Search
- Page Path
- HOME > Search
Review Article
- Adrenal gland
- The Fascinating Interplay between Growth Hormone, Insulin-Like Growth Factor-1, and Insulin
- Eline C. Nijenhuis-Noort, Kirsten A. Berk, Sebastian J. C. M. M. Neggers, Aart J. van der Lely
- Endocrinol Metab. 2024;39(1):83-89. Published online January 9, 2024
- DOI: https://doi.org/10.3803/EnM.2024.101
- 1,667 View
- 125 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - This review intends to provide the reader with a practical overview of several (patho)physiological conditions in which knowledge of the interplay between growth hormone (GH), insulin-like growth factor-1 (IGF-1), and insulin is important. This might help treating physicians in making the right decisions on how to intervene and improve metabolism for the benefit of patients, and to understand why and how metabolism responds in their specific cases. We will specifically address the interplay between GH, IGF-1, and insulin in type 1 and 2 diabetes mellitus, liver cirrhosis, and acromegaly as examples in which this knowledge is truly necessary.
-
Citations
Citations to this article as recorded by- IGF-1 and IGF-2 as Molecules Linked to Causes and Consequences of Obesity from Fetal Life to Adulthood: A Systematic Review
Justyna Szydlowska-Gladysz, Adrianna Edyta Gorecka, Julia Stepien, Izabela Rysz, Iwona Ben-Skowronek
International Journal of Molecular Sciences.2024; 25(7): 3966. CrossRef
- IGF-1 and IGF-2 as Molecules Linked to Causes and Consequences of Obesity from Fetal Life to Adulthood: A Systematic Review

Original Articles
- Diabetes, obesity and metabolism
- Protective Effects of Melatonin in High-Fat Diet-Induced Hepatic Steatosis via Decreased Intestinal Lipid Absorption and Hepatic Cholesterol Synthesis
- Hyungjune Ku, Yeonji Kim, Alvin Lyle Kim, Garam Lee, Youngsik Choi, Bukyung Kim
- Endocrinol Metab. 2023;38(5):557-567. Published online September 1, 2023
- DOI: https://doi.org/10.3803/EnM.2023.1672

- 1,974 View
- 94 Download
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
The preventative effect of melatonin on the development of obesity and the progression of fatty liver under a high-fat diet (HFD) has been well elucidated through previous studies. We investigated the mechanism behind this effect regarding cholesterol biosynthesis and regulation of cholesterol levels.
Methods
Mice were divided into three groups: normal chow diet (NCD); HFD; and HFD and melatonin administration group (HFD+M). We assessed the serum lipid profile, mRNA expression levels of proteins involved in cholesterol synthesis and reabsorption in the liver and nutrient transporters in the intestines, and cytokine levels. Additionally, an in vitro experiment using HepG2 cells was performed.
Results
Expression of hepatic sterol regulatory element-binding protein 2 (SREBP-2), 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMGCR), and low-density lipoprotein receptor (LDLR) demonstrated that melatonin administration significantly reduces hepatic cholesterol synthesis in mice fed an HFD. Expression of intestinal sodium-glucose transporter 1 (SGLT1), glucose transporter 2 (GLUT2), GLUT5, and Niemann-pick C1-like 1 (NPC1L1) demonstrated that melatonin administration significantly reduces intestinal carbohydrate and lipid absorption in mice fed an HFD. There were no differences in local and circulatory inflammatory cytokine levels among the NCD, HFD, and HFD+M group. HepG2 cells stimulated with palmitate showed reduced levels of SREBP, LDLR, and HMGCR indicating these results are due to the direct mechanistic effect of melatonin on hepatocytes.
Conclusion
Collectively, these data indicate the mechanism behind the protective effects of melatonin from weight gain and liver steatosis under HFD is through a reduction in intestinal caloric absorption and hepatic cholesterol synthesis highlighting its potential in the treatment of obesity and fatty liver disease. -
Citations
Citations to this article as recorded by- Influence of dark deprivation on the ultrastructure and mitochondrial apparatus of rat hepatocytes
D Areshidze
Morphology.2024;[Epub] CrossRef
- Influence of dark deprivation on the ultrastructure and mitochondrial apparatus of rat hepatocytes

- Diabetes, obesity and metabolism
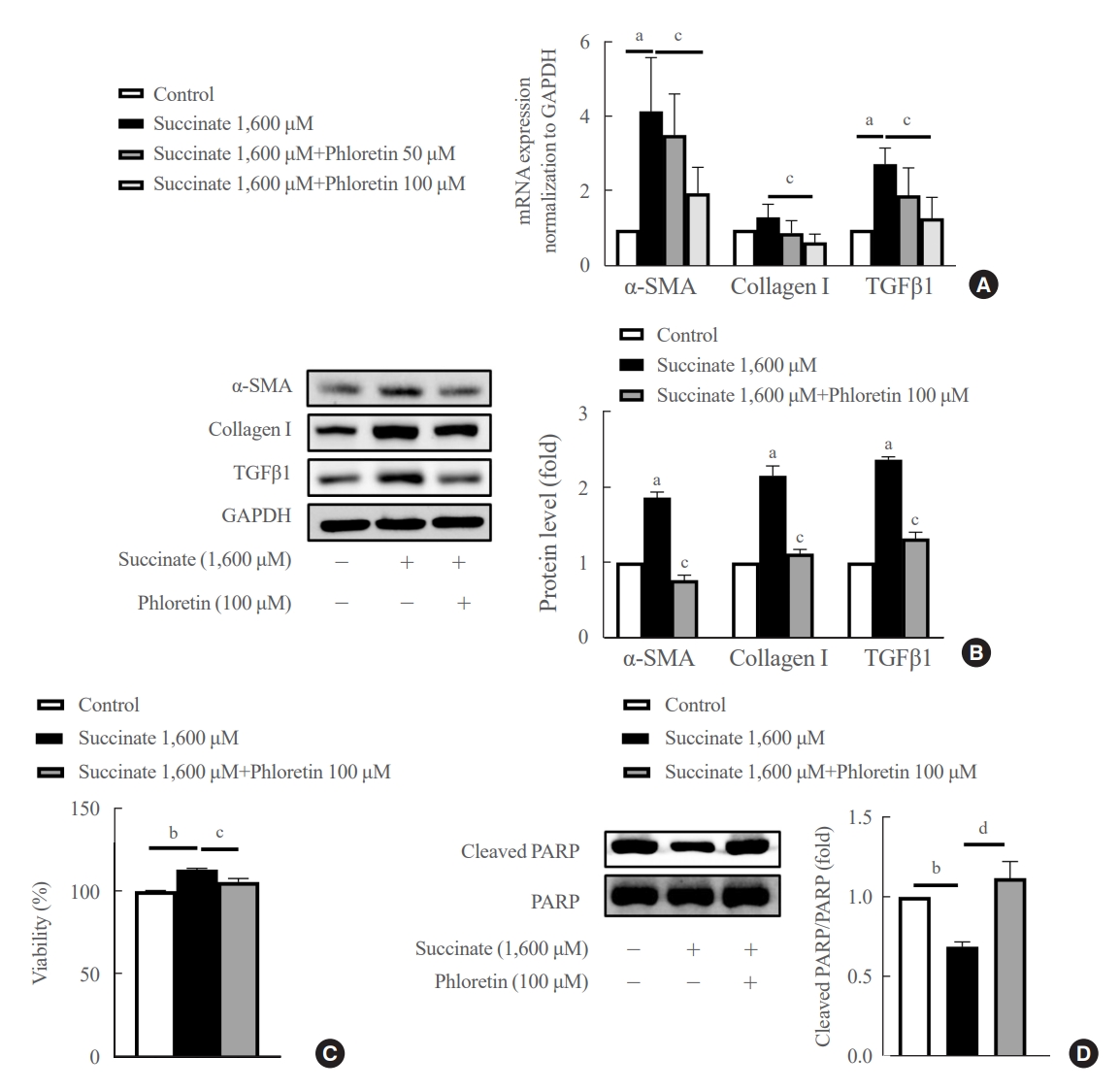
- Phloretin Ameliorates Succinate-Induced Liver Fibrosis by Regulating Hepatic Stellate Cells
- Cong Thuc Le, Giang Nguyen, So Young Park, Hanh Nguyen Dong, Yun Kyung Cho, Jae-Ho Lee, Seung-Soon Im, Dae-Hee Choi, Eun-Hee Cho
- Endocrinol Metab. 2023;38(4):395-405. Published online August 3, 2023
- DOI: https://doi.org/10.3803/EnM.2023.1661
- 1,472 View
- 105 Download
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
Hepatic stellate cells (HSCs) are the major cells which play a pivotal role in liver fibrosis. During injury, extracellular stimulators can induce HSCs transdifferentiated into active form. Phloretin showed its ability to protect the liver from injury, so in this research we would like to investigate the effect of phloretin on succinate-induced HSCs activation in vitro and liver fibrosis in vivo study.
Methods
In in vitro, succinate was used to induce HSCs activation, and then the effect of phloretin on activated HSCs was examined. In in vivo, succinate was used to generated liver fibrosis in mouse and phloretin co-treated to check its protection on the liver.
Results
Phloretin can reduce the increase of fibrogenic markers and inhibits the proliferation, migration, and contraction caused by succinate in in vitro experiments. Moreover, an upregulation of proteins associated with aerobic glycolysis occurred during the activation of HSCs, which was attenuated by phloretin treatment. In in vivo experiments, intraperitoneal injection of phloretin decreased expression of fibrotic and glycolytic markers in the livers of mice with sodium succinate diet-induced liver fibrosis. These results suggest that aerobic glycolysis plays critical role in activation of HSCs and succinate can induce liver fibrosis in mice, whereas phloretin has therapeutic potential for treating hepatic fibrosis.
Conclusion
Intraperitoneal injection of phloretin attenuated succinate-induced hepatic fibrosis and alleviates the succinate-induced HSCs activation.

- Diabetes, obesity and metabolism
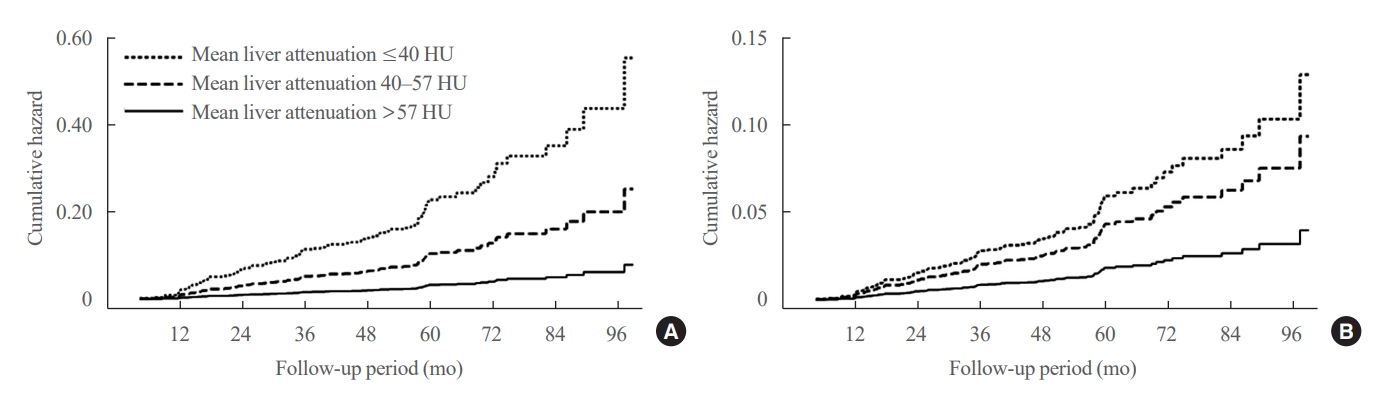
- Greater Severity of Steatosis Is Associated with a Higher Risk of Incident Diabetes: A Retrospective Longitudinal Study
- Ji Min Han, Jung Hwan Cho, Hye In Kim, Sunghwan Suh, Yu-Ji Lee, Jung Won Lee, Kwang Min Kim, Ji Cheol Bae
- Endocrinol Metab. 2023;38(4):418-425. Published online July 12, 2023
- DOI: https://doi.org/10.3803/EnM.2023.1729
- 1,058 View
- 77 Download
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Background
Fatty liver is associated with increased risk of developing type 2 diabetes. We aimed to evaluate whether the severity of hepatic steatosis is associated with incident diabetes.
Methods
We conducted a longitudinal analysis using data from 1,798 participants who underwent a comprehensive health checkup and abdominal computed tomography (CT). We assessed the association between baseline liver attenuation value on non-contrast CT images and risk of incident diabetes. All the participants were categorized into three groups based on the baseline liver attenuation value on non-contrast CT images: without hepatic steatosis (>57 Hounsfield unit [HU]), mild hepatic steatosis (41–57 HU), and moderate to severe hepatic steatosis (≤40 HU).
Results
During a median follow-up period of 5 years, 6.0% of the study participants progressed to diabetes. The incidence of diabetes was 17.3% in the moderate to severe hepatic steatosis group, 9.0% in the mild steatosis group, and 2.9% in those without hepatic steatosis. In a multivariate adjustment model, as compared with participants without hepatic steatosis, those with moderate to severe steatosis had a hazard ratio (HR) of 3.24 (95% confidence interval [CI], 1.64 to 4.2) for the development of diabetes, and those in the mild steatosis group had a HR of 2.33 (95% CI, 1.42 to 3.80). One standard deviation decrease in mean CT attenuation values of the liver was associated with a 40% increase in the development of diabetes (multivariate adjusted HR, 1.40; 95% CI, 1.2 to 1.63).
Conclusion
We found a positive association between severity of hepatic steatosis and risk of incident diabetes. Greater severity of steatosis was associated with a higher risk of incident diabetes.

Brief Report
- Diabetes, obesity and metabolism
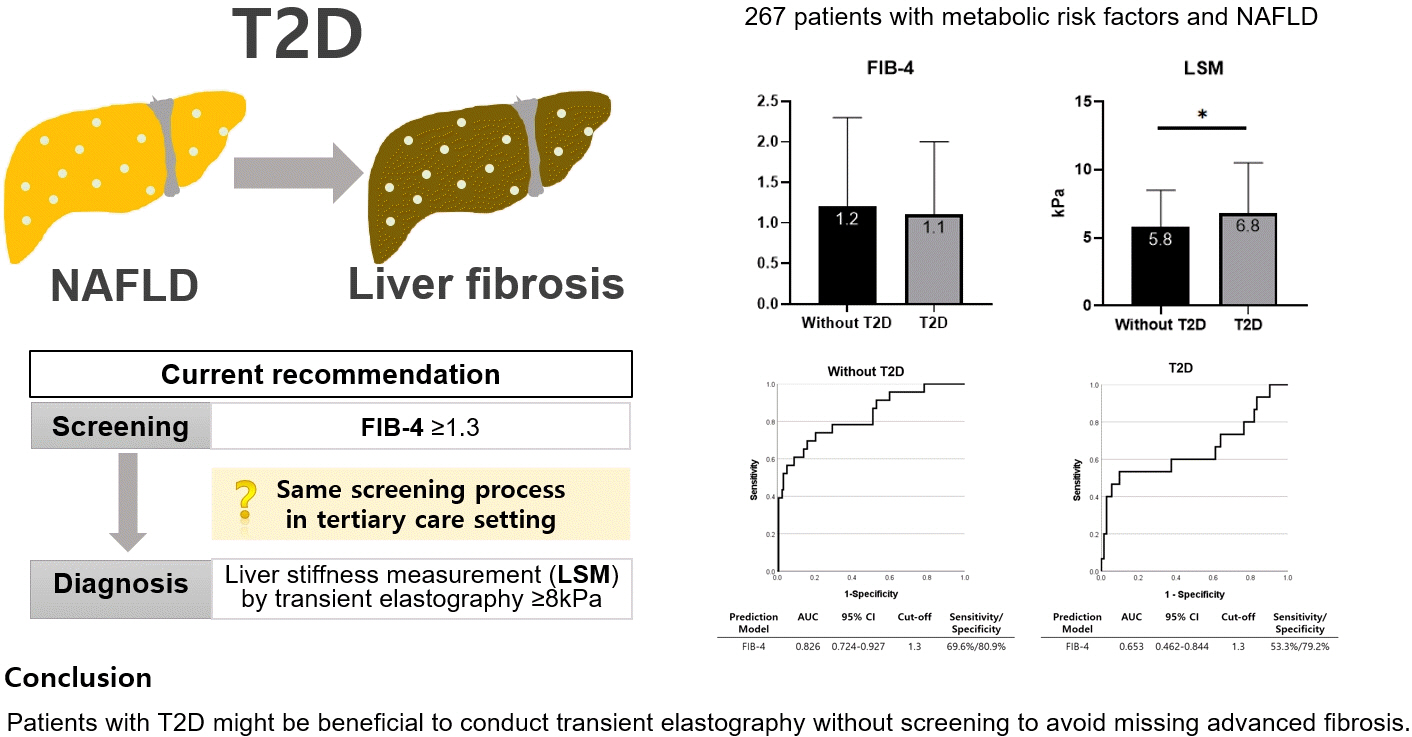
- Performance of Simple Fibrosis Score in Non-Alcoholic Fatty Liver Disease with and without Type 2 Diabetes
- Seung Min Chung, Min Kyu Kang, Jun Sung Moon, Jung Gil Park
- Endocrinol Metab. 2023;38(2):277-281. Published online March 13, 2023
- DOI: https://doi.org/10.3803/EnM.2022.1635
- 1,830 View
- 97 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - This cross-sectional study enrolled 267 patients with metabolic risk factors and established non-alcoholic fatty liver disease in the prospective cohort. The performance of fibrosis-4 (FIB-4) score (≥1.3) to diagnose advanced fibrosis using transient elastography (liver stiffness measurement [LSM] ≥8 kPa) was analyzed. Comparing patients with type 2 diabetes (T2D, n=87) and without (n=180), not FIB-4, but LSM was significantly higher in T2D (P=0.026). The prevalence of advanced fibrosis was 17.2% in T2D and 12.8% in non-T2D. FIB-4 exhibited higher proportion of false negatives in T2D patients (10.9%) than those without (5.2%). The diagnostic performance of FIB-4 was suboptimal in T2D (area under curve [AUC], 0.653; 95% confidence interval [CI], 0.462 to 0.844) compared to that in non-T2D (AUC, 0.826; 95% CI, 0.724 to 0.927). In conclusion, patients with T2D might be beneficial to conduct transient elastography without screening to avoid missing advanced fibrosis.
-
Citations
Citations to this article as recorded by- Prevalence of High and Moderate Risk of Liver Fibrosis Among Patients With Diabetes at a Noncommunicable Diseases (NCD) Clinic in a Primary Healthcare Center in Northern India
Anubhav Mondal, Aninda Debnath, Ghurumourthy Dhandapani, Abhishek Sharma, Shveta Lukhmana, Geeta Yadav
Cureus.2023;[Epub] CrossRef
- Prevalence of High and Moderate Risk of Liver Fibrosis Among Patients With Diabetes at a Noncommunicable Diseases (NCD) Clinic in a Primary Healthcare Center in Northern India

Original Articles
- Diabetes, Obesity and Metabolism
- The Effects of Irisin on the Interaction between Hepatic Stellate Cell and Macrophage in Liver Fibrosis
- Dinh Vinh Do, So Young Park, Giang Thi Nguyen, Dae Hee Choi, Eun-Hee Cho
- Endocrinol Metab. 2022;37(4):620-629. Published online July 22, 2022
- DOI: https://doi.org/10.3803/EnM.2022.1412

- 4,569 View
- 198 Download
- 4 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Hepatic stellate cells (HSCs) are the central players interacting with multiple cell types in liver fibrosis. The crosstalk between HSCs and macrophages has recently become clearer. Irisin, an exercise-responsive myokine, was known to have a potentially protective role in liver and renal fibrosis, especially in connection with stellate cells. This study investigated the effects of irisin on the interaction between HSCs and macrophages.
Methods
Tamm-Horsfall protein-1 (THP-1) human monocytes were differentiated into macrophages, polarized into the inflammatory M1 phenotype with lipopolysaccharide. Lieming Xu-2 (LX-2) cells, human HSCs, were treated with conditioned media (CM) from M1 macrophages, with or without recombinant irisin. HSCs responses to CM from M1 macrophages were evaluated regarding activation, proliferation, wound healing, trans-well migration, contractility, and related signaling pathway.
Results
CM from M1 macrophages significantly promoted HSC proliferation, wound healing, transwell migration, and contractility, but not activation of HSCs. Irisin co-treatment attenuated these responses of HSCs to CM. However, CM and irisin treatment did not induce any changes in HSC activation. Further, irisin co-treatment alleviated CM-induced increase of phopho-protein kinase B (pAKT), matrix metalloproteinase-9 (MMP-9), and tissue inhibitor of metalloproteinases-1 (TIMP-1).
Conclusion
These findings suggested that irisin may play a protective role in the pathogenesis of liver fibrosis, especially when working in the crosstalk between HSCs and macrophages. -
Citations
Citations to this article as recorded by- Matrix metalloproteinases induce extracellular matrix degradation through various pathways to alleviate hepatic fibrosis
Liang Shan, Fengling Wang, Dandan Zhai, Xiangyun Meng, Jianjun Liu, Xiongwen Lv
Biomedicine & Pharmacotherapy.2023; 161: 114472. CrossRef - Potential role of irisin in digestive system diseases
Yueming Zhang, Linxian Zhao, Huan Gao, Jinghui Zhai, Yanqing Song
Biomedicine & Pharmacotherapy.2023; 166: 115347. CrossRef - The effect of sarcopenia and serum myokines on prognosis and survival in cirrhotic patients: a multicenter cross-sectional study
Salih Boga, Abdullah Emre Yildirim, Enver Ucbilek, Ali Riza Koksal, Sevil Tokdemir Sisman, Ibrahim Durak, Ilker Sen, Beril Dogu, Erdinc Serin, Ayse Bolat Ucbilek, Makbule Ozge Yildirim, Sukru Mehmet Erturk, Huseyin Alkim, Canan Alkim
European Journal of Gastroenterology & Hepatology.2022; 34(12): 1261. CrossRef
- Matrix metalloproteinases induce extracellular matrix degradation through various pathways to alleviate hepatic fibrosis

- Diabetes, Obesity and Metabolism
- The Impact of Insulin Resistance on Hepatic Fibrosis among United States Adults with Non-Alcoholic Fatty Liver Disease: NHANES 2017 to 2018
- Ji Cheol Bae, Lauren A. Beste, Kristina M. Utzschneider
- Endocrinol Metab. 2022;37(3):455-465. Published online June 21, 2022
- DOI: https://doi.org/10.3803/EnM.2022.1434
- 4,206 View
- 135 Download
- 9 Web of Science
- 11 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
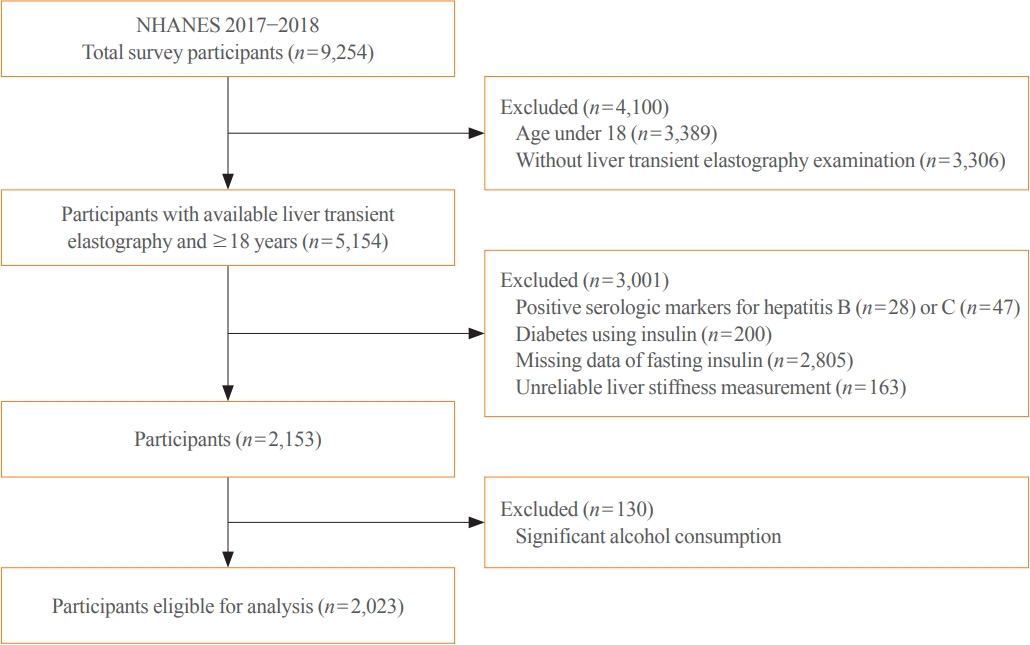
ePub - Background
We aimed to investigate the association of hepatic steatosis with liver fibrosis and to assess the interactive effects of hepatic steatosis and insulin resistance on liver fibrosis in a nationally representative sample of United States adults.
Methods
We conducted a cross-sectional analysis using data from National Health and Nutrition Examination Survey 2017 to 2018, which for the first time included transient elastography to assess liver stiffness and hepatic steatosis. We evaluated the association between hepatic steatosis (using controlled attenuation parameter [CAP]) and clinically significant liver fibrosis (defined as liver stiffness ≥7.5 kPa) using logistic regression with an interaction term for hepatic steatosis and insulin resistance (defined as homeostatic model assessment of insulin resistance ≥3.0).
Results
Among adults undergoing transient elastography (n=2,023), 45.9% had moderate or greater hepatic steatosis and 11.3% had clinically significant liver fibrosis. After adjustment for demographic and metabolic factors, the odds of significant liver fibrosis increased as CAP score rose (odds ratio, 1.35 per standard deviation increment; 95% confidence interval, 1.11 to 1.64). We detected a significant interaction effect between CAP score and insulin resistance on the probability of significant liver fibrosis (P=0.016 for interaction). The probability of significant liver fibrosis increased in the presence of insulin resistance with increasing CAP score, while those without insulin resistance had low probability of significant liver fibrosis, even with high CAP scores.
Conclusion
Individuals with hepatic steatosis had higher odds of fibrosis when insulin resistance was present. Our findings emphasize the importance of the metabolic aspects of the disease on fibrosis risk and suggest a need to better identify patients with metabolic associated fatty liver disease. -
Citations
Citations to this article as recorded by- Association of insulin resistance indicators with hepatic steatosis and fibrosis in patients with metabolic syndrome
Tzu-chia Kuo, Yang-bor Lu, Chieh-lun Yang, Bin Wang, Lin-xin Chen, Ching-ping Su
BMC Gastroenterology.2024;[Epub] CrossRef - No More NAFLD: The Term Is Now MASLD
Ji Cheol Bae
Endocrinology and Metabolism.2024; 39(1): 92. CrossRef - Insulin Resistance/Sensitivity Measures as Screening Indicators of Metabolic-Associated Fatty Liver Disease and Liver Fibrosis
Mohammad E. Khamseh, Mojtaba Malek, Soodeh Jahangiri, Sohrab Nobarani, Azita Hekmatdoost, Marieh Salavatizadeh, Samira Soltanieh, Haleh Chehrehgosha, Hoda Taheri, Zeinab Montazeri, Fereshteh Attaran, Faramarz Ismail-Beigi, Fariba Alaei-Shahmiri
Digestive Diseases and Sciences.2024;[Epub] CrossRef - The association of Neuromedin U levels and non-alcoholic fatty liver disease: A comparative analysis
Murat Keskin, Sercan Avul, Aylin Beyaz, Nizameddin Koca
Heliyon.2024; 10(5): e27291. CrossRef - Oral Insulin Alleviates Liver Fibrosis and Reduces Liver Steatosis in Patients With Metabolic Dysfunction-associated Steatohepatitis and Type 2 Diabetes: Results of Phase II Randomized, Placebo-controlled Feasibility Clinical Trial
Yuval Ishay, Joel Neutel, Yotam Kolben, Ram Gelman, Orly Sneh Arbib, Oliver Lopez, Helena Katchman, Rizwana Mohseni, Miriam Kidron, Yaron Ilan
Gastro Hep Advances.2024; 3(3): 417. CrossRef - Comparative and Predictive Significance of Serum Leptin Levels in Non-alcoholic Fatty Liver Disease
Mehwish Qamar, Abeer Fatima, Ambreen Tauseef, Muhammad I Yousufzai, Ibrahim Liaqat, Qanbar Naqvi
Cureus.2024;[Epub] CrossRef - Greater Severity of Steatosis Is Associated with a Higher Risk of Incident Diabetes: A Retrospective Longitudinal Study
Ji Min Han, Jung Hwan Cho, Hye In Kim, Sunghwan Suh, Yu-Ji Lee, Jung Won Lee, Kwang Min Kim, Ji Cheol Bae
Endocrinology and Metabolism.2023; 38(4): 418. CrossRef - Hepatic T-cell senescence and exhaustion are implicated in the progression of fatty liver disease in patients with type 2 diabetes and mouse model with nonalcoholic steatohepatitis
Byeong Chang Sim, Yea Eun Kang, Sun Kyoung You, Seong Eun Lee, Ha Thi Nga, Ho Yeop Lee, Thi Linh Nguyen, Ji Sun Moon, Jingwen Tian, Hyo Ju Jang, Jeong Eun Lee, Hyon-Seung Yi
Cell Death & Disease.2023;[Epub] CrossRef - Familial clustering of nonalcoholic fatty liver disease in first‐degree relatives of adults with lean nonalcoholic fatty liver disease
Sorachat Niltwat, Chanin Limwongse, Natthinee Charatcharoenwitthaya, Duangkamon Bunditvorapoom, Wimolrak Bandidniyamanon, Phunchai Charatcharoenwitthaya
Liver International.2023; 43(12): 2713. CrossRef - Metabolic Score for Insulin Resistance Is Inversely Related to Incident Advanced Liver Fibrosis in Patients with Non-Alcoholic Fatty Liver Disease
Jun-Hyuk Lee, Yu-Jin Kwon, Kyongmin Park, Hye Sun Lee, Hoon-Ki Park, Jee Hye Han, Sang Bong Ahn
Nutrients.2022; 14(15): 3039. CrossRef - DPP-4 Inhibitor in Type 2 Diabetes Mellitus Patient with Non-Alcoholic Fatty Liver Disease: Achieving Two Goals at Once?
Ji Cheol Bae
Endocrinology and Metabolism.2022; 37(6): 858. CrossRef
- Association of insulin resistance indicators with hepatic steatosis and fibrosis in patients with metabolic syndrome

Review Article
- Diabetes, Obesity and Metabolism
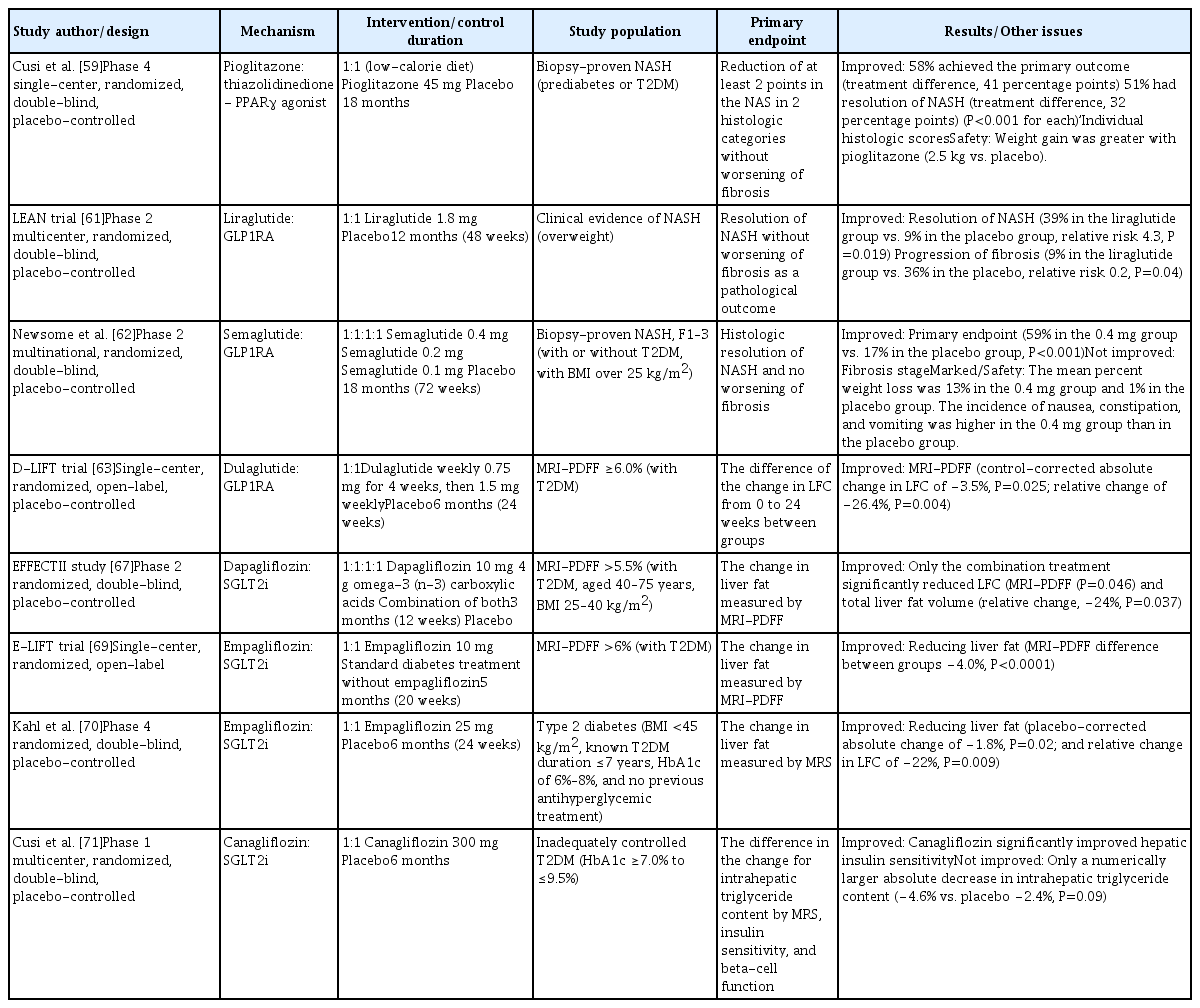
- State-of-the-Art Overview of the Pharmacological Treatment of Non-Alcoholic Steatohepatitis
- Yongin Cho, Yong-ho Lee
- Endocrinol Metab. 2022;37(1):38-52. Published online February 28, 2022
- DOI: https://doi.org/10.3803/EnM.2022.102
- 4,270 View
- 284 Download
- 2 Web of Science
- 3 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - Non-alcoholic fatty liver disease (NAFLD) is the most common cause of chronic liver disease worldwide, and non-alcoholic steatohepatitis (NASH), a subtype of NAFLD, can progress to cirrhosis, hepatocellular carcinoma, and death. Nevertheless, the current treatment for NAFLD/NASH is limited to lifestyle modifications, and no drugs are currently officially approved as treatments for NASH. Many global pharmaceutical companies are pursuing the development of medications for the treatment of NASH, and results from phase 2 and 3 clinical trials have been published in recent years. Here, we review data from these recent clinical trials and reports on the efficacy of newly developed antidiabetic drugs in NASH treatment.
-
Citations
Citations to this article as recorded by- Association of non-alcoholic fatty liver disease with cardiovascular disease and all cause death in patients with type 2 diabetes mellitus: nationwide population based study
Kyung-Soo Kim, Sangmo Hong, Kyungdo Han, Cheol-Young Park
BMJ.2024; : e076388. CrossRef - Mitochondrial Quality Control: Its Role in Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD)
Soyeon Shin, Jaeyoung Kim, Ju Yeon Lee, Jun Kim, Chang-Myung Oh
Journal of Obesity & Metabolic Syndrome.2023; 32(4): 289. CrossRef - Sodium-glucose cotransporter 2 inhibitors for non-alcoholic fatty liver disease in patients with type 2 diabetes mellitus: A nationwide propensity-score matched cohort study
Jinyoung Kim, Kyungdo Han, Bongsung Kim, Ki-Hyun Baek, Ki-Ho Song, Mee Kyoung Kim, Hyuk-Sang Kwon
Diabetes Research and Clinical Practice.2022; 194: 110187. CrossRef
- Association of non-alcoholic fatty liver disease with cardiovascular disease and all cause death in patients with type 2 diabetes mellitus: nationwide population based study

Original Articles
- Diabetes, Obesity and Metabolism
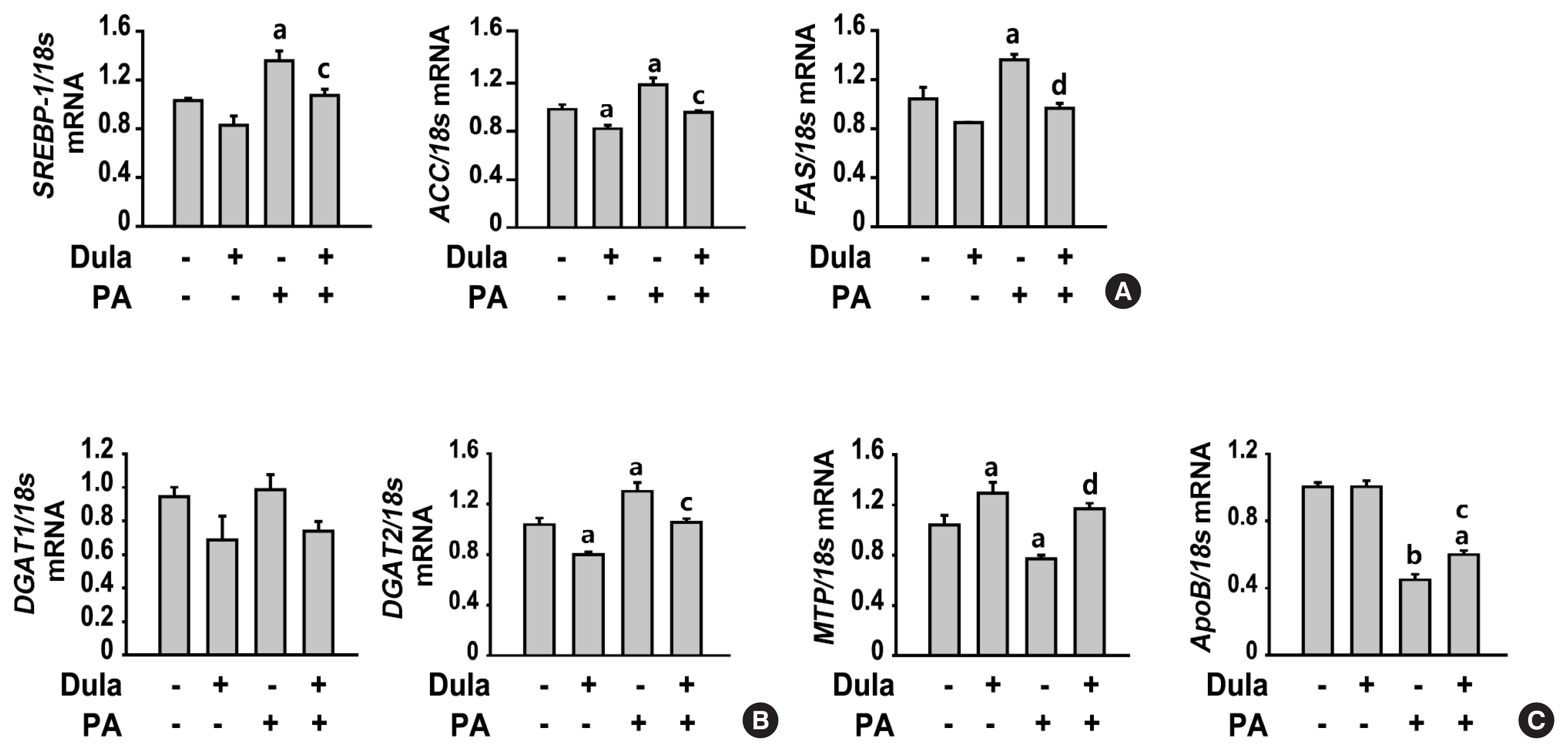
- Dulaglutide Ameliorates Palmitic Acid-Induced Hepatic Steatosis by Activating FAM3A Signaling Pathway
- Jinmi Lee, Seok-Woo Hong, Min-Jeong Kim, Sun Joon Moon, Hyemi Kwon, Se Eun Park, Eun-Jung Rhee, Won-Young Lee
- Endocrinol Metab. 2022;37(1):74-83. Published online February 9, 2022
- DOI: https://doi.org/10.3803/EnM.2021.1293
- 4,901 View
- 235 Download
- 5 Web of Science
- 5 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Dulaglutide, a long-acting glucagon-like peptide-1 receptor agonist (GLP-1RA), has been shown to reduce body weight and liver fat content in patients with type 2 diabetes. Family with sequence similarity 3 member A (FAM3A) plays a vital role in regulating glucose and lipid metabolism. The aim of this study was to determine the mechanisms by which dulaglutide protects against hepatic steatosis in HepG2 cells treated with palmitic acid (PA).
Methods
HepG2 cells were pretreated with 400 μM PA for 24 hours, followed by treatment with or without 100 nM dulaglutide for 24 hours. Hepatic lipid accumulation was determined using Oil red O staining and triglyceride (TG) assay, and the expression of lipid metabolism-associated factor was analyzed using quantitative real time polymerase chain reaction and Western blotting.
Results
Dulaglutide significantly decreased hepatic lipid accumulation and reduced the expression of genes associated with lipid droplet binding proteins, de novo lipogenesis, and TG synthesis in PA-treated HepG2 cells. Dulaglutide also increased the expression of proteins associated with lipolysis and fatty acid oxidation and FAM3A in PA-treated cells. However, exendin-(9-39), a GLP-1R antagonist, reversed the expression of FAM3A, and fatty acid oxidation-associated factors increased due to dulaglutide. In addition, inhibition of FAM3A by siRNA attenuated the reducing effect of dulaglutide on TG content and its increasing effect on regulation of fatty acid oxidation.
Conclusion
These results suggest that dulaglutide could be used therapeutically for improving nonalcoholic fatty liver disease, and its effect could be mediated in part via upregulation of FAM3A expression through a GLP-1R-dependent pathway. -
Citations
Citations to this article as recorded by- GLP-1/GLP-1RAs: New Options for the Drug Treatment of NAFLD
Haoran Jiang, Linquan Zang
Current Pharmaceutical Design.2024; 30(2): 100. CrossRef - GLP-1 Receptor Agonists in Non-Alcoholic Fatty Liver Disease: Current Evidence and Future Perspectives
Riccardo Nevola, Raffaella Epifani, Simona Imbriani, Giovanni Tortorella, Concetta Aprea, Raffaele Galiero, Luca Rinaldi, Raffaele Marfella, Ferdinando Carlo Sasso
International Journal of Molecular Sciences.2023; 24(2): 1703. CrossRef - FAM3A mediates the phenotypic switch of human aortic smooth muscle cells stimulated with oxidised low-density lipoprotein by influencing the PI3K-AKT pathway
Lei Yang, Baoshun Du, Shitao Zhang, Maode Wang
In Vitro Cellular & Developmental Biology - Animal.2023; 59(6): 431. CrossRef - ATP Secretion and Metabolism in Regulating Pancreatic Beta Cell Functions and Hepatic Glycolipid Metabolism
Jing Li, Han Yan, Rui Xiang, Weili Yang, Jingjing Ye, Ruili Yin, Jichun Yang, Yujing Chi
Frontiers in Physiology.2022;[Epub] CrossRef - Targeted therapeutics and novel signaling pathways in non-alcohol-associated fatty liver/steatohepatitis (NAFL/NASH)
Xiaohan Xu, Kyle L. Poulsen, Lijuan Wu, Shan Liu, Tatsunori Miyata, Qiaoling Song, Qingda Wei, Chenyang Zhao, Chunhua Lin, Jinbo Yang
Signal Transduction and Targeted Therapy.2022;[Epub] CrossRef
- GLP-1/GLP-1RAs: New Options for the Drug Treatment of NAFLD

- Diabetes, Obesity and Metabolism
- The Effects of PPAR Agonists on Atherosclerosis and Nonalcoholic Fatty Liver Disease in ApoE−/−FXR−/− Mice
- Yenna Lee, Bo-Rahm Kim, Geun-Hyung Kang, Gwan Jae Lee, Young Joo Park, Haeryoung Kim, Hak Chul Jang, Sung Hee Choi
- Endocrinol Metab. 2021;36(6):1243-1253. Published online December 28, 2021
- DOI: https://doi.org/10.3803/EnM.2021.1100

- 5,629 View
- 159 Download
- 11 Web of Science
- 12 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
Farnesoid X receptor (FXR), a bile acid–activated nuclear receptor, is a potent regulator of glucose and lipid metabolism as well as of bile acid metabolism. Previous studies have demonstrated that FXR deficiency is associated with metabolic derangements, including atherosclerosis and nonalcoholic fatty liver disease (NAFLD), but its mechanism remains unclear. In this study, we investigated the role of FXR in atherosclerosis and NAFLD and the effect of peroxisome proliferator-activated receptor (PPAR) agonists in mouse models with FXR deficiency.
Methods
En face lipid accumulation analysis, liver histology, serum levels of glucose and lipids, and mRNA expression of genes related to lipid metabolism were compared between apolipoprotein E (ApoE)−/− and ApoE−/−FXR−/− mice. The effects of PPARα and PPARγ agonists were also compared in both groups of mice.
Results
Compared with ApoE−/− mice, ApoE−/−FXR−/− mice showed more severe atherosclerosis, hepatic steatosis, and higher levels of serum cholesterol, low-density lipoprotein cholesterol, and triglycerides, accompanied by increased mRNA expression of FAS, ApoC2, TNFα, IL-6 (liver), ATGL, TGH, HSL, and MGL (adipocytes), and decreased mRNA expressions of CPT2 (liver) and Tfam (skeletal muscle). Treatment with a PPARα agonist, but not with a PPARγ agonist, partly reversed atherosclerosis and hepatic steatosis, and decreased plasma triglyceride levels in the ApoE−/−FXR−/− mice, in association with increased mRNA expression of CD36 and FATP and decreased expression of ApoC2 and ApoC3 (liver).
Conclusion
Loss of FXR is associated with aggravation of atherosclerosis and hepatic steatosis in ApoE-deficient mice, which could be reversed by a PPARα agonist through induction of fatty acid uptake, β-oxidation, and triglyceride hydrolysis. -
Citations
Citations to this article as recorded by- Evaluation of the hepatotoxicity of Psoralea corylifolia L. based on a zebrafish model
Shu-Yan Gao, Jing-Cheng Zhao, Qing Xia, Chen Sun, Maimaiti Aili, Ainiwaer Talifu, Shi-Xia Huo, Yun Zhang, Zhi-Jian Li
Frontiers in Pharmacology.2024;[Epub] CrossRef - Advances in management of metabolic dysfunction-associated steatotic liver disease: from mechanisms to therapeutics
Yuxiao Jiang, Lili Wu, Xiaopeng Zhu, Hua Bian, Xin Gao, Mingfeng Xia
Lipids in Health and Disease.2024;[Epub] CrossRef - Mitochondrial carnitine palmitoyltransferase-II dysfunction: A possible novel mechanism for nonalcoholic fatty liver disease in hepatocarcinogenesis
Min Yao, Ping Zhou, Yan-Yan Qin, Li Wang, Deng-Fu Yao
World Journal of Gastroenterology.2023; 29(12): 1765. CrossRef - Emerging Roles of Gut Microbial Modulation of Bile Acid Composition in the Etiology of Cardiovascular Diseases
Tess Yntema, Debby P. Y. Koonen, Folkert Kuipers
Nutrients.2023; 15(8): 1850. CrossRef - The interplay between nonalcoholic fatty liver disease and atherosclerotic cardiovascular disease
Alexandra C. Finney, Sandeep Das, Dhananjay Kumar, M. Peyton McKinney, Bishuang Cai, Arif Yurdagul, Oren Rom
Frontiers in Cardiovascular Medicine.2023;[Epub] CrossRef - Targeting PPARs for therapy of atherosclerosis: A review
Miao Miao, Xue Wang, Tian Liu, Yan-Jie Li, Wen-Qian Yu, Tong-Mei Yang, Shou-Dong Guo
International Journal of Biological Macromolecules.2023; 242: 125008. CrossRef - Cabernet sauvignon dry red wine ameliorates atherosclerosis in mice by regulating inflammation and endothelial function, activating AMPK phosphorylation, and modulating gut microbiota
Xinlong Cheng, Xue Han, Liangfu Zhou, Yasai Sun, Qian Zhou, Xuan Lin, Zhe Gao, Jie Wang, Wen Zhao
Food Research International.2023; 169: 112942. CrossRef - Impacts of dietary lipids derived from animal or vegetable sources on healthy rats
Mostafa M Dalal, Gamal M Edrees, Hanaa A Hassan, Mamdouh Abdel-Mogib, Mai Alaa El-Dein
Egyptian Journal of Basic and Applied Sciences.2023; 10(1): 618. CrossRef - Whey protein hydrolysate alleviated atherosclerosis and hepatic steatosis by regulating lipid metabolism in apoE-/- mice fed a Western diet
Kai Wang, Zixin Fu, Xiaoyi Li, Hui Hong, Xin Zhan, Xiaohong Guo, Yongkang Luo, Yuqing Tan
Food Research International.2022; 157: 111419. CrossRef - Melatonin alleviates PM2.5‐induced glucose metabolism disorder and lipidome alteration by regulating endoplasmic reticulum stress
Zhou Du, Junjie Hu, Lisen Lin, Qingqing Liang, Mengqi Sun, Zhiwei Sun, Junchao Duan
Journal of Pineal Research.2022;[Epub] CrossRef - Lipoprotein Lipase: Is It a Magic Target for the Treatment of Hypertriglyceridemia
Joon Ho Moon, Kyuho Kim, Sung Hee Choi
Endocrinology and Metabolism.2022; 37(4): 575. CrossRef - The role of the gut microbiota in health and cardiovascular diseases
Lu Wang, Shiqi Wang, Qing Zhang, Chengqi He, Chenying Fu, Quan Wei
Molecular Biomedicine.2022;[Epub] CrossRef
- Evaluation of the hepatotoxicity of Psoralea corylifolia L. based on a zebrafish model

Review Article
- Diabetes, Obesity and Metabolism
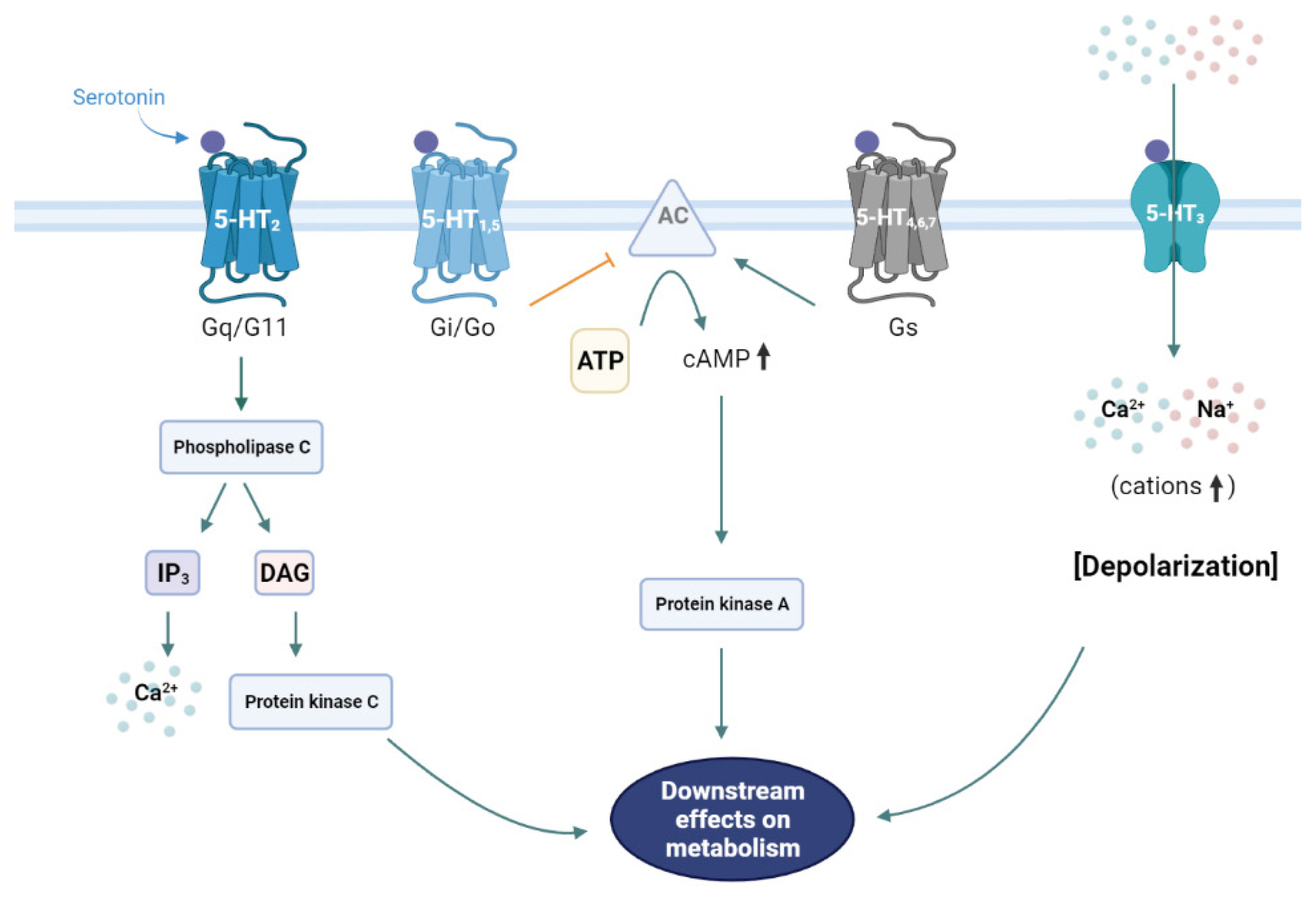
- Serotonergic Regulation of Hepatic Energy Metabolism
- Jiwon Park, Wooju Jeong, Chahyeon Yun, Hail Kim, Chang-Myung Oh
- Endocrinol Metab. 2021;36(6):1151-1160. Published online December 16, 2021
- DOI: https://doi.org/10.3803/EnM.2021.1331
- 5,970 View
- 219 Download
- 7 Web of Science
- 7 Crossref
-
 Abstract
Abstract
 PDF
PDF PubReader
PubReader  ePub
ePub - The liver is a vital organ that regulates systemic energy metabolism and many physiological functions. Nonalcoholic fatty liver disease (NAFLD) is the commonest cause of chronic liver disease and end-stage liver failure. NAFLD is primarily caused by metabolic disruption of lipid and glucose homeostasis. Serotonin (5-hydroxytryptamine [5-HT]) is a biogenic amine with several functions in both the central and peripheral systems. 5-HT functions as a neurotransmitter in the brain and a hormone in peripheral tissues to regulate systemic energy homeostasis. Several recent studies have proposed various roles of 5-HT in hepatic metabolism and inflammation using tissue-specific knockout mice and 5-HT-receptor agonists/antagonists. This review compiles the most recent research on the relationship between 5-HT and hepatic metabolism, and the role of 5-HT signaling as a potential therapeutic target in NAFLD.
-
Citations
Citations to this article as recorded by- Maternal nutrient metabolism in the liver during pregnancy
Hongxu Fang, Qingyang Li, Haichao Wang, Ying Ren, Leying Zhang, Ling Yang
Frontiers in Endocrinology.2024;[Epub] CrossRef - Metabolic and Molecular Response to High-Fat Diet Differs between Rats with Constitutionally High and Low Serotonin Tone
Petra Baković, Maja Kesić, Darko Kolarić, Jasminka Štefulj, Lipa Čičin-Šain
International Journal of Molecular Sciences.2023; 24(3): 2169. CrossRef - Roles of gut microbes in metabolic-associated fatty liver disease
Chun-Yao Chen, Han-Chen Ho
Tzu Chi Medical Journal.2023; 35(4): 279. CrossRef - Imidazoles as Serotonin Receptor Modulators for Treatment of Depression: Structural Insights and Structure–Activity Relationship Studies
Kapil Kumar Goel, Somesh Thapliyal, Rajeev Kharb, Gaurav Joshi, Arvind Negi, Bhupinder Kumar
Pharmaceutics.2023; 15(9): 2208. CrossRef - Serotonin in the regulation of systemic energy metabolism
Joon Ho Moon, Chang‐Myung Oh, Hail Kim
Journal of Diabetes Investigation.2022; 13(10): 1639. CrossRef - Involvement of the liver-gut peripheral neural axis in nonalcoholic fatty liver disease pathologies via hepatic HTR2A
Takashi Owaki, Kenya Kamimura, Masayoshi Ko, Itsuo Nagayama, Takuro Nagoya, Osamu Shibata, Chiyumi Oda, Shinichi Morita, Atsushi Kimura, Takeki Sato, Toru Setsu, Akira Sakamaki, Hiroteru Kamimura, Takeshi Yokoo, Shuji Terai
Disease Models & Mechanisms.2022;[Epub] CrossRef - Non-alcoholic fatty liver disease (NAFLD) and mental illness: Mechanisms linking mood, metabolism and medicines
Anwesha Gangopadhyay, Radwa Ibrahim, Karli Theberge, Meghan May, Karen L. Houseknecht
Frontiers in Neuroscience.2022;[Epub] CrossRef
- Maternal nutrient metabolism in the liver during pregnancy

Original Articles
- Diabetes, Obesity and Metabolism
- The Leg Fat to Total Fat Ratio Is Associated with Lower Risks of Non-Alcoholic Fatty Liver Disease and Less Severe Hepatic Fibrosis: Results from Nationwide Surveys (KNHANES 2008–2011)
- Hyun Min Kim, Yong-ho Lee
- Endocrinol Metab. 2021;36(6):1232-1242. Published online November 23, 2021
- DOI: https://doi.org/10.3803/EnM.2021.1087

- 3,810 View
- 130 Download
- 2 Web of Science
- 2 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
The prevalence of non-alcoholic fatty liver disease (NAFLD) has rapidly increased worldwide. The aim of this study was to investigate whether there is an independent relationship between regional fat distribution, especially leg fat mass, and the presence of NAFLD using nationally representative data in Korea.
Methods
This cross-sectional study analyzed data from 14,502 participants in the Korea National Health and Nutrition Examination Survey 2008 to 2011. Total fat mass, leg fat mass, and appendicular skeletal muscle mass were measured by dual-energy X-ray absorptiometry. Validated NAFLD prediction models and scoring systems for hepatic fibrosis were used.
Results
The leg fat to total fat (LF/TF) ratio showed a negative relationship with many factors, including body mass index, waist circumference, blood pressure, fasting blood glucose, and liver enzyme levels. When the LF/TF ratio and indices of hepatic steatosis were stratified by quartiles, the LF/TF ratio showed a negative correlation with the scoring systems that were used. The LF/TF ratio showed better accuracy in predicting NAFLD than total fat mass or leg fat mass alone. After adjusting for various traditional and lifestyle factors, a low LF/TF ratio remained a risk factor for NAFLD. Among NAFLD subjects, the LF/TF ratio showed a negative relationship with hepatic fibrosis.
Conclusion
A lower LF/TF ratio was markedly associated with a higher risk of hepatic steatosis and advanced hepatic fibrosis using various predictive models in a Korean population. Therefore, the LF/TF ratio could be a useful anthropometric parameter to predict NAFLD or advanced hepatic fibrosis. -
Citations
Citations to this article as recorded by- A greater ratio of thigh subcutaneous fat to abdominal fat is associated with protection against non-alcoholic fatty liver disease
Yebei Liang, Peizhu Chen, Siyu Chen, Dan Liu, Fusong Jiang, Zhijun Zhu, Keqing Dong, Li Wei, Xuhong Hou
JHEP Reports.2023; 5(7): 100730. CrossRef - Association between Alcohol Consumption and Metabolic Dysfunction-Associated Steatotic Liver Disease Based on Alcohol Flushing Response in Men: The Korea National Health and Nutrition Examination Survey 2019–2021
Dae Eon Kang, Si Nae Oh
Nutrients.2023; 15(18): 3901. CrossRef
- A greater ratio of thigh subcutaneous fat to abdominal fat is associated with protection against non-alcoholic fatty liver disease

- Diabetes, Obesity and Metabolism
- Changes in Insulin Resistance Index and the Risk of Liver Fibrosis in Patients with Nonalcoholic Fatty Liver Disease without Diabetes: Kangbuk Samsung Health Study
- Dae-Jeong Koo, Mi Yeon Lee, Inha Jung, Sun Joon Moon, Hyemi Kwon, Se Eun Park, Eun-Jung Rhee, Won-Young Lee
- Endocrinol Metab. 2021;36(5):1016-1028. Published online October 21, 2021
- DOI: https://doi.org/10.3803/EnM.2021.1110
- 4,158 View
- 128 Download
- 5 Web of Science
- 7 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
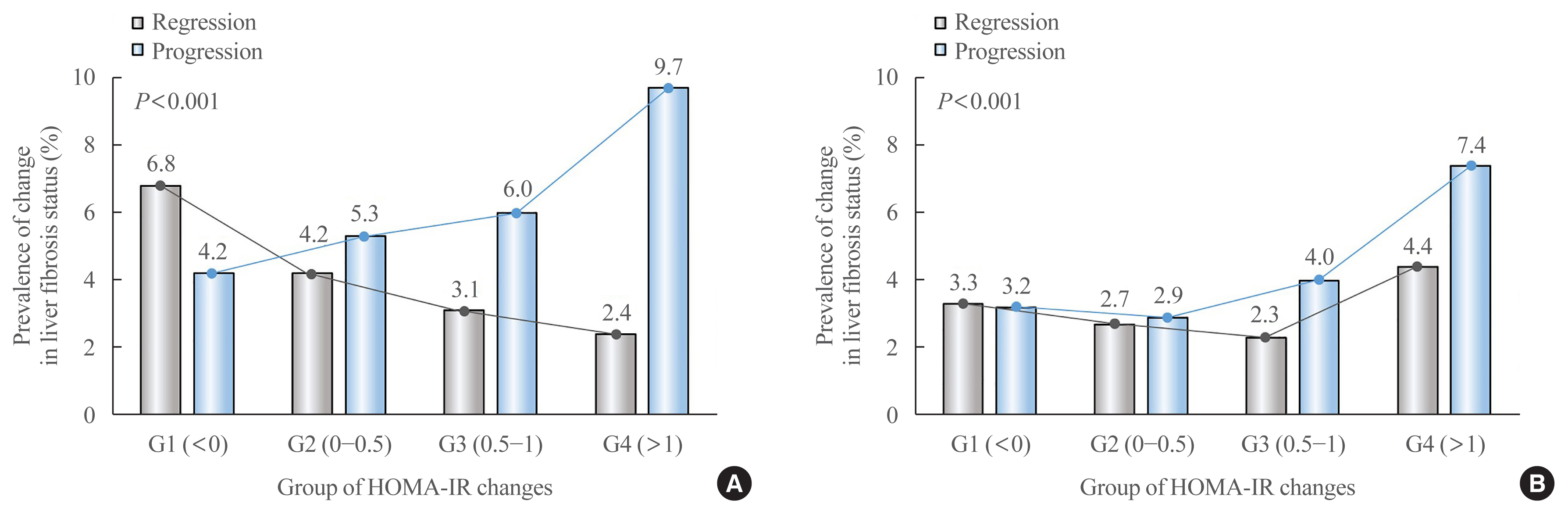
Fibrosis is the most important prognostic factor for nonalcoholic fatty liver disease (NAFLD). Insulin resistance plays a key role of fibrosis progression. We evaluated the association between changes in homeostasis model assessment of insulin resistance (HOMA-IR) values and changes in fibrosis status in NAFLD.
Methods
We analyzed the data of 15,728 participants with NAFLD (86% men, mean age 40.5 years) who had no diabetes at baseline and visited our centers for health check-ups both in 2012 and 2016. The participants were classified into four groups according to the degree of change in HOMA-IR values from baseline to the end of follow-up: G1 (<0), G2 (0–0.50), G3 (0.51–1.00), and G4 (>1.00). NAFLD was assessed by ultrasonography, and fibrosis status was evaluated by the NAFLD fibrosis score (NFS) and the aspartate aminotransferase to platelet ratio index (APRI).
Results
After the 4-year follow-up, the multivariable-adjusted odds ratio (OR) for progression of fibrosis probability increased with increasing HOMA-IR values (OR, 2.25; 95% confidence interval [CI], 1.87 to 2.71 for NFS; and OR, 2.55; 95% CI, 2.05 to 3.18 for APRI, G4). This tendency remained consistent throughout the subgroup analyses, except in those for female sex and a body mass index <25 kg/m2. The OR for regression of fibrosis probability decreased with increasing HOMA-IR values (OR, 0.33; 95% CI, 0.25 to 0.43 for NFS, G4).
Conclusion
Changes in HOMA-IR values were associated with changes in fibrosis status in patients with NAFLD without diabetes, which underscores the role of insulin resistance in liver fibrosis. -
Citations
Citations to this article as recorded by- Insulin Resistance/Sensitivity Measures as Screening Indicators of Metabolic-Associated Fatty Liver Disease and Liver Fibrosis
Mohammad E. Khamseh, Mojtaba Malek, Soodeh Jahangiri, Sohrab Nobarani, Azita Hekmatdoost, Marieh Salavatizadeh, Samira Soltanieh, Haleh Chehrehgosha, Hoda Taheri, Zeinab Montazeri, Fereshteh Attaran, Faramarz Ismail-Beigi, Fariba Alaei-Shahmiri
Digestive Diseases and Sciences.2024; 69(4): 1430. CrossRef - Association between nonalcoholic fatty liver disease and left ventricular diastolic dysfunction: A 7-year retrospective cohort study of 3,496 adults using serial echocardiography
Gyuri Kim, Tae Yang Yu, Jae Hwan Jee, Ji Cheol Bae, Mira Kang, Jae Hyeon Kim
Diabetes & Metabolism.2024; : 101534. CrossRef - Factors Associated with Liver Fibrosis in Chinese Patients with Type 2 Diabetes Mellitus and Non-Alcoholic Fatty Liver Disease
Yu Luo, Cuiyu Wang, Tian Zhang, Xiaoyu He, Jianan Hao, Andong Shen, Hang Zhao, Shuchun Chen, Luping Ren
International Journal of General Medicine.2023; Volume 16: 293. CrossRef - Impact of COVID-19 Lockdown on Non-Alcoholic Fatty Liver Disease and Insulin Resistance in Adults: A before and after Pandemic Lockdown Longitudinal Study
Ángel Arturo López-González, Bárbara Altisench Jané, Luis Masmiquel Comas, Sebastiana Arroyo Bote, Hilda María González San Miguel, José Ignacio Ramírez Manent
Nutrients.2022; 14(14): 2795. CrossRef - Metabolic Score for Insulin Resistance Is Inversely Related to Incident Advanced Liver Fibrosis in Patients with Non-Alcoholic Fatty Liver Disease
Jun-Hyuk Lee, Yu-Jin Kwon, Kyongmin Park, Hye Sun Lee, Hoon-Ki Park, Jee Hye Han, Sang Bong Ahn
Nutrients.2022; 14(15): 3039. CrossRef - Machine learning models including insulin resistance indexes for predicting liver stiffness in United States population: Data from NHANES
Kexing Han, Kexuan Tan, Jiapei Shen, Yuting Gu, Zilong Wang, Jiayu He, Luyang Kang, Weijie Sun, Long Gao, Yufeng Gao
Frontiers in Public Health.2022;[Epub] CrossRef - The crosstalk between insulin resistance and nonalcoholic fatty liver disease/metabolic dysfunction-associated fatty liver disease: a culprit or a consequence?
Dae-Jeong Koo, Won-Young Lee
Cardiovascular Prevention and Pharmacotherapy.2022; 4(4): 132. CrossRef
- Insulin Resistance/Sensitivity Measures as Screening Indicators of Metabolic-Associated Fatty Liver Disease and Liver Fibrosis

- Diabetes, Obesity and Metabolism
- Increased Risk of Nonalcoholic Fatty Liver Disease in Individuals with High Weight Variability
- Inha Jung, Dae-Jeong Koo, Mi Yeon Lee, Sun Joon Moon, Hyemi Kwon, Se Eun Park, Eun-Jung Rhee, Won-Young Lee
- Endocrinol Metab. 2021;36(4):845-854. Published online August 27, 2021
- DOI: https://doi.org/10.3803/EnM.2021.1098
- 4,930 View
- 140 Download
- 7 Web of Science
- 9 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
ePub - Background
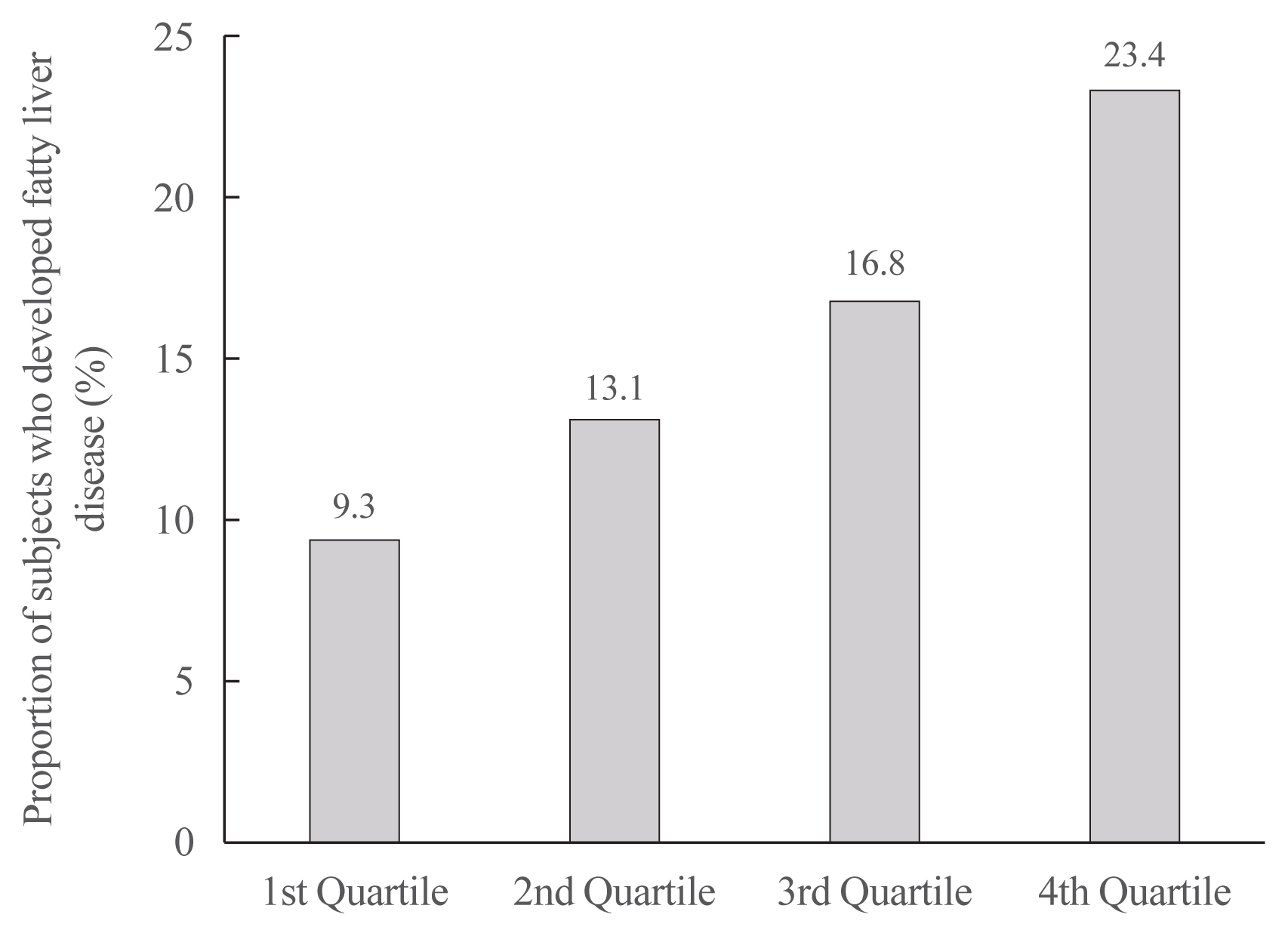
Weight loss through lifestyle modification is recommended for patients with nonalcoholic fatty liver disease (NAFLD). Recent studies have suggested that repeated loss and gain of weight is associated with worse health outcomes. This study aimed to examine the association between weight variability and the risk of NAFLD in patients without diabetes.
Methods
We examined the health-checkup data of 30,708 participants who had undergone serial examinations between 2010 and 2014. Weight variability was assessed using coefficient of variation and the average successive variability of weight (ASVW), which was defined as the sum of absolute weight changes between successive years over the 5-year period divided by 4. The participants were classified according to the baseline body mass index and weight difference over 4 years.
Results
On dividing the participants into four groups according to ASVW quartile groups, those in the highest quartile showed a significantly increased risk of NAFLD compared to those in the lowest quartile (odds ratio [OR], 1.89; 95% confidence interval [CI], 1.63 to 2.19). Among participants without obesity at baseline, individuals with high ASVW showed increased risk of NAFLD (OR, 1.80; 95% CI, 1.61 to 2.01). Participants with increased weight over 4 years and high ASVW demonstrated higher risk of NAFLD compared to those with stable weight and low ASVW (OR, 4.87; 95% CI, 4.29 to 5.53).
Conclusion
Regardless of participant baseline obesity status, high weight variability was associated with an increased risk of developing NAFLD. Our results suggest that further effort is required to minimize weight fluctuations after achieving a desirable body weight. -
Citations
Citations to this article as recorded by- Changes in Macronutrients during Dieting Lead to Weight Cycling and Metabolic Complications in Mouse Model
Anouk Charlot, Anthony Bringolf, Léa Debrut, Joris Mallard, Anne-Laure Charles, Emilie Crouchet, Delphine Duteil, Bernard Geny, Joffrey Zoll
Nutrients.2024; 16(5): 646. CrossRef - Body weight variability and the risk of liver‐related outcomes in type 2 diabetes and steatotic liver disease: a cohort study
Nathalie C. Leite, Claudia R. L. Cardoso, Cristiane A. Villela‐Nogueira, Gil F. Salles
Obesity.2024;[Epub] CrossRef - Weight variability, physical functioning and incident disability in older adults
Katie J. McMenamin, Tamara B. Harris, Joshua F. Baker
Journal of Cachexia, Sarcopenia and Muscle.2023; 14(4): 1648. CrossRef - Dulaglutide Ameliorates Palmitic Acid-Induced Hepatic Steatosis by Activating FAM3A Signaling Pathway
Jinmi Lee, Seok-Woo Hong, Min-Jeong Kim, Sun Joon Moon, Hyemi Kwon, Se Eun Park, Eun-Jung Rhee, Won-Young Lee
Endocrinology and Metabolism.2022; 37(1): 74. CrossRef - Triglyceride and glucose index is a simple and easy‐to‐calculate marker associated with nonalcoholic fatty liver disease
Kyung‐Soo Kim, Sangmo Hong, Hong‐Yup Ahn, Cheol‐Young Park
Obesity.2022; 30(6): 1279. CrossRef - Metabolic (dysfunction)-associated fatty liver disease in individuals of normal weight
Mohammed Eslam, Hashem B. El-Serag, Sven Francque, Shiv K. Sarin, Lai Wei, Elisabetta Bugianesi, Jacob George
Nature Reviews Gastroenterology & Hepatology.2022; 19(10): 638. CrossRef - Impact of COVID-19 Lockdown on Non-Alcoholic Fatty Liver Disease and Insulin Resistance in Adults: A before and after Pandemic Lockdown Longitudinal Study
Ángel Arturo López-González, Bárbara Altisench Jané, Luis Masmiquel Comas, Sebastiana Arroyo Bote, Hilda María González San Miguel, José Ignacio Ramírez Manent
Nutrients.2022; 14(14): 2795. CrossRef - Higher Weight Variability Could Bring You a Fatty Liver
Yeoree Yang, Jae-Hyoung Cho
Endocrinology and Metabolism.2021; 36(4): 766. CrossRef - Autonomic Imbalance Increases the Risk for Non-alcoholic Fatty Liver Disease
Inha Jung, Da Young Lee, Mi Yeon Lee, Hyemi Kwon, Eun-Jung Rhee, Cheol-Young Park, Ki-Won Oh, Won-Young Lee, Sung-Woo Park, Se Eun Park
Frontiers in Endocrinology.2021;[Epub] CrossRef
- Changes in Macronutrients during Dieting Lead to Weight Cycling and Metabolic Complications in Mouse Model

- Diabetes, Obesity and Metabolism
- Non-Laboratory-Based Simple Screening Model for Nonalcoholic Fatty Liver Disease in Patients with Type 2 Diabetes Developed Using Multi-Center Cohorts
- Jiwon Kim, Minyoung Lee, Soo Yeon Kim, Ji-Hye Kim, Ji Sun Nam, Sung Wan Chun, Se Eun Park, Kwang Joon Kim, Yong-ho Lee, Joo Young Nam, Eun Seok Kang
- Endocrinol Metab. 2021;36(4):823-834. Published online August 27, 2021
- DOI: https://doi.org/10.3803/EnM.2021.1074
- 4,445 View
- 137 Download
- 1 Web of Science
- 1 Crossref
-
 Abstract
Abstract
 PDF
PDF Supplementary Material
Supplementary Material PubReader
PubReader  ePub
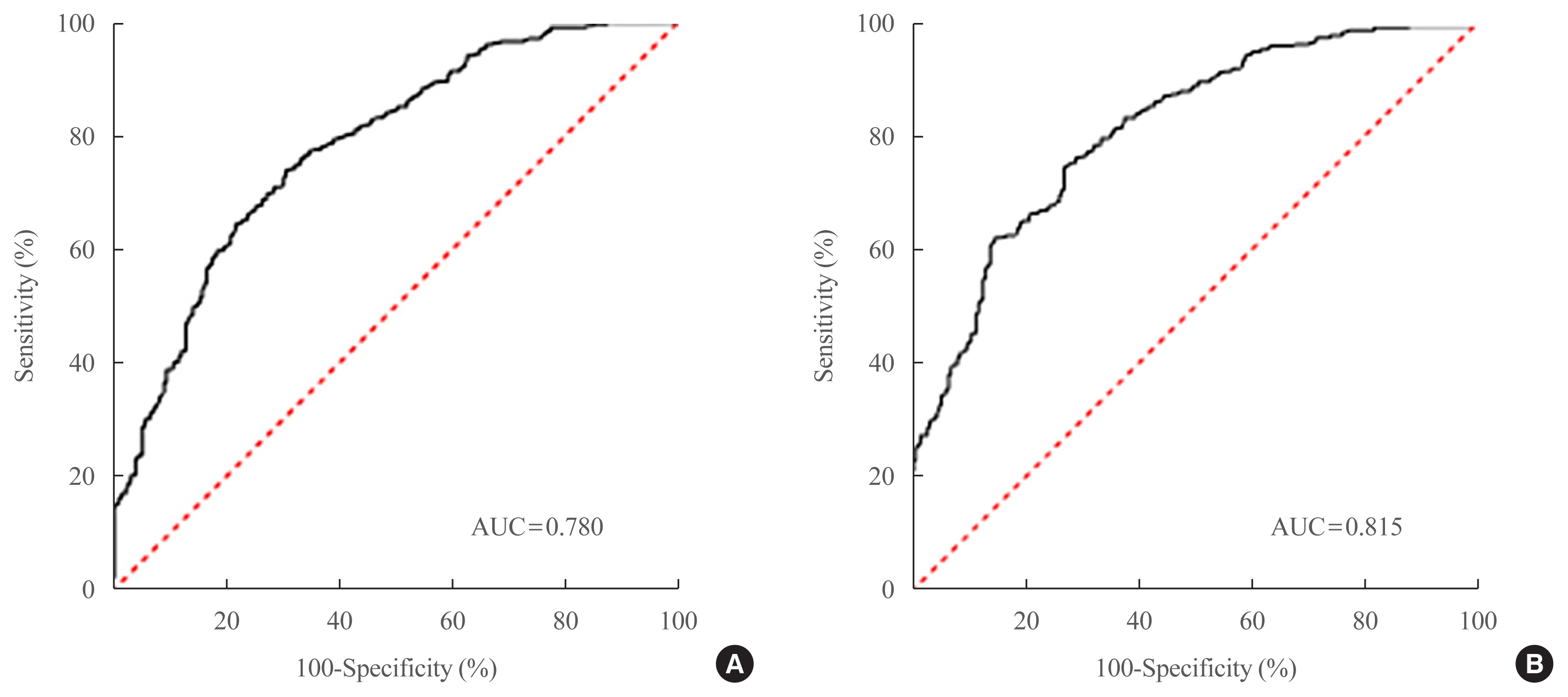
ePub - Background
Nonalcoholic fatty liver disease (NAFLD) is the most prevalent cause of chronic liver disease worldwide. Type 2 diabetes mellitus (T2DM) is a risk factor that accelerates NAFLD progression, leading to fibrosis and cirrhosis. Thus, here we aimed to develop a simple model to predict the presence of NAFLD based on clinical parameters of patients with T2DM.
Methods
A total of 698 patients with T2DM who visited five medical centers were included. NAFLD was evaluated using transient elastography. Univariate logistic regression analyses were performed to identify potential contributors to NAFLD, followed by multivariable logistic regression analyses to create the final prediction model for NAFLD.
Results
Two NAFLD prediction models were developed, with and without serum biomarker use. The non-laboratory model comprised six variables: age, sex, waist circumference, body mass index (BMI), dyslipidemia, and smoking status. For a cutoff value of ≥60, the prediction accuracy was 0.780 (95% confidence interval [CI], 0.743 to 0.817). The second comprehensive model showed an improved discrimination ability of up to 0.815 (95% CI, 0.782 to 0.847) and comprised seven variables: age, sex, waist circumference, BMI, glycated hemoglobin, triglyceride, and alanine aminotransferase to aspartate aminotransferase ratio. Our non-laboratory model showed non-inferiority in the prediction of NAFLD versus previously established models, including serum parameters.
Conclusion
The new models are simple and user-friendly screening methods that can identify individuals with T2DM who are at high-risk for NAFLD. Additional studies are warranted to validate these new models as useful predictive tools for NAFLD in clinical practice. -
Citations
Citations to this article as recorded by- Non-Alcoholic Fatty Liver Disease or Type 2 Diabetes Mellitus—The Chicken or the Egg Dilemma
Marcin Kosmalski, Agnieszka Śliwińska, Józef Drzewoski
Biomedicines.2023; 11(4): 1097. CrossRef
- Non-Alcoholic Fatty Liver Disease or Type 2 Diabetes Mellitus—The Chicken or the Egg Dilemma


 KES
KES

 First
First Prev
Prev



