablation for patients who have undergone thyroidectomy in Korea.
- Total or near-total thyroidectomy followed by 131I radioiodine ablation constitutes the initial treatment for differentiated thyroid cancer (DTC) [12]. The aim of radioiodine ablation is to facilitate detection of recurrent disease and to decrease the risk of recurrence by destroying remnant thyroid tissue [2]. For ablation to be successful, patients must have elevated levels of thyroid stimulating hormone (TSH). Traditionally, endogenous stimulation by thyroid hormone withdrawal (THW) has been used to elevate serum TSH, which can take 3 to 8 weeks after thyroidectomy. Although hypothyroidism by THW elevates serum TSH levels suitable for radioiodine uptake, it is commonly associated with a negative impact on quality of life in DTC patients [3456]. Symptoms of induced hypothyroidism by THW may be severe and can prevent some patients from performing usual daily activities. Also, there is considerable evidence suggesting endogenous stimulation results in greater work absenteeism [47].
- An alternative to rendering the patients hypothyroid is exogenous stimulation of TSH using recombinant human thyroid stimulating hormone (rhTSH; Thyrogen, Genzyme Transgenics Corp., Cambridge, MA, USA). This allows patients to continue thyroid hormone replacement with thyroxine (T4) after thyroidectomy and therefore renders the patients euthyroid. Recent randomized prospective studies have reported that rhTSH was as effective as THW for successful ablation in low-dose and high-dose radioiodine ablation [89]. Furthermore, some studies indicate considerable quality of life benefits with exogenous stimulation using rhTSH [10]. However, exogenous stimulation with rhTSH is more costly than endogenous stimulation. Therefore, economic evaluation is necessary whether the use of rhTSH is cost effective alternative to THW to offset the incremental cost.
INTRODUCTION
- Economic model structure
- The economic model is based on an analysis published by Mernagh et al. [11] that was previously modified for the Swedish market, and that supported reimbursement. The present economic model takes a healthcare system perspective, capturing only those costs and outcomes that directly impact the Korean public healthcare system.
- This economic evaluation was undertaken to assess the incremental benefits gained from rhTSH in the course of ablation, in the context of costs relative to patient benefit. The benefits were expressed in units of quality-adjusted life years (QALYs) using "utility weights" from 0 (death) to 1 (perfect health), a standard method in health economics.
- The model utilized a Markov chain structure to simulate the costs and outcomes of low to intermediate risk thyroid cancer patients requiring 100 mCi of radioiodine ablation. The model consisted of two arms: one with exogenous stimulation with rhTSH, the other with endogenous stimulation. A Markov model is a well-accepted method of economic evaluation for following patients as they move between different health states.
- The model was run for 17 one-week cycles. This 17-week duration is a departure from a previous version by Mernagh et al. [11], which followed patients over the course of a lifetime and suggested the theoretical advantage of a reduction in secondary malignancies because of faster clearance of radioiodine from the body. The 17-week evaluation enables all immediate and downstream quality of life benefits offered by rhTSH to be adequately captured. That is, the avoidance of hypothyroidism and the more rapid recovery to full health are captured. Furthermore, as all differences in costs and quality of life are already captured, the model ceases at this point. The relatively short duration of the model is reflective of the short-term nature of the quality of life gains offered, which is in itself, reflective of the short-term morbidity caused by hypothyroidism. Half-cycle correction is not employed as patients are not able to move between health states in either the first or final stages of the model.
- The model only relates to low- to intermediate-risk radioiodine ablation in thyroid cancer patients similar to those in the pivotal randomized controlled study [10], and does not include patients with stage IV or distant metastases.
- There are key assumptions in the economic evaluation, as follows: (1) the model assumes equal efficacy of ablation (100%) in both arms; this assumption is based on the pivotal clinical trial by Pacini et al. [10]; (2) It was assumed that exogenously-stimulated patients are ablated 1 week following thyroidectomy, while endogenously stimulated patients are ablated at various points in time, often more than 4 weeks after the thyroidectomy; the latter is based on a survey conducted among Korean clinicians describing their current practice; (3) the model applies quality of life results observed over time in each arm of the pivotal clinical trial (SF-6D results, reported by Schroeder et al. [12]); (4) patients are released from the radioprotective ward earlier when prepared for ablation via exogenous stimulation, based on findings by Borget et al. [13] and Pacini et al. [10]; and (5) the cost of radioactive waste disposal has not been independently estimated. Instead, it is assumed to have been captured in the fees for ablation and/or radio-protective ward stay.
- In Korea, patients are eligible for a one-time reimbursement for rhTSH if they meet one the following criteria: (1) have experienced a serious adverse event due to T4 withdrawal in previous treatment; (2) are over 65 years old; (3) have cardiac or pulmonary dysfunction; (4) have hypopituitarism; (5) show evidence of fast-growing cancer; or (6) underwent a total or near total thyroidectomy for remnant ablation. The last criterion was added in the revised Korea reimbursement system [14].
- Economic model variables
- The estimation of a number of the variables used to populate the economic model relied upon the results of a treatment survey conducted among clinical experts currently practicing endocrinology, nuclear medicine, and surgery in Korea (Appendix 1).
- The objective of the survey was to establish the current practice of radioiodine ablation (100 mCi) of well-DTC, in Korean patients who are endogenously-stimulated prior to ablation. Optum Australia developed the survey questionnaire and conducted the data analysis. The survey itself was administered by a local representative of Genzyme, based in Korea. The survey consisted of twelve questions, eleven of which related to endogenously-stimulated patients and one related to the administration of rhTSH. Though the majority of questions were framed to generate a quantitative response, qualitative answers were appropriate in some cases. The survey was conducted via face-to-face interviews with six experts based in endocrinology, nuclear medicine or surgical departments around Korea. Each clinical expert was presented with the survey during a meeting with a Genzyme representative. A note describing the purpose and rationale behind each question was also provided to the interviewer.
- In general, the survey was well understood by all of the clinical experts, which was reflected by the consistency of the answers given. The six experts treated a total of approximately 2,000 newly diagnosed thyroid cancer patients with radioiodine ablation each year. Standard protocol dictates that patients undergo approximately 4 weeks of levothyroxine withdrawal prior to radioiodine ablation. All six clinicians indicated that 100% of their patients would commence T4 replacement therapy directly after thyroidectomy, to be withdrawn at a later time. The aim of this question was to assess the extent of potential supply constraints in terms of radio-ablation facilities in the Korean healthcare system. The indication that all thyroidectomy patients would commence T4 immediately after surgery suggests that this may be a substantial issue in Korea. However, the duration of T4 replacement therapy prior to withdrawal was not defined in the responses to the treatment survey. Since the availability of radio-ablation facilities is independent of the choice of pre-ablative treatment, it would affect both arms in the model equally. As such, this pre-ablative T4 supplementation period is assumed to occur prior to the beginning of the modelled evaluation; and thus, it does not impact the duration or outcomes of the evaluation itself. The majority of respondents also indicated that patients undergoing endogenous hypothyroid stimulation are not completely withdrawn from thyroid hormone replacement therapy. Rather, patients transition from T4 to a 2-week treatment of triiodothyronine (T3) therapy, followed by complete withdrawal. After transition to T3 therapy, the majority of patients are ablated within 4 weeks. All patients were ablated within 6 weeks.
- Clinical inputs
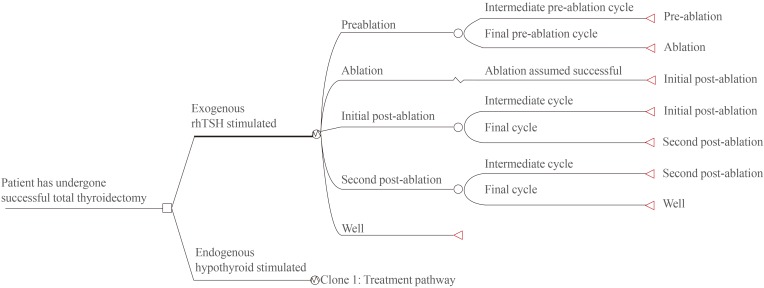
- Four distinct health states in each of the two arms are incorporated into the economic model (Table 1). A schematic of the model is depicted in Fig. 1. The interval between thyroidectomy and ablation for endogenously-stimulated patients was determined via the clinician survey described above. As patients can only undergo ablation once TSH levels have naturally elevated to a suitable level, it is intuitive that the interval will vary from patient to patient. As a consequence, the model enables patients to move from pre-ablation to ablation according to a set of probabilities. The clinician survey was used to derive these probabilities, which are presented in Table 2. Note that the results of the survey generate an expected mean duration of approximately 4.4 weeks (standard deviation ±1.12 weeks).
- With respect to exogenously-stimulated patients, radioiodine ablation was assumed to take place closer to the time of total thyroidectomy (which occurs prior to the beginning of the model). This assumption is consistent with previous iterations of the economic model, and was a consequence of discussions with clinicians who indicated that ablation would occur earlier, presumably because: (1) patients would no longer need to wait several weeks for their TSH levels to be suitably elevated and (2) the clinical aim is to ablate any cancer remnants as soon as possible to reduce the risk of metastases. One week is required; however, to allow adequate time for recovery from thyroidectomy.
- The results of the survey were also used to inform the duration of time spent in the radioprotective ward following ablation for endogenously-stimulated patients. As with the time between thyroidectomy and ablation, these results varied from patient to patient. The weighted average proportion of patients, and the overall average expected duration in a radioprotective ward, is presented in Table 3.
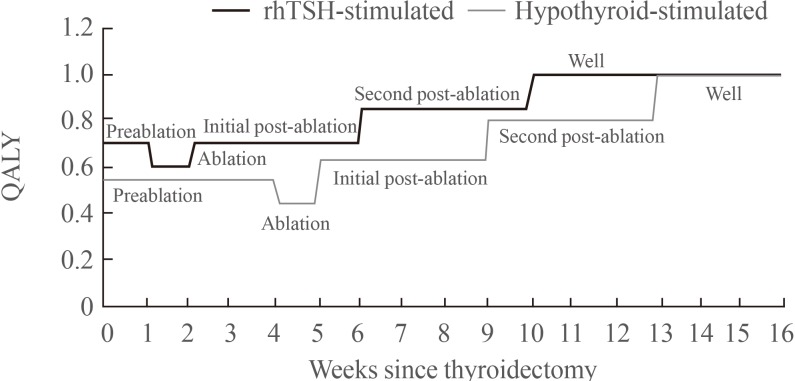
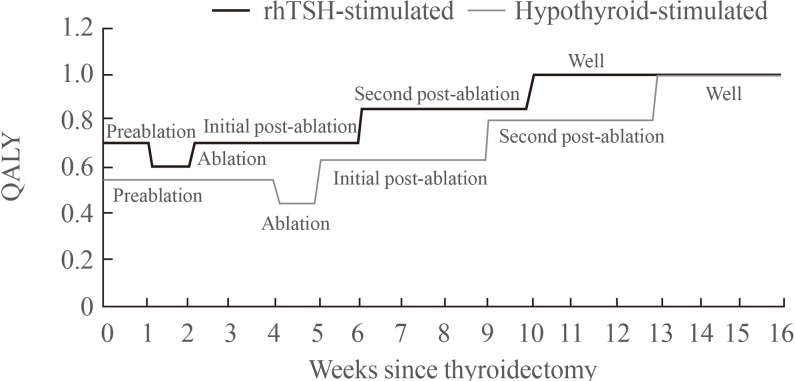
- The utility weights applied to the economic model were based upon the SF-36 data collected during the randomized controlled trial in the ablation setting [10]. SF-36 results are widely reported in the literature, consistently showing a significant impairment in quality of life with induced hypothyroidism [1215]. These data were transformed to SF-6D QALY weights based on the method outlined by Brazier et al. [16]. Fig. 2 presents a summary of the utility weights associated with each of the health states.
- Economic inputs
- The economic inputs used in the model are presented in Table 4. All unit costs are presented in South Korean won (₩). The costs applied to each arm of the economic model are presented in Table 5. The main difference between the two treatment groups is the presence of rhTSH and its administration (2×nurse visits for injections) as a cost in the exogenously-stimulated arm only. Other differences include a lower expected cost of both time spent in the radioprotective ward in the exogenously-stimulated (rhTSH) arm and the cost of 2 weeks of T3 therapy in the endogenously-stimulated arm (THW) during the pre-ablation health state. Four of the six respondents to the Korean treatment survey indicated that for endogenously-stimulated patients, TSH quantification would be performed once, after 4 weeks of T4 withdrawal. If the patient's TSH level had not reached the desired value for ablation, it would not be measured again. This is reflected in the ablation timeframes given in the treatment survey, which showed >99% of patients will be ablated between 4 and 6 weeks following T4 withdrawal, with the majority undergoing ablation at week 4 (72%). Based on these data, the model assumes that patients in the endogenously-stimulated arm of the model receive one TSH quantification test in the pre-ablation health state. The clinician surveys also reveal that a small proportion (1% to 2%) of endogenously-stimulated patients would seek additional treatment from a specialist for treatment of symptoms related to hypothyroidism. A rate of 1.5% was applied to the model to capture the cost of these additional specialist visits.
METHODS
- Base case analysis
- The results of the economic model are presented in Table 6. The cost of rhTSH in this radioiodine ablation setting (₩2,250,393) is higher than endogenous stimulation of TSH (₩1,289,288). The use of rhTSH is also associated with an improved health-related quality of life, as illustrated by an increase in QALY terms (0.036).
- Sensitivity analyses
- A number of sensitivity analyses were conducted to test the effect of altering key assumptions and to determine the key drivers of the model. Many of these tests are extreme scenarios. The results of the sensitivity analyses are presented in Table 7. The analyses suggested that difference in productivity was a major driver of the model results. These sensitivity analyses are discussed in greater detail below.
RESULTS
- The present economic investigation indicates that the use of rhTSH in preparation for radioiodine remnant ablation has economic benefits in addition to health benefits, with an incremental cost-effectiveness ratio (ICER) of ₩26,697,361/QALY. New health care interventions are typically accepted if the ICER is below ₩30,000,000, and therefore the use of rhTSH represents cost-effective technology (i.e., the additional benefits to the patients and society are achieved for an acceptable net cost).
- In addition to improved quality of life for patients, there is also a productivity benefit associated with rhTSH. A number of reports have indicated significantly reduced productivity and increased employment absenteeism in the hypothyroid state compared with the euthyroid state [347]. The sensitivity analysis performed in this economic evaluation takes a similar approach to Mernagh et al. [11], by basing the estimate of work days lost on data from Luster et al. [3]. Luster et al. [3] reported that German patients rendered hypothyroid in the diagnostic setting were absent from work for an average of 11 days. Another study by Borget et al. [7] reported that patients undergoing rhTSH preparation in a diagnostic setting take an average of 3 days off work, compared to 11 days for the hypothyroid group. In a pivotal remnant ablation study, 15.6% of the subjects in the endogenous-stimulation arm reported markedly reduced daily productivity compared with 8.6% of subjects who received exogenously stimulation [17]. Therefore, the loss of work productivity in exogenously-stimulated patients was crudely estimated to be 50% of that of the endogenously-stimulated patients who withdrew from thyroid hormone, i.e., 50% of 11 days, or 5.5 days. It should be noted that, although the Luster et al. [3] data was sourced from patients undergoing diagnostic 131I scanning, they are assumed to be representative of pre-ablation use, because in both cases patients are rendered equally hypothyroid. Considering the number of days of work lost in the rhTSH arm is half of that experienced by patients in the endogenously-stimulated arm, the incremental cost difference between the two groups is reduced with the inclusion of indirect costs. The incremental utility difference is unchanged; therefore, the overall ICER is lower in this scenario, indicating that rhTSH is more cost-effective.
- The most significant cost-offset included in this economic model is that associated with the shorter duration of time spent in the radioprotective ward following radioiodine ablation. The sensitivity analyses demonstrate the impact of the removal of this cost-offset on the overall cost-effectiveness. A number of studies have reported faster clearance of 131I from the body after radioiodine ablation when patients are prepared for ablation with rhTSH rather than endogenous stimulation [10181920]. Regardless of the measure used, the radiation clearance is approximately 35% faster with rhTSH-stimulation than with hypothyroid-stimulation. This is not unexpected given the impairment of renal function that is known to occur with hypothyroidism. Based on a study by Borget et al. [13], the model assumes that hospital discharge occurs 27 hours earlier for patients receiving rhTSH. However, if the advantage of less time spent in the radioprotective ward is ignored entirely, rhTSH generated a modest increase in ICER of ₩33,909,333 per QALY. Even under these extreme assumptions, exogenous stimulation using rhTSH may be acceptable value for money.
- The sensitivity analysis shows that increasing the period between surgery and ablation from 1 to 2 weeks for exogenously-stimulated patients with rhTSH has little impact on overall cost-effectiveness of rhTSH after reimbursement. That said, there is a practical advantage to the shorter duration of time between thyroidectomy and ablation prepared by exogenously-stimulated patients. By removing the requirement for TSH levels to naturally elevate over time, timing of ablation can be more flexible, resulting in fewer scheduling issues. This may free up hospital beds for other uses or for additional patients. However, this advantage is not captured in the present economic evaluation.
- A sensitivity analysis also was conducted to test the impact of the utility differential between being rendered hypothyroid or not. The utility difference between the pre-ablation health states was reduced by 50% by increasing the utility associated with hypothyroidism from 0.548 to 0.631. The model showed that change in utility difference in the pre-ablation health state by 50% generated in ICER of ₩33,693,724 per QALY. However, the advantage of more rapid recovery to full health and other cost off-sets of rhTSH should be considered under these extreme situation.
- The current economic evaluation relates to the ablation setting and therefore differs from the models evaluating the use of rhTSH in the diagnostic setting. The amount of radioiodine transmitted to the patient in the ablation setting (3.7 GBq) is approximately 20-fold higher than the amount typically provided in a diagnostic setting (150 MBq), and therefore faster clearance of radioiodine with rhTSH is of greater importance in this ablation setting. However, the two settings are similar in terms of quality of life benefits of rhTSH, regardless of whether the setting is diagnostic testing or remnant ablation.
- Recently, the national reimbursement criteria for using rhTSH was expanded as of October 2013 in Korea [14]. The revised reimbursement system allows patients who undergo total thyroidectomy to receive an one-time reimbursement for radioiodine ablation during their lifetime. Patients who have had serious adverse effects due to T4 withdrawal in previous radioiodine ablation are also eligible for reimbursement, per the previous criteria. After reimbursement, rhTSH may cost a patient as little as ₩58,695, compared to ₩1,219,000 before reimbursement. Therefore, the cost-effectiveness of rhTSH can be maximized under the newly expanded Korean insurance reimbursement criteria.
- In summary, the present analyses indicate that the therapeutic use of rhTSH is a cost-effective method of preparation for radioiodine ablation following thyroidectomy in Korea. This finding is consistent with analyses conducted in other international jurisdictions [1121]. In the Korean context, rhTSH offers substantial quality of life benefits over conventional endogenous stimulation at a reduced health-related cost after reimbursement. These advantages are magnified when important societal costs such as loss of productivity are included in the analysis. Furthermore, the base case assumptions were shown to be largely robust to extreme manipulation during sensitivity analysis.
- This economic evaluation does not capture any flow-on benefits associated with rhTSH use, such as more efficient use hospital resources, nor does the model incorporate any reduction in secondary cancers that could result from faster clearance of radioiodine. Regardless of these omissions, the use of rhTSH is a cost-effective alternative to endogenous hypothyroid stimulation prior to radioiodine ablation for patients who have undergone thyroidectomy in Korea. It is particularly cost-effective given the newly expanded Korean insurance reimbursement criteria.
DISCUSSION
-
CONFLICTS OF INTEREST: JHC is medical advisor in Genzyme Corporation. Genzyme Corporation funded the development of the economic model and the application of this model to the Korean setting.
Article information
- 1. Pacini F, Schlumberger M, Dralle H, Elisei R, Smit JW, Wiersinga W, et al. European consensus for the management of patients with differentiated thyroid carcinoma of the follicular epithelium. Eur J Endocrinol 2006;154:787–803. ArticlePubMed
- 2. American Thyroid Association (ATA) Guidelines Taskforce on Thyroid Nodules and Differentiated Thyroid Cancer. Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 2009;19:1167–1214. ArticlePubMed
- 3. Luster M, Felbinger R, Dietlein M, Reiners C. Thyroid hormone withdrawal in patients with differentiated thyroid carcinoma: a one hundred thirty-patient pilot survey on consequences of hypothyroidism and a pharmacoeconomic comparison to recombinant thyrotropin administration. Thyroid 2005;15:1147–1155. ArticlePubMed
- 4. Dow KH, Ferrell BR, Anello C. Quality-of-life changes in patients with thyroid cancer after withdrawal of thyroid hormone therapy. Thyroid 1997;7:613–619. ArticlePubMed
- 5. Rubic M, Kuna SK, Tesic V, Samardzic T, Despot M, Huic D. The most common factors influencing on quality of life of thyroid cancer patients after thyroid hormone withdrawal. Psychiatr Danub 2014;26(Suppl 3):520–527. PubMed
- 6. Lee J, Yun MJ, Nam KH, Chung WY, Soh EY, Park CS. Quality of life and effectiveness comparisons of thyroxine withdrawal, triiodothyronine withdrawal, and recombinant thyroid-stimulating hormone administration for low-dose radioiodine remnant ablation of differentiated thyroid carcinoma. Thyroid 2010;20:173–179. ArticlePubMed
- 7. Borget I, Corone C, Nocaudie M, Allyn M, Iacobelli S, Schlumberger M, et al. Sick leave for follow-up control in thyroid cancer patients: comparison between stimulation with Thyrogen and thyroid hormone withdrawal. Eur J Endocrinol 2007;156:531–538. ArticlePubMed
- 8. Schlumberger M, Catargi B, Borget I, Deandreis D, Zerdoud S, Bridji B, et al. Strategies of radioiodine ablation in patients with low-risk thyroid cancer. N Engl J Med 2012;366:1663–1673. ArticlePubMed
- 9. Mallick U, Harmer C, Yap B, Wadsley J, Clarke S, Moss L, et al. Ablation with low-dose radioiodine and thyrotropin alfa in thyroid cancer. N Engl J Med 2012;366:1674–1685. ArticlePubMed
- 10. Pacini F, Ladenson PW, Schlumberger M, Driedger A, Luster M, Kloos RT, et al. Radioiodine ablation of thyroid remnants after preparation with recombinant human thyrotropin in differentiated thyroid carcinoma: results of an international, randomized, controlled study. J Clin Endocrinol Metab 2006;91:926–932. ArticlePubMed
- 11. Mernagh P, Suebwongpat A, Silverberg J, Weston A. Cost-effectiveness of using recombinant human thyroid-stimulating hormone before radioiodine ablation for thyroid cancer: the Canadian perspective. Value Health 2010;13:180–187. ArticlePubMed
- 12. Schroeder PR, Haugen BR, Pacini F, Reiners C, Schlumberger M, Sherman SI, et al. A comparison of short-term changes in health-related quality of life in thyroid carcinoma patients undergoing diagnostic evaluation with recombinant human thyrotropin compared with thyroid hormone withdrawal. J Clin Endocrinol Metab 2006;91:878–884. ArticlePubMed
- 13. Borget I, Remy H, Chevalier J, Ricard M, Allyn M, Schlumberger M, et al. Length and cost of hospital stay of radioiodine ablation in thyroid cancer patients: comparison between preparation with thyroid hormone withdrawal and thyrogen. Eur J Nucl Med Mol Imaging 2008;35:1457–1463. ArticlePubMedPDF
- 14. Health Insurance Review & Assessment Service. Korea reimbursement system [Internet]; Seoul: HIRA; c2013. cited 2015 Oct 22. Available from: http://www.hira.or.kr/rf/medicine/getStandardList.do?pgmid=HIRAA030035040000.
- 15. Botella-Carretero JI, Galan JM, Caballero C, Sancho J, Escobar-Morreale HF. Quality of life and psychometric functionality in patients with differentiated thyroid carcinoma. Endocr Relat Cancer 2003;10:601–610. ArticlePubMed
- 16. Brazier J, Usherwood T, Harper R, Thomas K. Deriving a preference-based single index from the UK SF-36 Health Survey. J Clin Epidemiol 1998;51:1115–1128. ArticlePubMed
- 17. Sanofi. THYR-008-00: a randomized, controlled, open-label, multi-national pilot study of thyroid remnant ablation comparing the safety and ablation rate following 131I administration using ThyrogenTM versus the safety and ablation rate following 131I administration in the hypothyroid state: final report 2 [Internet]; Cambridge: Genzyme Corporation; c2015. cited 2015 Oct 22. Available from: http://www.genzymeclinicalresearch.com/home/search_clinical_trial_results/thyrogen_study5.asp.
- 18. Luster M, Sherman SI, Skarulis MC, Reynolds JR, Lassmann M, Hanscheid H, et al. Comparison of radioiodine biokinetics following the administration of recombinant human thyroid stimulating hormone and after thyroid hormone withdrawal in thyroid carcinoma. Eur J Nucl Med Mol Imaging 2003;30:1371–1377. ArticlePubMedPDF
- 19. Menzel C, Kranert WT, Dabert N, Diehl M, Fietz T, Hamscho N, et al. rhTSH stimulation before radioiodine therapy in thyroid cancer reduces the effective half-life of (131)I. J Nucl Med 2003;44:1065–1068. PubMed
- 20. Hanscheid H, Lassmann M, Luster M, Thomas SR, Pacini F, Ceccarelli C, et al. Iodine biokinetics and dosimetry in radioiodine therapy of thyroid cancer: procedures and results of a prospective international controlled study of ablation after rhTSH or hormone withdrawal. J Nucl Med 2006;47:648–654. PubMed
- 21. Mernagh P, Campbell S, Dietlein M, Luster M, Mazzaferri E, Weston AR. Cost-effectiveness of using recombinant human TSH prior to radioiodine ablation for thyroid cancer, compared with treating patients in a hypothyroid state: the German perspective. Eur J Endocrinol 2006;155:405–414. ArticlePubMed
References
Appendix
Treatment Practice Survey
Simplified schematic of the structure of the Korean adaptation of economic model. rhTSH, recombinant human thyroid stimulating hormone.
Quality-adjusted life year (QALY) weights as used in the economic model. rhTSH, recombinant human thyroid stimulating hormone.
Duration of Health States Applied to the Economic Model
| Health state | Exogenously-stimulated (rhTSH) | Endogenously-stimulated (hypothyroid) |
|---|---|---|
| Pre-ablation | 1 week | Between 2 and 7 weeks (refer to Table 2) |
| Ablation | 1 week | 1 week |
| Initial post-ablation period | 4 weeks | 4 weeks |
| Second post-ablation period | 4 weeks | 4 weeks |
| Patient recovered (well state) | Remainder of the model (7 weeks per patient) | Remainder of the model (average 3.6 weeks per patient)a |
rhTSH, recombinant human thyroid stimulating hormone.
aAlthough the average duration of this health state was 3.6 weeks, the model was still run for 17 weeks. This ensures consistency with Mernagh et al. [11] and similarly ensures that all patients reach the 'well' health state, and remain there for at least one full cycle (1 week).
Distribution of Time Spent between Thyroidectomy and Ablation in the Endogenously-Stimulated Patient Group
Duration of Time Spent It the Radioprotective Ward for Endogenously-Stimulated (Recombinant Human Thyroid Stimulating Hormone) Patients
| Duration | Proportion of patients, % | Expected duration, days |
|---|---|---|
| Less than 1 day | 0 | - |
| 2 days | 5.70 | - |
| 3 days | 91.30 | - |
| 4 days | 3.00 | - |
| 5 days | 0 | - |
| Average expected duration | - | 2.97 |
Unit Costs Incorporated into the Economic Evaluation
Costs Applied to Each Treatment Arm
Base Case Analysis
Sensitivity Analysis
Figure & Data
References
Citations

- Comparison of Recombinant Human Thyroid-Stimulating Hormone and Thyroid Hormone Withdrawal for 131I Therapy in Patients With Intermediate- to High-Risk Thyroid Cancer
Sohyun Park, Ji-In Bang, Keunyoung Kim, Youngduk Seo, Ari Chong, Chae Moon Hong, Dong-Eun Lee, Miyoung Choi, Sang-Woo Lee, So Won Oh
Clinical Nuclear Medicine.2024; 49(3): e96. CrossRef - Thyroid Hormone Withdrawal versus Recombinant Human TSH as Preparation for I-131 Therapy in Patients with Metastatic Thyroid Cancer: A Systematic Review and Meta-Analysis
Luca Giovanella, Maria Luisa Garo, Alfredo Campenní, Petra Petranović Ovčariček, Rainer Görges
Cancers.2023; 15(9): 2510. CrossRef - Health-related quality of life of thyroid cancer patients undergoing radioiodine therapy: a cohort real-world study in a reference public cancer hospital in Brazil
Jayda Eiras Ramim, Marcella Araugio Soares Cardoso, Gessen Lopes Carneiro de Oliveira, Maria Luisa Gomes, Tiago Teixeira Guimarães, Rossana Corbo Ramalho de Mello, Anke Bergmann, Priscilla Brunelli Pujatti
Supportive Care in Cancer.2020; 28(8): 3771. CrossRef - Predictive factors determining incomplete response to radioiodine therapy in patients with differentiated thyroid cancer
Ewelina Szczepanek-Parulska, Magdalena Wojewoda-Korbelak, Martyna Borowczyk, Malgorzata Kaluzna, Barbara Brominska, Katarzyna Ziemnicka, Rafal Czepczynski, Maciej Baczyk, Marek Ruchala
The Quarterly Journal of Nuclear Medicine and Molecular Imaging.2020;[Epub] CrossRef - Initial Adoption of Recombinant Human Thyroid-Stimulating Hormone Following Thyroidectomy in the Medicare Thyroid Cancer Patient Population
Michaela A. Dinan, Yanhong Li, Shelby D. Reed, Julie Ann Sosa
Endocrine Practice.2019; 25(1): 31. CrossRef - Triennial Report ofEndocrinology and Metabolism, 2015 to 2017
Eun-Jung Rhee, Hey Yeon Jang, Won-Young Lee
Endocrinology and Metabolism.2018; 33(2): 195. CrossRef - Recombinant human TSH stimulated thyroglobulin levels at remnant ablation predict structural incomplete response to treatment in patients with differentiated thyroid cancer
Jeonghoon Ha, Min Hee Kim, Kwanhoon Jo, Yejee Lim, Ja Seong Bae, Sohee Lee, Moo Il Kang, Bong Yun Cha, Dong Jun Lim
Medicine.2017; 96(29): e7512. CrossRef - Does the Risk of Metabolic Syndrome Increase in Thyroid Cancer Survivors?
Min-Hee Kim, Jin-young Huh, Dong-jun Lim, Moo-Il Kang
Thyroid.2017; 27(7): 936. CrossRef

 KES
KES
 PubReader
PubReader Cite
Cite