Articles
- Page Path
- HOME > Endocrinol Metab > Volume 36(2); 2021 > Article
-
Original ArticleClinical Study High Serum-Induced AhRL Is Associated with Prevalent Metabolic Syndrome and Future Impairment of Glucose Tolerance in the Elderly
-
Youngmi Kim Pak1,2
 , Hoon Sung Choi3, Wook Ha Park1, Suyeol Im2, P. Monica Lind4, Lars Lind5, Hong Kyu Lee6
, Hoon Sung Choi3, Wook Ha Park1, Suyeol Im2, P. Monica Lind4, Lars Lind5, Hong Kyu Lee6
-
Endocrinology and Metabolism 2021;36(2):436-446.
DOI: https://doi.org/10.3803/EnM.2020.883
Published online: April 19, 2021
1Department of Physiology, Kyung Hee University School of Medicine, Seoul, Korea
2Department of Neuroscience, Medical Research Center for Bioreaction to Reactive Oxygen Species and Biomedical Science Institute, Kyung Hee University School of Medicine, Graduate School, Seoul, Korea
3Department of Internal Medicine, Kangwon National University School of Medicine, Chuncheon, Korea
4Occupational and Environmental Medicine, Uppsala University, Uppsala, Sweden
5Acute and Internal Medicine, Department of Medicine, Uppsala University Hospital, Uppsala, Sweden
6Department of Internal Medicine, Seoul National University College of Medicine, Seoul, Korea
- Corresponding author: Hong Kyu Lee. Department of Internal Medicine, Seoul National University College of Medicine, 101 Daehak-ro, Jongno-gu, Seoul 03080, Korea, Tel: +82-2-379-1694, E-mail: hongkyu414@gmail.com
Copyright © 2021 Korean Endocrine Society
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (https://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
ABSTRACT
-
Background
- High circulating levels of dioxins and dioxin-like chemicals, acting via the aryl hydrocarbon receptor (AhR), have previously been linked to diabetes. We now investigated whether the serum AhR ligands (AhRL) were higher in subjects with metabolic syndrome (MetS) and in subjects who had developed a worsened glucose tolerance over time.
-
Methods
- Serum AhRL at baseline was measured by a cell-based AhRL activity assay in 70-year-old subjects (n=911) in the Prospective Investigation of the Vasculature in Uppsala Seniors (PIVUS) study. The main outcome measures were prevalent MetS and worsening of glucose tolerance over 5 years of follow-up.
-
Results
- AhRL was significantly elevated in subjects with prevalent MetS as compared to those without MetS, following adjustment for sex, smoking, exercise habits, alcohol intake and educational level (P=0.009). AhRL at baseline was higher in subjects who developed impaired fasting glucose or diabetes at age 75 years than in those who remained normoglycemic (P=0.0081). The odds ratio (OR) of AhRL for worsening glucose tolerance over 5 years was 1.43 (95% confidence interval [CI], 1.13 to 1.81; P=0.003, continuous variables) and 2.81 (95% CI, 1.31 to 6.02; P=0.008, in the highest quartile) adjusted for sex, life style factors, body mass index, and glucose.
-
Conclusion
- These findings support a large body of epidemiologic evidence that exposure to AhR transactivating substances, such as dioxins and dioxin-like chemicals, might be involved in the pathogenesis of MetS and diabetes development. Measurement of serum AhRL in humans can be a useful tool in predicting the onset of metabolic disorders.
- The clustering of risk factors for cardiovascular diseases, such as abdominal obesity, hyperglycemia, high blood pressure, and hyperlipidemia in an individual has been known for a long time [1,2]. Reaven [3] proposed to study this state as an unknown disease syndrome X, which was renamed to metabolic syndrome (MetS) [4]. Many epidemiologic studies have revealed a high serum concentration of various environmental endocrine disrupting chemicals (EDCs) [5,6] is strongly associated with MetS [7–9] or its component phenotypes, such as abdominal obesity [10] and diabetes mellitus (DM) [11–13].
- Among the EDCs, persistent organic pollutants (POPs) are the most important. They are banned by an international treaty, the Stockholm Convention, as they persist for many years in the environment, bioaccumulate in human fat tissues, reach high level with time, and have been suggested to cause risks to human health [14]. Some organochlorine POPs, such as trans-nonachlor, dichlorodiphenyldichloroethylene, polychlorinated biphenyls (PCBs), and dioxins and dioxin-like chemicals show a strong relationship to diabetes [15,16], whereas other non-organochlorine POPs, such as perfluoroalkyl acids and brominated compounds are weakly associated with diabetes. In addition, dioxin exposures altered glucose and lipid metabolisms, possibly leading to MetS, in both epidemiological and animal studies [17].
- However, there is a large data gap in the linkage between POP exposure and the onset of diabetes. To fill this data gap, the U.S. National Toxicological Program recommended developing improved methods to measure POPs in small blood volumes using high throughput technologies at a reasonable cost, which could be used in epidemiologic studies [15]. The Endocrine Society made similar recommendations in its executive summary, including the need for longitudinal and multigenerational analyses of EDCs in humans and implementing emerging and sensitive testing systems [5,18].
- The Prospective Investigation of the Vasculature in Uppsala Seniors (PIVUS) study was started in 2001, aiming primarily to investigate the endothelial function and arterial compliance in a random sample of 1,000 70-year-old subjects, living in the community of Uppsala, Sweden [19]. The levels of various environmental pollutants were measured with high-resolution chromatography, coupled with a high-resolution mass spectrometry system. This found several POPs to be associated with MetS [8] and type 2 diabetes [10,13].
- We developed a cell-based reporter assay that monitors the cumulative biological activity of an aryl hydrocarbon receptor (AhR) ligand mixture in a tiny amount of serum without a lipid extraction process [20,21]. Some POPs, such as dioxins and dioxin-like PCBs, exhibit their biological effects by activating AhR [22,23], a ligand-activated nuclear receptor that has been characterized as a “dioxin receptor” or “xenobiotic receptor,” sensing xenotoxicants [24,25]. Therefore, serum-induced AhR ligands (AhRL) bioactivity could be the level of AhR-dependent biological activity induced by a circulating POPs mixture. The serum AhRL was elevated in subjects with an abnormal glucose tolerance and associated with several components of MetS among Koreans [20]. It was confirmed that there was a strong dose-dependent relationship between serum AhRL and total toxic equivalence (TEQtotal) values (adjusted for sex, serum cholesterol, and serum triglycerides), calculated from total dioxin-like PCBs and dioxins in the PIVUS participants [21]. Therefore, serum AhRL bioactivity could represent a contamination level of POPs in humans and be a good serum biomarker for MetS.
- We also developed a similar cell-based assay for serum mitochondrial inhibiting substances (MIS), which were measured by intracellular ATP contents of cultured cells (MIS-ATP) treated with serum. With these assays we found there were dose-dependent relationships between AhRL and a TEQtotal values (adjusted for sex, serum cholesterol and serum triglycerides) calculated from total dioxin-like PCBs and dioxins in the PIVUS participants [21]. Also, MIS-ATP was related to TEQ. Recently, Lee et al. [26] had reported serum AhRL and MIS-ATP could predict the future development of impaired glucose tolerance (IGT) and diabetes in a large, well-characterized, community-based prospective epidemiologic study, the Korean Genome and Epidemiology Study (KoGES).
- In the present study, we investigated the relation between serum AhRL and prevalent MetS as well as worsening of glucose tolerance during 5 years of follow-up in the PIVUS cohort. In addition, MIS-ATP was also determined, which was reversely correlated with AhRL and was reduced in subjects with glucose intolerance and MetS in a Korean population [20]. Here, we report serum AhRL is elevated in the Uppsala subjects with MetS, as well as in those who develop glucose intolerance following a 5-year follow-up.
INTRODUCTION
- Subjects
- The research design for the first part of this study is a cross-sectional investigation within the cohort study on the relationship between serum AhRL and prevalent MetS. The second part is a longitudinal investigation of the association between serum AhRL and the development of glucose intolerance over a 5-year follow-up.
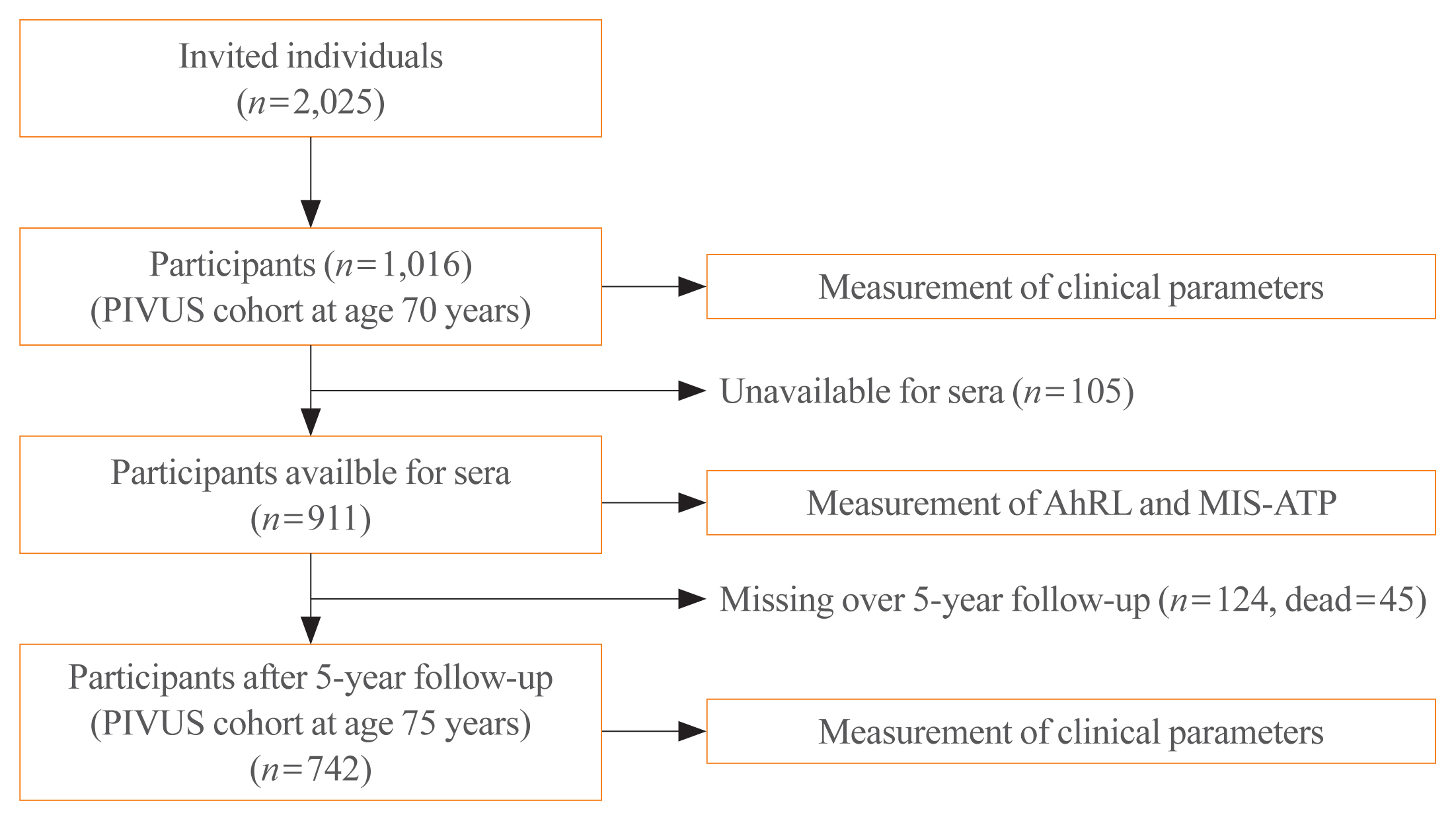
- The details of the PIVUS study (https://www.medsci.uu.se/pivus/) have previously been reported [19,27]. In short, eligible subjects were all aged 70±0.08 years and lived in the community of Uppsala, Sweden, who had been randomly chosen from a register of community living (n=2,025). A total of 1,016 subjects participated (female, 50.2%), giving a participation rate of 50.1%. When the subjects became 75 years of age, a reinvestigation of the cohort was done with a follow-up rate of 81.4% (n=826). A total of 52 individuals had died during the follow-up period. The study was approved by the Ethics Committee of the University of Uppsala, and the participants gave written informed consent. Measurements of AhRL and MIS-ATP were performed in 911 of the subjects whose serum samples are available, and this constitutes the baseline sample of this study. Among the 911 participants, 742 subjects enrolled at age 75 during a 5-year follow-up. Fig. 1 summarizes a flow chart for the selection of this study population.
- Basic investigation
- All subjects were investigated in the morning after an overnight fast. No medication or smoking was allowed after midnight. Participants were asked to answer a questionnaire about their medical history, smoking habits, and regular medication. Various clinical parameters including weight, height, blood pressure, heart rate, and waist circumference were measured, and batches of blood samples were obtained for various biochemical tests. Some serum samples were stored at −80°C for later studies, which were used in this study. Basic characteristics are given in Table 1. Approximately 10% of the cohort reported a history of coronary heart disease, 4% reported stroke, and 9% reported DM. Nearly half (45%) of the cohort reported the use of at least one medication for a cardiovascular condition, with antihypertensive medication being the most prevalent (32%). Fifteen percent reported the use of statins, and insulin and oral antihyperglycemic drug use was reported by 2% and 6% of the participants, respectively [19].
- As the participation rate in this cohort was only 50%, we conducted an evaluation of cardiovascular disorders and medications in 100 consecutive non-participants. The prevalence of cardiovascular drug intake, history of myocardial infarction, coronary revascularization, antihypertensive medication, and statin use and insulin treatment were similar to those in the investigated sample, whereas the prevalence of diabetes, congestive heart failure and stroke tended to be higher among the non-participants [8].
- Baseline measurement
- Baseline characteristics of participants were measured as described previously [10]. A questionnaire was given to the participants to answer about their medical history and regular medication, educational level, exercise habits, and smoking habits. The educational levels were divided into three groups: <9, 9–12, and >12 years of education. Exercise habits were divided into four groups: <2 times light exercise (no sweat) per week; ≥2 times light exercise per week; 1–2 times heavy exercise (sweat) per week; and >2 times heavy exercise (sweat) per week. The dietary intakes of total calories and alcohol were assessed by use of a 7-day food diary. Computer software was used to calculate the daily amount of calorie and alcohol intake from the recorded food and drink items.
- All subjects were investigated in the morning following an overnight fast, with no medication or smoking allowed after midnight. Waist circumference was measured in the supine position midway between the lowest rib and the iliac crest. Plasma cholesterol and triglyceride concentrations were determined in an enzymatic assay, and fasting blood glucose was analyzed with the hexokinase method using ARCHITECT (Abbott, Abbott Park, IL, USA).
- CALA assay for AhRL bioactivity
- To measure AhRL bioactivity in the serum samples, we performed cell-based AhR ligand activity (CALA) assay [20] with minor modifications, as described previously [21,28]. Briefly, the assay was performed using puromycin (1 μg/mL)-resistant stable cells of Hepa1c1c7 mouse hepatoma cells (CRL-2026), which co-expresses pRL-mTK and pGL4-DRE-luc (puromycin+) reporter plasmid containing four dioxin-responsive element consensus sequences. The transfected stable cells seeded at 5×104/well in a 96-well plate were cultured for 24 hours in α-minimum essential medium (α-MEM) supplemented with 10% fetal bovine serum and 1% penicillin and streptomycin. After the media had been changed to 90 μL of phenol red-free Dulbecco’s Modified Eagle’s Medium, the cells were treated for 24 hours with 10 μL of heat-inactivated human serum samples or charcoal stripped human serum (control, CS-HS). Luciferase activities were measured using a Dual-Glo Luciferase assay system (Promega, Madison, WI, USA) and a luminometer (Berthold, Bad Wildbad, Germany) and subsequently normalized against Renilla luciferase activity to normalize cell numbers. The AhRL bioactivities of serum-treated cells were presented as fold induction (FI) over the AhRL bioactivity of the 10% CS-HS-treated control cells. All assays were conducted in duplicate. The intra- and interassay coefficients of variation of these methods were less than 4.0%.
- MIS-ATP assay for mitochondrial inhibition induced by serum samples
- The mitochondrial inhibition induced by human serum samples was evaluated by determining intracellular ATP content [20]. In short, the Hepa1c1c7 cells (5×104/well) showing stable Renilla luciferase activity, were treated with 10% human serum samples or 10% CS-HS for 48 hours in a 96-well plate. The intracellular ATP content of the treated cells was determined using the luciferin-luciferase reaction and CellTiter-Glo luciferase kits (Promega) [29]. The measured ATP contents were normalized to Renilla luciferase activity, determined by adding an equal amount of Stop & Glo substrate solution of Dual-Glo Luciferase assay system (Promega). All data is presented as a percentage of control of ATP contents in 10% CS-HS-treated control cells. The intracellular ATP contents of the control cells were 65.1±2.7 nM. The ATP concentration of 10% sample serum-treated cells can be calculated from the standard curve of ATP concentration (nM)=(% Control+18.24)/1.817. The intra- and interassay coefficients of variation of these methods were less than 6.0%.
- Biochemical measurements
- Blood pressure was measured by a calibrated mercury sphygmomanometer in the non-cannulated arm to the nearest mmHg after at least 30 minutes of rest. We used an average of three recordings. Traditional lipid variables and fasting blood glucose were measured by standard laboratory techniques. Basic risk factor characteristics, medical history, and regular medications were reported [19].
- Criteria for MetS and classification of glucose intolerance
- MetS was defined by the National Cholesterol Education Program Expert Panel and Adult Treatment Panel III (NCEP/ATP III) criteria [30]. Three of the following five criteria needed to be fulfilled: Blood pressure >130/85 mm Hg or antihypertensive treatment, fasting plasma glucose >6.2 mmol/L or antidiabetic treatment, serum triglycerides >1.7 mmol/L, waist circumference >102 cm in men and >88 cm in women, high density lipoprotein (HDL)-cholesterol <1.0 mmol/L in men and <1.3 in women.
- We divided the glucose tolerance spectra in three categories; normal (fasting plasma glucose <5.6 mmol/L), impaired fasting glucose (IFG; fasting plasma glucose 5.6 to 6.9 mmol/L), DM (fasting plasma glucose ≥7.0 mmol/L and/or antidiabetic treatment).
- Statistical analysis
- In the cross-sectional part of the study, the mean±standard deviation (SD) for AhRL and MIS-ATP content were given for subjects with and without MetS. Additionally, quartile analysis of AhRL and MIS-ATP was performed and the lowest quartile group (Q1) was considered as the reference group in linear and logistic regression. To evaluate the risk of AhRL and MIS-ATP for the presence of MetS, multivariate logistic regression for quartile categories was performed with adjustment for sex-only (Model 1), and sex plus the life style factors smoking, exercise habits, energy and alcohol intake, educational level, body mass index (BMI), and fasting glucose (Model 2). AhRL and MIS-ATP variables were transformed to the SD-scale. The linear regression analysis was performed for continuous variables or quartile groups. Comparison of mean number of MetS components was performed by linear regression adjusted for same eight covariates. The P value for the difference between the two groups was calculated using analysis of covariance (ANCOVA) adjusted for the same eight covariates as above. Since we evaluated two exposures, the P value for significance was set to 0.025 (=0.05/2). As a secondary supportive analysis, we used similar statistics to correlate AhRL and MIS-ATP content with the presence of the five MetS criteria without performing formal adjustments for multiple comparisons. A linear regression analysis was performed to correlate AhRL (and MIS-ATP) with the number of components of MetS seen in each subject. We also used the quartiles of AhRL and MIS-ATP to investigate the relationship between AhRL and MIS-ATP and number of MetS components.
- In longitudinal analysis, two analyses were performed: logistic regression and analysis of variance (ANOVA) analyses. Multiple logistic regression for continuous variables or quartile categories was performed to obtain the odds ratio (OR) and 95% confidence intervals (CIs) of AhRL (or MIS-ATP) for worsening glucose tolerance over 5 years. AhRL and MIS-ATP were also transformed to the SD-scale. A worsening of the glucose tolerance was defined as either moving from the normal group to the IFG or DM groups, or from the IFG group to the DM group. To adjust covariates, Model 1 included only sex as a covariate. Model 2 additionally included smoking status, exercise habits, energy and alcohol intake, education level, BMI and fasting glucose. Using ANOVA, we compared subjects who had remained normoglycemic at age 75 years with those who had developed either IFG or DM at age 75 years, within the group that was normoglycemic at baseline (age 70 years). We used STATA version 14 (StataCorp LP, College Station, TX, USA) or R packages for statistical calculations (R Foundation for Statistical Computing, Vienna, Austria).
METHODS
- Among 911 subjects in the cross-sectional analyses, diabetes and IFG occurred in 10.0% and 10.9%, respectively (Table 1). Subject who were examined at age 75 years and had measurements of AhRL at age 70 years, thereby being eligible for the prospective analyses (n=742), had baseline characteristics similar to those in the cross-sectional analysis. Supplemental Table S1 summarizes the number of subjects according to glucose tolerance in the AhRL and MIS-ATP quartile groups aged 70 and 75 years.
- Cross-sectional analyses regarding prevalent MetS
- AhRL was significantly elevated in subjects with MetS (n=214) as compared to those without MetS (n=697), following adjustment for sex, smoking, exercise habits, alcohol intake, and educational level (2.21±0.24 vs. 2.16±0.24, P=0.009). However, MIS-ATP was not different between groups with or without MetS (80.2±8.18 vs. 80.6±8.4, P=0.775). Compared to subjects with lower quartile of AhRL, those in higher quartile of AhRL showed more frequent prevalence of MetS with statistical significance, and there was significant risk effect in the Q3 and Q4 quartiles (Table 2). The ORs of AhRL for MetS in the Q3 and Q4 quartiles were 1.76 (95% CI, 1.03 to 3.01; P=0.039) and 2.23 (95% CI, 1.33 to 3.74; P=0.003), respectively. Again, MIS-ATP did not show statistical significances in these quartile analyses for MetS.
- AhRL, but not MIS-ATP, was significantly related to the number of components of MetS present following adjustment for sex, smoking, exercise habits, alcohol intake, and educational level (P=0.020) (Table 3) as calculated using AhRL or MIS-ATP as continuous variables being transformed to the SD-scale. When AhRL was divided into quartiles (Table 3), the relationship with the number of MetS components was limited to the highest quartile Q4 in both Model 1 and Model 2. The subjects in Q4 of AhRL tends to have more components of MetS with statistical significance. MIS-ATP did not show significant correlation with the number of components of MetS.
- Of the five components of MetS, the presence of lower HDL-criteria only had significant difference of AhRL in multivariate analyses with adjustment for sex, smoking, exercise habits, alcohol intake, and educational level (P=0.019) (Table 4). The proportions of the HDL-criteria-present in Q1 and Q4 of AhRL were 15% and 22%, respectively. However, MIS-ATP was not different between groups with or without any of the five MetS components.
- Prospective analyses regarding worsening of glucose tolerance over 5 years
- Seventy-seven subjects showed impairment in glucose tolerance over a 5-year follow-up. AhRL levels in subject who had worsened their glucose tolerance from age 70 to 75 years (normal to IFG/DM or IFG to DM, n=77) were higher than those who had not (2.25±0.26 FI vs. 2.17±0.24 FI, P=0.010). AhRL showed a significant risk effect on worsening of glucose tolerance in multivariate analyses (Table 5). The ORs of AhRL on worsening of glucose tolerance were 1.31 (95% CI, 1.03 to 1.67; P= 0.028) adjusted for sex (Model 1) and 1.41 (95% CI, 1.10 to 1.82; P=0.007) adjusted for sex, smoking, exercise habits, energy and alcohol intake, education level, BMI, and glucose (Model 2). However, MIS-ATP did not show significant risk. There was no interaction term between AhRL and MIS-ATP for worsening of glucose tolerance (P=0.453). In quartile analysis, the only highest quartile (Q4) of AhRL showed the increased risk of a worsening of glucose tolerance in both Model 1 (OR, 2.02; 95% CI, 1.03 to 3.95; P=0.040) and Model 2 (OR, 2.81; 95% CI, 1.31 to 6.02; P=0.008).
- In a separate analysis restricted to only those with a normal glucose tolerance at baseline, AhRL at baseline was higher in the subjects who had developed IFG or DM at age 75 years (n=60) than in those subjects who had remained normal at age 75 (2.26±0.27 vs. 2.17±0.24, P=0.0081).
RESULTS
- This study showed that serum AhRL measured by a cell-based CALA assay is elevated in the sera of subjects with prevalent MetS and is significantly related to the number of components of MetS present in the cross-sectional part of the study. Furthermore, serum AhR bioactivity was related to a deterioration in the glucose tolerance in this cohort during a 5-year follow-up.
- In previous studies on this cohort, the PIVUS investigators had observed relationships between concentrations of various POPs and different components of MetS, such as hypertension, diabetes, and abdominal obesity [11,13]. An environment-wide association study on MetS [8] also revealed the associations between certain PCBs and MetS. However, the analysis of POPs in this cohort sample found no association between MetS and TEQ values being calculated from the chemical measurements. This difference could arise simply from the differences between the nature of the CALA assay and calculated TEQ. The former measures the total AhRL-dependent transactivation in serum, whereas the latter depends upon the number of dioxin-like compounds measured and the quality of the analysis.
- AhR mediates most toxic/biological effects of dioxin-like compounds [24,31–33]. From an analysis of sera collected from this PIVUS cohort, Park et al. [20] observed that AhRL showed a significant correlation to the serum concentrations of several chemically measured POPs and the derived TEQ value. This suggests that it could be a biomarker of POPs mixture in the sera. However, those results should be interpreted carefully, since not all dioxin-like compounds activating AhR were chemically determined in the PIVUS study, and those unmeasured AhRL could have contributed to the total AhR-dependent transactivation. Furthermore, some AhRL might have acted as antagonists, instead of agonists [34].
- Another major observation made in this study was that serum AhRL was higher in those subjects who had normal fasting blood glucose at baseline and then developed IGT after 5 years (at age 75 years) than in those subjects who remain normal at follow-up. This was also observed when AhRL was related to a worsening in glucose tolerance over a 5-year-period. This novel finding strongly suggests that the exposure to environmental toxicants being AhRL may be an independent risk factor for the development of glucose intolerance in this population. Although experimental evidence also supports the concept that exposure to EDCs, such as the herbicide atrazine [35] or contaminated fish oil containing POPs, can cause insulin resistance in animals [36], a hallmark of MetS and glucose intolerance, there are still large data gaps in establishing a causal relationship in humans.
- Although a previous study done in Korea showed both serum AhRL and MIS-ATP could predict the future development of IFG and diabetes [26], we could not find any association between MIS-ATP and the prevalence of MetS or its components, nor the future development of glucose intolerance in this study. An immense variety of chemical mixtures, which differ between populations and over time, and differences between the ages of the populations studied, could have contributed to these discrepancies. The PIVUS cohort consisted of 70-year-old people living in a city, whereas the KoGES cohort was mostly middle age farmers between 40 to 60 years of age.
- It might look paradoxical that AhRL was not significantly related to the glucose criteria of MetS, while AhRL was significantly related to future impairment in glucose control. There are two most probable explanation for this. First, although the correlation between AhRL and the glucose criteria of MetS is not significant, the direction and magnitude of the relationship is the same as for the prospective outcome. Second, the relationship between AhRL and the glucose criteria of MetS is based on cross-sectional data that includes factors related to poor glucose tolerance, such as advice on diet and exercise habits, and antidiabetic medications may affect AhRL levels. This is a common major drawback of cross-sectional analysis, which is unlikely to influence the prospective analysis as in that case all participants at risk are free from the disease at baseline.
- The strengths of this study include the use of serum samples from a well-established population-based epidemiologic study, the PIVUS study, and a double-blind approach to data analysis, eliminating any observer bias. Analyses of serum with cell-based assays were done at Pak’s laboratory in South Korea, and statistical analysis was done by the Swedish group. Major limitations of the study stem from the nature of bioassays detecting dioxin-like substances [37], because AhR-mediated responses to dibenzo-p-dioxins and -furans differ according to the cell lines [32]. Our CALA assay is standardized to 2,3,7,8-tetrachlorodibenzodioxin (TCDD), but not to different types of AhRL in Hepa1c1c7 cells. However, the use of in vitro assays as a screening tool to characterize various environmental chemical mixtures is becoming increasingly popular in the field of environmental toxicology [25,37]. Our cell-based assays for AhRL and MIS-ATP are easy and inexpensive, require tiny amounts of serum and no sophisticated instruments. This provides mechanistic insight into the metabolic abnormalities. In conclusion, this study provides evidence that serum AhRL are associated with MetS and glucose intolerance and that the CALA assay can be a valuable tool to evaluate the exposure degree of POPs, especially when appropriate cell lines are employed [34].
DISCUSSION
Supplementary Information
-
Acknowledgements
- This research was supported by the National Research Foundation of Korea (NRF) grants (2020R1A2C1008699 and 2018R1A6A1A03025124) funded by the Korean government (MSIT, to Youngmi Kim Pak) and by Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (FORMAS, to Lars Lind).
-
CONFLICTS OF INTEREST
No potential conflict of interest relevant to this article was reported.
-
AUTHOR CONTRIBUTIONS
Conception or design: Y.K.P., H.S.C., H.K.L. Acquisition, analysis, or interpretation of data: Y.K.P., W.H.P., S.I., L.L. Drafting the work or revising: M.L. Final approval of the manuscript: M.L., H.K.L.
Article information
Values are expressed as mean±standard deviation. The sample serum-induced AhRL and MIS-ATP are presented as fold induction and % control (%) over those of the 10% CS-HS-treated control cells, respectively.
PIVUS, Prospective Investigation of the Vasculature in Uppsala Seniors; BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; LDL-C, low density lipoprotein cholesterol; HDL-C, high density lipoprotein cholesterol; AhRL, arylhydrocarbon receptor ligand activity; CS-HS, charcoal stripped human serum; MIS-ATP, mitochondrial inhibiting substance activity measured by intracellular ATP content.
| Quartile | AhRL | MIS-ATP | ||||
|---|---|---|---|---|---|---|
|
|
|
|||||
| Prevalence, % | OR (95% CI) | P value | Prevalence, % | OR (95% CI) | P value | |
| Q1 | 18.1 | Reference | - | 25.1 | Reference | - |
|
|
||||||
| Q2 | 22.8 | 1.48 (0.86–2.55) | 0.160 | 23.7 | 0.98 (0.59–1.60) | 0.921 |
|
|
||||||
| Q3 | 24.1 | 1.76 (1.03–3.01) | 0.158a | 25.0 | 1.15 (0.70–1.89) | 0.583 |
|
|
||||||
| Q4 | 28.9 | 2.23 (1.33–3.74) | 0.003a | 20.2 | 0.71 (0.42–1.20) | 0.203 |
P values were calculated using multivariate logistic regression for quartile categories adjusted for sex, smoking, exercise habits, energy and alcohol intake, education level, body mass index, and fasting glucose.
AhRL, arylhydrocarbon receptor ligand activity; MIS-ATP, mitochondrial inhibiting substance activity measured by intracellular ATP content; OR, odds ratio; CI, confidence interval.
a P values <0.05 were considered significant.
| Variable | AhRLa | MIS-ATPa | ||||
|---|---|---|---|---|---|---|
|
|
|
|||||
| Beta | SE | P value | Beta | SE | P value | |
| Continuous variable | ||||||
| Model 1 | 0.053 | 0.037 | 0.025b | 0.025 | 0.038 | 0.502 |
| Model 2 | 0.092 | 0.039 | 0.020b | 0.022 | 0.040 | 0.594 |
|
|
||||||
| Quartilec | ||||||
| Model 1 | ||||||
| Q1 | Reference | - | - | Reference | - | - |
| Q2 | 0.024 | 0.105 | 0.819 | 0.014 | 0.105 | 0.897 |
| Q3 | −0.046 | 0.105 | 0.662 | 0.076 | 0.105 | 0.471 |
| Q4 | 0.243 | 0.105 | 0.021b | −0.003 | 0.105 | 0.972 |
| Model 2 | ||||||
| Q1 | Reference | - | - | Reference | - | - |
| Q2 | −0.012 | 0.116 | 0.914 | 0.024 | 0.116 | 0.835 |
| Q3 | 0.008 | 0.117 | 0.943 | 0.094 | 0.116 | 0.417 |
| Q4 | 0.241 | 0.114 | 0.035b | −0.027 | 0.117 | 0.812 |
P values were calculated using linear regression for continuous variables and quartile categories. Model 1, adjusted for sex; Model 2, adjusted for Model 1+smoking, exercise habits, energy and alcohol intake, education level, body mass index, and fasting glucose.
AhRL, arylhydrocarbon receptor ligand activity; MIS-ATP, mitochondrial inhibiting substance activity measured by intracellular ATP content; Beta, regression coefficient; SE, standard error.
a All variables were transformed to the SD-scale;
b P values <0.05 were considered significant;
c The lowest quartile Q1 was used as the reference group.
| Component | AhRL (FI) | MIS-ATP (%) | ||
|---|---|---|---|---|
|
|
|
|||
| Mean±SD | P value | Mean±SD | P value | |
| High glucose | ||||
| Present (n=193) | 2.20±0.25 | 0.191 | 80.13±8.36 | 0.265 |
| Absent (n=718) | 2.17±0.24 | 80.56±8.30 | ||
|
|
||||
| Hypertension | ||||
| Present (n=759) | 2.18±0.25 | 0.282 | 80.46±8.38 | 0.788 |
| Absent (n=152) | 2.16±0.23 | 80.51±7.97 | ||
|
|
||||
| High triglyceride | ||||
| Present (n=162) | 2.20±0.26 | 0.219 | 80.51±8.63 | 0.844 |
| Absent (n=749) | 2.17±0.24 | 80.46±8.24 | ||
|
|
||||
| Low HDL | ||||
| Present (n=163) | 2.21±0.25 | 0.019a | 80.98±7.51 | 0.238 |
| Absent (n=748) | 2.17±0.24 | 80.36±8.47 | ||
|
|
||||
| Large waist circumference | ||||
| Present (n=311) | 2.18±0.25 | 0.479 | 80.48±8.17 | 0.344 |
| Absent (n=600) | 2.17±0.24 | 80.46±8.39 | ||
The presence of each component of MetS was defined by following criteria; high glucose, glucose >6.2 mmol/L or antidiabetic treatment; hypertension, blood pressure >130/85 mm Hg or antihypertensive treatment; high triglyceride, triglycerides >1.7 mmol/L; low HDL, HDL <1.0 mmol/L in men and <1.3 mmol/L in women; large waist circumference, waist circumference >102 cm in men and >88 cm in women. Serum AhRL and MIS-ATP are presented as FI and % of charcoal stripped human serum-treated control. P values were calculated using analysis of covariance (ANCOVA) adjusted for sex, smoking, exercise habits, energy and alcohol intake, education level, body mass index, and fasting glucose.
AhRL, arylhydrocarbon receptor ligand activity; MIS-ATP, mitochondrial inhibiting substance activity measured by intracellular ATP content; MetS, metabolic syndrome; FI, fold induction; SD, standard deviation; HDL, high density lipoprotein.
a P values <0.05 were considered significant.
| AhRLa | MIS-ATPa | |||||
|---|---|---|---|---|---|---|
|
|
|
|||||
| No./totalb | OR (95% CI) | P value | No./totalb | OR (95% CI) | P value | |
| Continuous variable | ||||||
| Model 1 | 1.30 (1.03–1.62) | 0.022c | 0.86 (0.68–1.08) | 0.210 | ||
| Model 2 | 1.43(1.13–1.81) | 0.003c | 0.82 (0.63–1.05) | 0.120 | ||
|
|
||||||
| Quartilesd | ||||||
| Model 1 | ||||||
| Q1 | 15/180 | Reference | 19/191 | Reference | ||
| Q2 | 16/181 | 1.21 (0.52–2.57) | 0.620 | 20/183 | 1.11 (0.57–2.17) | 0.740 |
| Q3 | 18/190 | 1.25 (0.60–2.60) | 0.540 | 26/181 | 1.57 (0.83–2.96) | 0.160 |
| Q4 | 28/191 | 2.02 (1.03–3.95) | 0.040c | 12/187 | 0.63 (0.29–1.38) | 0.230 |
| Model 2 | ||||||
| Q1 | 15/180 | Reference | 19/191 | Reference | ||
| Q2 | 16/181 | 1.39 (0.58–3.31) | 0.450 | 20/183 | 0.81 (0.39–1.69) | 0.570 |
| Q3 | 18/190 | 1.72 (0.75–3.94) | 0.200 | 26/181 | 1.38 (0.71–2.76) | 0.330 |
| Q4 | 28/191 | 2.81 (1.31–6.02) | 0.008c | 12/187 | 0.45 (0.20–1.04) | 0.062 |
A worsening glucose tolerance was defined as either moving from the normal group to the impaired fasting glucose (IFG) or diabetes mellitus (DM) group, or from the IFG group to the DM group. Model 1, adjusted for sex; Model 2, adjusted for Model 1+smoking, exercise habits, energy and alcohol intake, education level, body mass index, and fasting glucose. P values were calculated using multivariate logistic regression for continuous variables and quartile categories.
AhRL, arylhydrocarbon receptor ligand activity; MIS-ATP, mitochondrial inhibiting substance activity measured by intracellular ATP content; OR, odds ratio; CI, confidence interval.
a All variables were transformed to the standard deviation-scale;
b Number of subjects with worsening glucose tolerance over 5 years/total subjects;
c P values <0.05 were considered significant;
d The lowest quartile Q1 was used as the reference group.
- 1. Kylin ES. Studien ueber das hypertonie-hyperglykämie-hyperurikämie syndrome. Zentralblatt fur Innere Medizin 1923;44:105–27.
- 2. Cornier MA, Dabelea D, Hernandez TL, Lindstrom RC, Steig AJ, Stob NR, et al. The metabolic syndrome. Endocr Rev 2008;29:777–822.ArticlePubMedPMC
- 3. Reaven GM. Banting lecture 1988: role of insulin resistance in human disease. Diabetes 1988;37:1595–607.ArticlePubMed
- 4. Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med 1998;15:539–53.ArticlePubMed
- 5. Gore AC, Chappell VA, Fenton SE, Flaws JA, Nadal A, Prins GS, et al. EDC-2: The Endocrine Society’s second scientific statement on endocrine-disrupting chemicals. Endocr Rev 2015;36:E1–150.ArticlePubMedPMC
- 6. World Health Organization-United Nations Environment Programme (WHO-UNEP). State of the science of endocrine disrupting chemicals 2012 summary for decision-makers [Internet] Geneva: WHO; 2013 [cited 2021 Mar 22]. Available from: http://www.who.int/ceh/publications/endocrine/en/index.html.
- 7. Lee DH, Lee IK, Jin SH, Steffes M, Jacobs DR Jr. Association between serum concentrations of persistent organic pollutants and insulin resistance among nondiabetic adults: results from the National Health and Nutrition Examination Survey 1999–2002. Diabetes Care 2007;30:622–8.ArticlePubMed
- 8. Lind PM, Riserus U, Salihovic S, Bavel Bv, Lind L. An environmental wide association study (EWAS) approach to the metabolic syndrome. Environ Int 2013;55:1–8.ArticlePubMed
- 9. Uemura H, Arisawa K, Hiyoshi M, Kitayama A, Takami H, Sawachika F, et al. Prevalence of metabolic syndrome associated with body burden levels of dioxin and related compounds among Japan’s general population. Environ Health Perspect 2009;117:568–73.ArticlePubMed
- 10. Lee DH, Lind L, Jacobs DR Jr, Salihovic S, van Bavel B, Lind PM. Associations of persistent organic pollutants with abdominal obesity in the elderly: the Prospective Investigation of the Vasculature in Uppsala Seniors (PIVUS) study. Environ Int 2012;40:170–8.ArticlePubMed
- 11. Lee DH, Lee IK, Song K, Steffes M, Toscano W, Baker BA, et al. A strong dose-response relation between serum concentrations of persistent organic pollutants and diabetes: results from the National Health and Examination Survey 1999–2002. Diabetes Care 2006;29:1638–44.ArticlePubMed
- 12. Magliano DJ, Loh VH, Harding JL, Botton J, Shaw JE. Persistent organic pollutants and diabetes: a review of the epidemiological evidence. Diabetes Metab 2014;40:1–14.ArticlePubMed
- 13. Lind L, Zethelius B, Salihovic S, van Bavel B, Lind PM. Circulating levels of perfluoroalkyl substances and prevalent diabetes in the elderly. Diabetologia 2014;57:473–9.ArticlePubMed
- 14. Lind L, Lind PM, Lejonklou MH, Dunder L, Bergman A, Guerrero-Bosagna C, et al. Uppsala consensus statement on environmental contaminants and the global obesity epidemic. Environ Health Perspect 2016;124:A81–3.ArticlePubMedPMC
- 15. Taylor KW, Novak RF, Anderson HA, Birnbaum LS, Blystone C, Devito M, et al. Evaluation of the association between persistent organic pollutants (POPs) and diabetes in epidemiological studies: a national toxicology program workshop review. Environ Health Perspect 2013;121:774–83.ArticlePubMedPMC
- 16. Lind PM, Lind L. Endocrine-disrupting chemicals and risk of diabetes: an evidence-based review. Diabetologia 2018;61:1495–502.ArticlePubMedPMC
- 17. Warner M, Mocarelli P, Brambilla P, Wesselink A, Samuels S, Signorini S, et al. Diabetes, metabolic syndrome, and obesity in relation to serum dioxin concentrations: the Seveso women’s health study. Environ Health Perspect 2013;121:906–11.ArticlePubMedPMC
- 18. Gore AC, Chappell VA, Fenton SE, Flaws JA, Nadal A, Prins GS, et al. Executive summary to EDC-2: The Endocrine Society’s second scientific statement on endocrine-disrupting chemicals. Endocr Rev 2015;36:593–602.ArticlePubMedPMC
- 19. Lind L, Fors N, Hall J, Marttala K, Stenborg A. A comparison of three different methods to evaluate endothelium-dependent vasodilation in the elderly: the Prospective Investigation of the Vasculature in Uppsala Seniors (PIVUS) study. Arterioscler Thromb Vasc Biol 2005;25:2368–75.PubMed
- 20. Park WH, Jun DW, Kim JT, Jeong JH, Park H, Chang YS, et al. Novel cell-based assay reveals associations of circulating serum AhR-ligands with metabolic syndrome and mitochondrial dysfunction. Biofactors 2013;39:494–504.ArticlePubMed
- 21. Park WH, Kang S, Lee HK, Salihovic S, Bavel BV, Lind PM, et al. Relationships between serum-induced AhR bioactivity or mitochondrial inhibition and circulating polychlorinated biphenyls (PCBs). Sci Rep 2017;7:9383.ArticlePubMedPMC
- 22. Beischlag TV, Luis Morales J, Hollingshead BD, Perdew GH. The aryl hydrocarbon receptor complex and the control of gene expression. Crit Rev Eukaryot Gene Expr 2008;18:207–50.ArticlePubMedPMC
- 23. Hao N, Whitelaw ML. The emerging roles of AhR in physiology and immunity. Biochem Pharmacol 2013;86:561–70.ArticlePubMed
- 24. Okey AB. An aryl hydrocarbon receptor odyssey to the shores of toxicology: the Deichmann Lecture, International Congress of Toxicology-XI. Toxicol Sci 2007;98:5–38.ArticlePubMed
- 25. Brennan JC, He G, Tsutsumi T, Zhao J, Wirth E, Fulton MH, et al. Development of species-specific ah receptor-responsive third generation CALUX cell lines with enhanced responsiveness and improved detection limits. Environ Sci Technol 2015;49:11903–12.ArticlePubMedPMC
- 26. Lee HK, Park WH, Kang YC, Kang S, Im S, Park S, et al. Serum biomarkers from cell-based assays for AhRL and MIS strongly predicted the future development of diabetes in a large community-based prospective study in Korea. Sci Rep 2020;10:6339.ArticlePubMedPMC
- 27. Lee DH, Lind PM, Jacobs DR Jr, Salihovic S, van Bavel B, Lind L. Association between background exposure to organochlorine pesticides and the risk of cognitive impairment: a prospective study that accounts for weight change. Environ Int 2016;89–90:179–84.Article
- 28. Kim JT, Kim SS, Jun DW, Hwang YH, Park WH, Pak YK, et al. Serum arylhydrocarbon receptor transactivating activity is elevated in type 2 diabetic patients with diabetic nephropathy. J Diabetes Investig 2013;4:483–91.ArticlePubMedPMC
- 29. Piao Y, Kim HG, Oh MS, Pak YK. Overexpression of TFAM, NRF-1 and myr-AKT protects the MPP(+)-induced mitochondrial dysfunctions in neuronal cells. Biochim Biophys Acta 2012;1820:577–85.ArticlePubMed
- 30. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the third report of The National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 2001;285:2486–97.ArticlePubMed
- 31. Hankinson O. The aryl hydrocarbon receptor complex. Annu Rev Pharmacol Toxicol 1995;35:307–40.ArticlePubMed
- 32. Olsman H, Engwall M, Kammann U, Klempt M, Otte J, van Bavel B, et al. Relative differences in aryl hydrocarbon receptor-mediated response for 18 polybrominated and mixed halogenated dibenzo-p-dioxins and -furans in cell lines from four different species. Environ Toxicol Chem 2007;26:2448–54.ArticlePubMed
- 33. Kiss EA, Vonarbourg C, Kopfmann S, Hobeika E, Finke D, Esser C, et al. Natural aryl hydrocarbon receptor ligands control organogenesis of intestinal lymphoid follicles. Science 2011;334:1561–5.ArticlePubMed
- 34. Doan TQ, Berntsen HF, Verhaegen S, Ropstad E, Connolly L, Igout A, et al. A mixture of persistent organic pollutants relevant for human exposure inhibits the transactivation activity of the aryl hydrocarbon receptor in vitro. Environ Pollut 2019;254(Pt B):113098.ArticlePubMed
- 35. Lim S, Ahn SY, Song IC, Chung MH, Jang HC, Park KS, et al. Chronic exposure to the herbicide, atrazine, causes mitochondrial dysfunction and insulin resistance. PLoS One 2009;4:e5186.ArticlePubMedPMC
- 36. Ruzzin J, Petersen R, Meugnier E, Madsen L, Lock EJ, Lillefosse H, et al. Persistent organic pollutant exposure leads to insulin resistance syndrome. Environ Health Perspect 2010;118:465–71.ArticlePubMed
- 37. Eichbaum K, Brinkmann M, Buchinger S, Reifferscheid G, Hecker M, Giesy JP, et al. In vitro bioassays for detecting dioxin-like activity: application potentials and limits of detection, a review. Sci Total Environ 2014;487:37–48.ArticlePubMed
References
Figure & Data
References
Citations

- An Interactive Online App for Predicting Diabetes via Machine Learning from Environment-Polluting Chemical Exposure Data
Rosy Oh, Hong Kyu Lee, Youngmi Kim Pak, Man-Suk Oh
International Journal of Environmental Research and Public Health.2022; 19(10): 5800. CrossRef - A Novel Aryl Hydrocarbon Receptor Antagonist HBU651 Ameliorates Peripheral and Hypothalamic Inflammation in High-Fat Diet-Induced Obese Mice
Sora Kang, Aden Geonhee Lee, Suyeol Im, Seung Jun Oh, Hye Ji Yoon, Jeong Ho Park, Youngmi Kim Pak
International Journal of Molecular Sciences.2022; 23(23): 14871. CrossRef

 KES
KES
 PubReader
PubReader ePub Link
ePub Link Cite
Cite